Thulium (Tm)
lanthanideSolid
Standard Atomic Weight
168.93422 uElectron configuration
[Xe] 6s2 4f13Melting point
1544.85 °C (1818 K)Boiling point
1949.85 °C (2223 K)Density
9320 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.25Ionization energy (1st)
Discovery year
1879Atomic radius
175 pmDetails
Thulium is a lanthanide rare-earth metal and the least abundant stable lanthanide in Earth’s crust. It is chemically typical of the series, forming mainly Tm³⁺ compounds with a high affinity for oxygen and halogens. The element is not scarce enough to be unattainable, but it is dispersed and difficult to separate from neighboring lanthanides. Its technological importance is concentrated in isotope sources, specialty lasers, and optical materials rather than bulk structural use.
Thulium can be isolated by reduction of the oxide with lanthanum metal or by calcium reduction of a closed container. The element is silver-gray, soft, malleable, and ductile, and can be cut with a knife. Twenty five isotopes are known, with atomic masses ranging from 152 to 176. Natural thulium, which is 100% 169Tm, is stable.
The name derives from Thule, the earliest name for the northernmost part of the civilized world—Scandinavia. It was discovered in 1879 by the Swedish chemist Per Theodor Cleve in a sample of erbium mineral. Thulium was first isolated by the American chemist Charles James in 1911.
Thulium was discovered by Per Theodor Cleve, a Swedish chemist, in 1879. Cleve used the same method Carl Gustaf Mosander used to discover lanthanum, erbium and terbium, he looked for impurities in the oxides of other rare earth elements. He started with erbia, the oxide of erbium (Er2O3), and removed all of the known contaminants. After further processing, he obtained two new materials, one brown and the other green. Cleve named the brown material holmia and the green material thulia. Holmia is the oxide of the element holmium and thulia is the oxide of the element thulium. Today, thulium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements that can contain as much as 0.007% thulium.
Named after Thule, the earliest name for Scandinavia. Discovered in 1879 by Cleve. Thulium occurs in small quantities along with other rare earths in a number of minerals. It is obtained commercially from monazite, which contains about 0.007% of the element. Thulium is the least abundant of the rare earth elements, but with new sources recently discovered, it is now considered to be about as rare as silver,gold, or cadmium.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
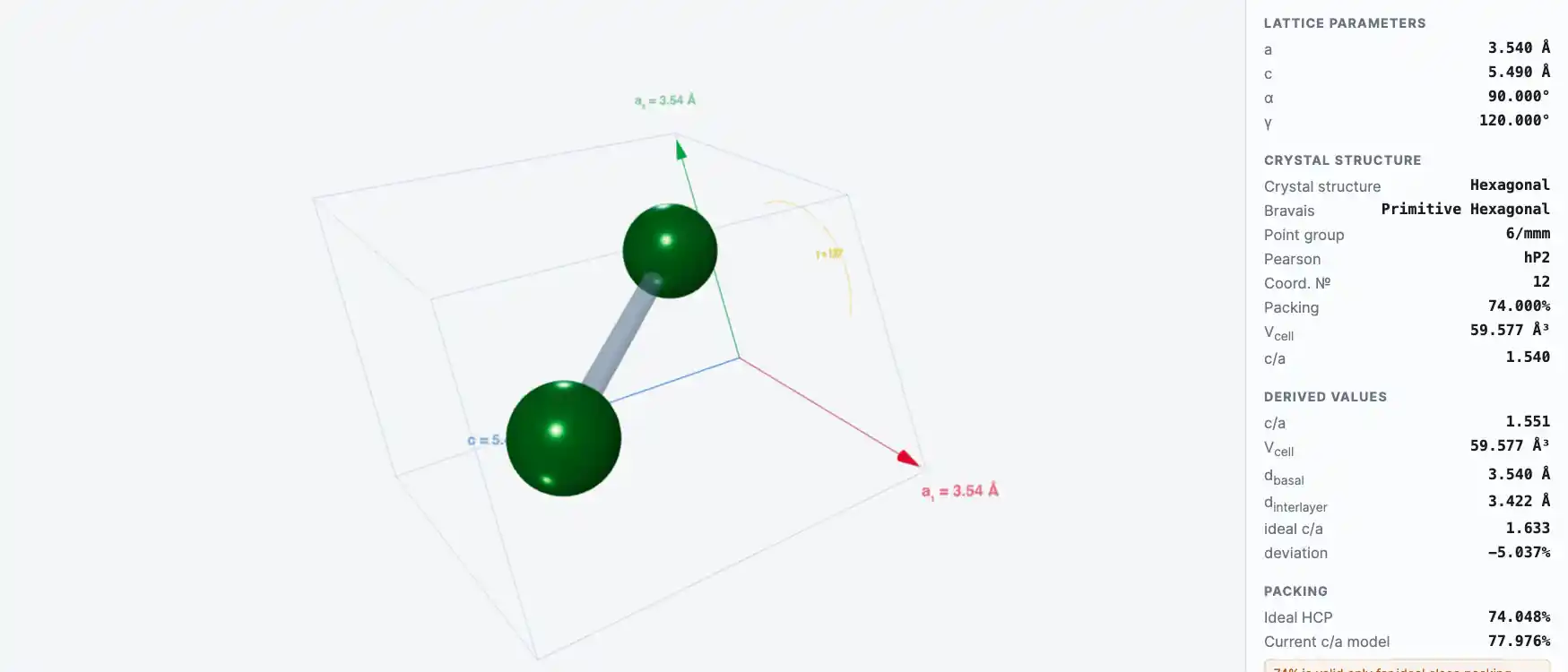
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Tm: 4f¹³ 6s²[Xe] 4f¹³ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹³ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 169 Stable | 168.9342179 ± 0.0000022 | 100.0000% | Stable |
Phase / State
Reason: 1519.8 °C below melting point (1544.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 69 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Tm I | 0 | 538 | 408 | 525 |
| Tm II | +1 | 267 | 13 | 13 |
| Tm III | +2 | 186 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Tm I | 0 | 631 |
| Tm II | +1 | 367 |
| Tm III | +2 | 128 |
| Tm IV | +3 | 8 |
| Tm V | +4 | 2 |
| Tm VI | +5 | 2 |
| Tm VII | +6 | 2 |
| Tm VIII | +7 | 2 |
| Tm IX | +8 | 2 |
| Tm X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 103 pm |
| +2 | 7 | N/A | 109.00000000000001 pm |
| +3 | 6 | N/A | 88 pm |
| +3 | 8 | N/A | 99.4 pm |
| +3 | 9 | N/A | 105.2 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 169 Stable | 168.9342179 ± 0.0000022 | 100.0000% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1818.15 K |
| Boiling point | 2223.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.3437 |
| 2 | p | 4.3588 |
| 2 | s | 18.0522 |
| 3 | d | 13.6257 |
| 3 | p | 20.5239 |
| 3 | s | 21.0816 |
| 4 | d | 36.056 |
| 4 | f | 40.366 |
| 4 | p | 33.012 |
| 4 | s | 31.8624 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 117 | ||
| 2 | VII | 123 | ||
| 3 | VI | 102 | from r^3 vs V plots, | |
| 3 | VIII | 113.4 | from r^3 vs V plots, | |
| 3 | IX | 119.2 | from r^3 vs V plots, |
Isotope Decay Modes (58)
| Isotope | Mode | Intensity |
|---|---|---|
| 144 | p | — |
| 144 | B+ | — |
| 145 | p | 100% |
| 146 | p | 100% |
| 146 | B+ | — |
| 146 | B+p | — |
| 147 | B+ | 85% |
| 147 | p | 15% |
| 148 | B+ | 100% |
| 148 | B+p | — |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.17918 |
| 10.1617 | — | 0.18356 |
| 10.3261 | — | 0.18805 |
| 10.4931 | — | 0.19265 |
| 10.6628 | — | 0.19737 |
| 10.8353 | — | 0.2022 |
| 11.0106 | — | 0.20714 |
| 11.1886 | — | 0.21221 |
| 11.3696 | — | 0.21741 |
| 11.5535 | — | 0.22273 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5.2×10-1 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.7×10-7 milligrams per liter
References (1)
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Thulium.
The element property data was retrieved from publications.