Cesium (Cs)
alkali-metalSolid
Standard Atomic Weight
132.905452 uElectron configuration
[Xe] 6s1Melting point
28.44 °C (301.59 K)Boiling point
670.85 °C (944 K)Density
1930 kg/m³Oxidation states
−1, +1Electronegativity (Pauling)
0.79Ionization energy (1st)
Discovery year
1860Atomic radius
260 pmDetails
Cesium is a soft alkali metal with very low ionization energy and an unusually low melting point for a metal. It occurs naturally as the single stable isotope ¹³³Cs, chiefly in rare granitic pegmatite minerals. Chemically it is the heaviest stable group 1 element and forms almost exclusively Cs⁺ salts. Its best-known technological role is in the definition of the second, based on the microwave transition of the ¹³³Cs atom.
The metal is characterized by a spectrum containing two bright lines in the blue along with several others in the red, yellow, and green wavelengths. It is silvery white, soft, and ductile. It is the most electropositive and most alkaline element.
Cesium, gallium, and mercury are the only three metals that are liquid at room temperature. Cesium reacts explosively with cold water, and reacts with ice at temperatures above -116C. Cesium hydroxide, the strongest base known, attacks glass.
The name derives from the Latin caesius for "sky blue", which was the colour of the caesium line in the spectroscope. Caesium was discovered by the German chemist Robert Wilhelm Bunsen and the German physicist Gustav Robert Kirchhoff in 1860. It was first isolated by the German chemist Carl Setterberg in 1882.
Cesium was discovered by Robert Wilhelm Bunsen and Gustav Robert Kirchhoff, German chemists, in 1860 through the spectroscopic analysis of Durkheim mineral water. They named cesium after the blue lines they observed in its spectrum. Today, cesium is primarily obtained from the mineral pollucite (CsAlSi2O6). Obtaining pure cesium is difficult since cesium ores are frequently contaminated with rubidium, an element that is chemically similar to cesium. To obtain pure cesium, cesium and rubidium ores are crushed and heated with sodium metal to 650°C, forming an alloy that can then be separated with a process known as fractional distillation. Metallic cesium is too reactive to easily handle and is usually sold in the form of cesium azide (CsN3). Cesium is recovered from cesium azide by heating it.
From the Latin word caesius, sky blue. Cesium was discovered spectroscopically in 1860 by Bunsen and Kirchhoff in mineral water from Durkheim.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
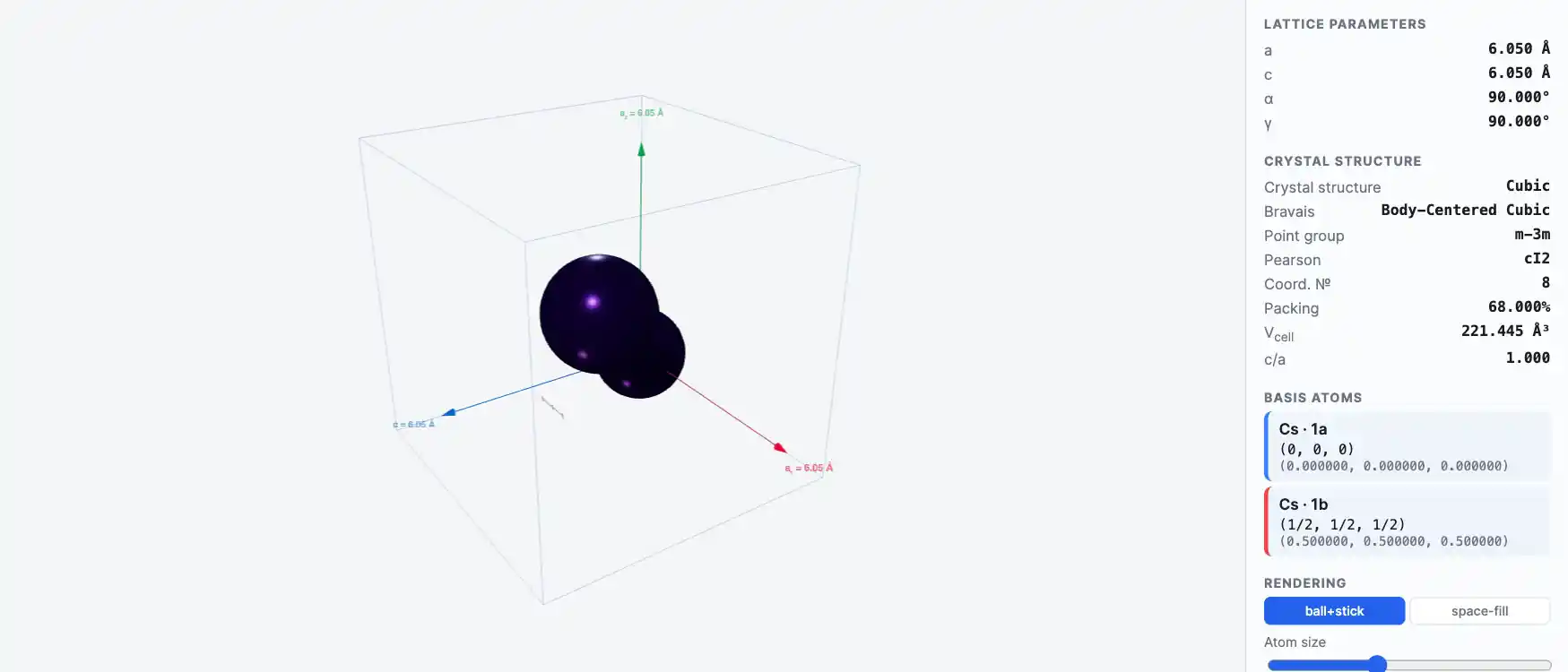
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Cs: 6s¹[Xe] 6s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 6s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 133 Stable | 132.905451961 ± 0.000000008 | 100.0000% | Stable |
Phase / State
Reason: 3.4 °C below melting point (28.44 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 55 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Cs I | 0 | 230 | 42 | 228 |
| Cs II | +1 | 1757 | 2 | 1737 |
| Cs III | +2 | 1010 | 1010 | 1010 |
| Cs IV | +3 | 207 | 0 | 207 |
| Cs V | +4 | 143 | 0 | 143 |
| Cs VI | +5 | 67 | 0 | 67 |
| Cs VII | +6 | 185 | 0 | 185 |
| Cs VIII | +7 | 111 | 13 | 111 |
| Cs IX | +8 | 50 | 16 | 50 |
| Cs X | +9 | 86 | 86 | 86 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Cs I | 0 | 179 |
| Cs II | +1 | 316 |
| Cs III | +2 | 174 |
| Cs IV | +3 | 116 |
| Cs V | +4 | 50 |
| Cs VI | +5 | 32 |
| Cs VII | +6 | 79 |
| Cs VIII | +7 | 55 |
| Cs IX | +8 | 69 |
| Cs X | +9 | 79 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 6 | N/A | 167 pm |
| +1 | 8 | N/A | 174 pm |
| +1 | 9 | N/A | 178 pm |
| +1 | 10 | N/A | 181 pm |
| +1 | 11 | N/A | 185 pm |
| +1 | 12 | N/A | 188 pm |
Compounds
Isotopes (1)
Cesium has more isotopes than any element32with masses ranging from 114 to 145.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 133 Stable | 132.905451961 ± 0.000000008 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 728 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 460.37908 nm | 10000000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 522.70372 nm | 7500000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 592.56312 nm | 5100000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[7/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 556.3024 nm | 3900000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 495.28523 nm | 3700000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 695.54998 nm | 3700000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[7/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 524.93849 nm | 2900000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 504.38026 nm | 2700000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 483.01864 nm | 2500000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 583.11404 nm | 2400000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 537.09876 nm | 2200000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 452.67416 nm | 2000000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 487.00392 nm | 1900000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 427.71303 nm | 1800000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 697.96684 nm | 1600000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 426.470255 nm | 1400000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[5/2] → 5p5.(2P*<3/2>).6d 2[7/2]* | Measured | NIST | |
| 721.9603 nm | 1400000 | Cs III | emission | 5s2.5p5 2P* → 5s2.5p5 2P* | Measured | NIST | |
| 714.95415 nm | 1300000 | Cs II | emission | 5p5.(2P*<1/2>).5d 2[5/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 450.15517 nm | 1200000 | Cs II | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 527.40539 nm | 1100000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 534.91319 nm | 1000000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 653.6445 nm | 1000000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 612.86072 nm | 980000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 672.44659 nm | 960000 | Cs II | emission | 5p5.(2P*<1/2>).5d 2[5/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 664.65663 nm | 880000 | Cs II | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 400.65447 nm | 860000 | Cs III | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).6p 2[3]* | Measured | NIST | |
| 649.55329 nm | 830000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[7/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 497.25963 nm | 820000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[5/2] → 5p5.(2P*<3/2>).7s 2[3/2]* | Measured | NIST | |
| 403.985602 nm | 800000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[5/2] → 5p5.(2P*<3/2>).6d 2[7/2]* | Measured | NIST | |
| 436.329875 nm | 760000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[3/2] → 5p5.(2P*<3/2>).6d 2[5/2]* | Measured | NIST | |
| 388.83763 nm | 740000 | Cs III | emission | 5s2.5p4.(3P<1>).5d 2[3] → 5s2.5p4.(3P<2>).6p 2[3]* | Measured | NIST | |
| 441.02226 nm | 720000 | Cs III | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).6p 2[2]* | Measured | NIST | |
| 450.67197 nm | 720000 | Cs III | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).6p 2[2]* | Measured | NIST | |
| 476.36362 nm | 700000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 520.95813 nm | 650000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 392.55957 nm | 620000 | Cs III | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).6p 2[3]* | Measured | NIST | |
| 442.56759 nm | 560000 | Cs III | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).6p 2[2]* | Measured | NIST | |
| 428.837507 nm | 510000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[3/2] → 5p5.(2P*<3/2>).6d 2[5/2]* | Measured | NIST | |
| 488.00516 nm | 490000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 581.41641 nm | 450000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 395.95055 nm | 420000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 461.61693 nm | 420000 | Cs II | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 453.896566 nm | 410000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[3/2] → 5p5.(2P*<3/2>).6d 2[3/2]* | Measured | NIST | |
| 440.525568 nm | 390000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[1/2] → 5p5.(2P*<3/2>).7s 2[3/2]* | Measured | NIST | |
| 437.30356 nm | 370000 | Cs II | emission | 5p5.(2P*<3/2>).5d 2[1/2]* → 5p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 452.28578 nm | 350000 | Cs III | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).6p 2[2]* | Measured | NIST | |
| 389.698641 nm | 340000 | Cs II | emission | 5p5.(2P*<3/2>).6p 2[1/2] → 5p5.(2P*<3/2>).6d 2[1/2]* | Measured | NIST | |
| 404.34262 nm | 310000 | Cs III | emission | 5s2.5p4.(1D<2>).6s 2[2] → 5s2.5p4.(1D<2>).6p 2[3]* | Measured | NIST | |
| 645.6318 nm | 310000 | Cs III | emission | 5s2.5p4.(3P<2>).7p 2[3]* → 5s2.5p4.(3P<2>).7d 2[4] | Measured | NIST | |
| 607.9854 nm | 300000 | Cs III | emission | 5s2.5p4.(3P<2>).5f 2[2]* → 5s2.5p4.(3P<2>).5g 2[3] | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 301.65 K |
| Boiling point | 944.15 K |
| Critical point (temperature) | 1938.15 K |
| Critical point (pressure) | 9.4 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (12)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.0957 |
| 2 | p | 4.1804 |
| 2 | s | 14.4884 |
| 3 | d | 14.0194 |
| 3 | p | 18.4222 |
| 3 | s | 18.6226 |
| 4 | d | 32.1616 |
| 4 | p | 29.1424 |
| 4 | s | 27.9576 |
| 5 | p | 41.349 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | VI | 181 | ||
| 1 | VIII | 188 | ||
| 1 | IX | 192 | ||
| 1 | X | 195 | ||
| 1 | XI | 199 | ||
| 1 | XII | 202 |
Isotope Decay Modes (74)
| Isotope | Mode | Intensity |
|---|---|---|
| 111 | p | — |
| 112 | p | 100% |
| 112 | A | 0.3% |
| 113 | p | 100% |
| 114 | B+ | 100% |
| 114 | A | 0% |
| 114 | B+p | 8.7% |
| 114 | B+A | 0.2% |
| 115 | B+ | 100% |
| 115 | B+p | 0.1% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.04294 |
| 10.1617 | — | 0.04199 |
| 10.3261 | — | 0.04106 |
| 10.4931 | — | 0.04015 |
| 10.6628 | — | 0.03925 |
| 10.8353 | — | 0.03838 |
| 11.0106 | — | 0.04186 |
| 11.1886 | — | 0.04987 |
| 11.3696 | — | 0.06291 |
| 11.5535 | — | 0.06823 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3×10-4 milligrams per liter
References (1)
Sources
Sources of this element.
Cesium, an alkali metal, occurs in lepidolite, pollucte (a hydrated silicate of aluminum and cesium), and in other sources. One of the world's richest sources of cesium is located at Bernic Lake, Manitoba. The deposits are estimated to contain 300,000 tons of pollucite, averaging 20% cesium.
It can be isolated by elecytrolysis of the fused cyanide and by a number of other methods. Very pure, gas-free cesium can be prepared by thermal decomposition of cesium azide.
References (1)
- [6] Cesium https://periodic.lanl.gov/55.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Cesium.
The element property data was retrieved from publications.