Cerium (Ce)
lanthanideSolid
Standard Atomic Weight
140.116 uElectron configuration
[Xe] 6s2 4f1 5d1Melting point
797.85 °C (1071 K)Boiling point
3423.85 °C (3697 K)Density
6770 kg/m³Oxidation states
+1, +2, +3, +4Electronegativity (Pauling)
1.12Ionization energy (1st)
Discovery year
1801Atomic radius
185 pmDetails
Cerium is the first lanthanide by atomic number and one of the most abundant rare-earth elements in the crust. It is a reactive, electropositive metal whose chemistry is unusual among lanthanides because both Ce³⁺ and Ce⁴⁺ are accessible in ordinary compounds. This Ce³⁺/Ce⁴⁺ redox pair, especially in oxides, makes cerium important in catalysts, polishing materials, glass treatment, and oxygen-storage applications.
Cerium is especially interesting because of its variable electronic structure. The energy of the inner 4f level is nearly the same as that of the outer (valence) electrons, and only small amounts of energy are required to change the relative occupancy of these electronic levels. This gives rise to dual valency states.
For example, a volume change of about 10 percent occurs when cerium is subjected to high pressures or low temperatures. Cesium's valence appears to change from about 3 to 4 when it is cooled or compressed. The low temperature behavior of cerium is complex.
Cerium is an iron-gray lustrous metal. It is malleable, and oxidizes very readily at room temperature, especially in moist air. Except for europium, cerium is the most reactive of the rare-earth metals. It decomposes slowly in cold water and rapidly in hot water.
Alkali solutions and dilute and concentrated acids attack the metal rapidly. The pure metal is likely to ignite if scratched with a knife.
Ceric slats are orange red or yellowish; cerous salts are usually white.
The name derives from the planetoid Ceres, which was discovered by the Italian astronomer Giuseppe Piazzi in 1801 and named for Ceres, the Roman goddess of agriculture and harvest. Two years later, the element cerium was discovered by the German chemist Martin-Heinrich Klaproth, who called it ochroeite earth because of its yellow colour.
Cerium was independently discovered at the same time by the Swedish chemist Jöns Jacob Berzelius and the Swedish mineralogist Wilhelm von Hisinger, who called it ceria. It was first isolated in 1875 by the American mineralogist and chemist William Frances Hillebrand and the American chemist Thomas H. Norton.
Cerium was discovered by Jöns Jacob Berzelius and Wilhelm von Hisinger, Swedish chemists, and independently by Martin Heinrich Klaproth, a German chemist, in 1803. Cerium is the most abundant of the rare earth elements and makes up about 0.0046% of the earth's crust. Today, cerium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements.
Cerium was named for the asteroid Ceres, which was discovered in 1801. The element was discovered two years later in 1803 by Klaproth and by Berzelius and Hisinger. In 1875 Hillebrand and Norton prepared the metal.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
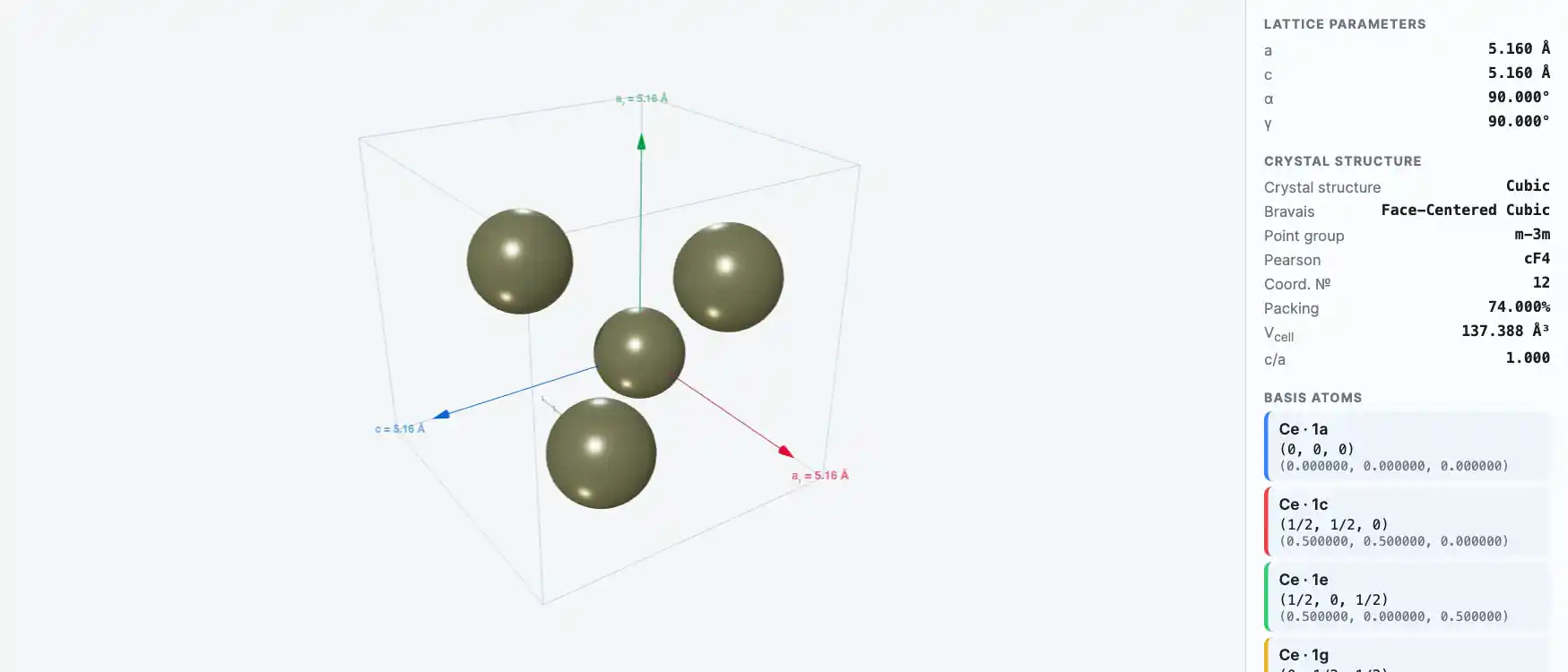
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ce: 4f¹ 5d¹ 6s²[Xe] 4f¹ 5d¹ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹ 5d¹ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 140 Stable | 139.9054431 ± 0.0000023 | 88.4500% | Stable |
Phase / State
Reason: 772.9 °C below melting point (797.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 58 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ce I | 0 | 209 | 67 | 209 |
| Ce II | +1 | 560 | 283 | 560 |
| Ce III | +2 | 262 | 0 | 0 |
| Ce IV | +3 | 27 | 0 | 0 |
| Ce V | +4 | 5 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ce I | 0 | 953 |
| Ce II | +1 | 491 |
| Ce III | +2 | 227 |
| Ce IV | +3 | 17 |
| Ce V | +4 | 12 |
| Ce VI | +5 | 4 |
| Ce VII | +6 | 2 |
| Ce VIII | +7 | 2 |
| Ce IX | +8 | 2 |
| Ce X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 101 pm |
| +3 | 7 | N/A | 107 pm |
| +3 | 8 | N/A | 114.3 pm |
| +3 | 9 | N/A | 119.6 pm |
| +3 | 10 | N/A | 125 pm |
| +3 | 12 | N/A | 134 pm |
| +4 | 6 | N/A | 87 pm |
| +4 | 8 | N/A | 97 pm |
| +4 | 10 | N/A | 97 pm |
| +4 | 12 | N/A | 113.99999999999999 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 140 Stable | 139.9054431 ± 0.0000023 | 88.4500% ± 0.0510% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1072.15 K |
| Boiling point | 3716.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.1519 |
| 2 | p | 4.2176 |
| 2 | s | 15.26 |
| 3 | d | 13.9147 |
| 3 | p | 19.0405 |
| 3 | s | 19.3408 |
| 4 | d | 32.3392 |
| 4 | f | 56.324 |
| 4 | p | 29.3936 |
| 4 | s | 28.32 |
Crystal Radii Detail (10)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 115 | from r^3 vs V plots, | |
| 3 | VII | 121 | estimated, | |
| 3 | VIII | 128.3 | from r^3 vs V plots, | |
| 3 | IX | 133.6 | from r^3 vs V plots, | |
| 3 | X | 139 | ||
| 3 | XII | 148 | calculated, | |
| 4 | VI | 101 | from r^3 vs V plots, | |
| 4 | VIII | 111 | from r^3 vs V plots, | |
| 4 | X | 121 | from r^3 vs V plots, | |
| 4 | XII | 128 |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 119 | B+ | — |
| 119 | B+p | — |
| 120 | B+ | — |
| 120 | B+p | — |
| 121 | B+ | 100% |
| 121 | B+p | 1% |
| 122 | B+ | — |
| 122 | B+p | — |
| 123 | B+ | 100% |
| 123 | B+p | — |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.28369 |
| 10.1617 | — | 1.26389 |
| 10.3261 | — | 1.24441 |
| 10.4931 | — | 1.22522 |
| 10.6628 | — | 1.20632 |
| 10.8353 | — | 1.18772 |
| 11.0106 | — | 1.16941 |
| 11.1886 | — | 1.15138 |
| 11.3696 | — | 1.13362 |
| 11.5535 | — | 1.11614 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
6.65×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.2×10-6 milligrams per liter
References (1)
Sources
Sources of this element.
Cerium is the most abundant so-called rare-earth metals. It is found in a number of minerals including allanite (also known as orthite), monazite, bastnasite, cerite, and samarskite. Monazite and bastnasite are presently the more important sources of cerium.
Large deposits of monazite (found on the beaches of Travancore, India and in river sands in Brazil), allanite (in the western United States), and bastnasite (in Southern California) will supply cerium, thorium, and the other rare-earth metals for many years to come.
Metallic cerium is prepared by metallothermic reduction techniques, such as reducing cerous fluoride with calcium, or using electrolysis of molten cerous chloride or others processes. The metallothermic technique produces high-purity cerium.
References (1)
- [6] Cerium https://periodic.lanl.gov/58.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Cerium.
The element property data was retrieved from publications.