Gold (Au)
transition-metalSolid
Standard Atomic Weight
196.966569 uElectron configuration
[Xe] 6s1 4f14 5d10Melting point
1064.18 °C (1337.33 K)Boiling point
2855.85 °C (3129 K)Density
1.928200e+4 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +5Electronegativity (Pauling)
2.54Ionization energy (1st)
Discovery year
N/AAtomic radius
135 pmDetails
Gold is a dense, soft transition metal with exceptional resistance to oxidation and corrosion. It is usually found native or alloyed with silver and other precious metals, rather than as common simple ores. Its chemistry is dominated by relativistic effects, which help give the metal its yellow color and influence stable oxidation states. Gold combines high electrical conductivity, malleability, chemical nobility, and cultural value in a way unmatched by most elements.
It is estimated that all the gold in the world, so far refined, could be placed in a single cube 60 ft. on a side. Of all the elements, gold in its pure state is undoubtedly the most beautiful. It is metallic, having a yellow color when in a mass, but when finely divided it may be black, ruby, or purple. The Purple of Cassius is a delicate test for auric gold. It is the most malleable and ductile metal; 1 oz. of gold can be beaten out to 300 ft2. It is a soft metal and is usually alloyed to give it more strength. It is a good conductor of heat and electricity, and is unaffected by air and most reagents.
The name derives from the Sanskrit jval for "shine", the Teutonic word gulth for "shining metal", and the Anglo-Saxon gold of unknown origin. The symbol Au derives from the Latin aurum, for Aurora, the goddess of dawn. Gold was known and highly valued in prehistoric times.
An attractive and highly valued metal, gold has been known for at least 5500 years. Gold is sometimes found free in nature but it is usually found in conjunction with silver, quartz (SiO2), calcite (CaCO3), lead, tellurium, zinc or copper. There is roughly 1 milligram of gold dissolved in every ton of seawater, although extracting it currently costs more than the gold is worth. It has been estimated that all of the gold that has currently been refined could be placed in a cube measuring 20 meters on a side.
Known and highly valued from earliest times, gold is found in nature as the free metal and in tellurides; it is very widely distributed and is almost always associated with quartz or pyrite.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
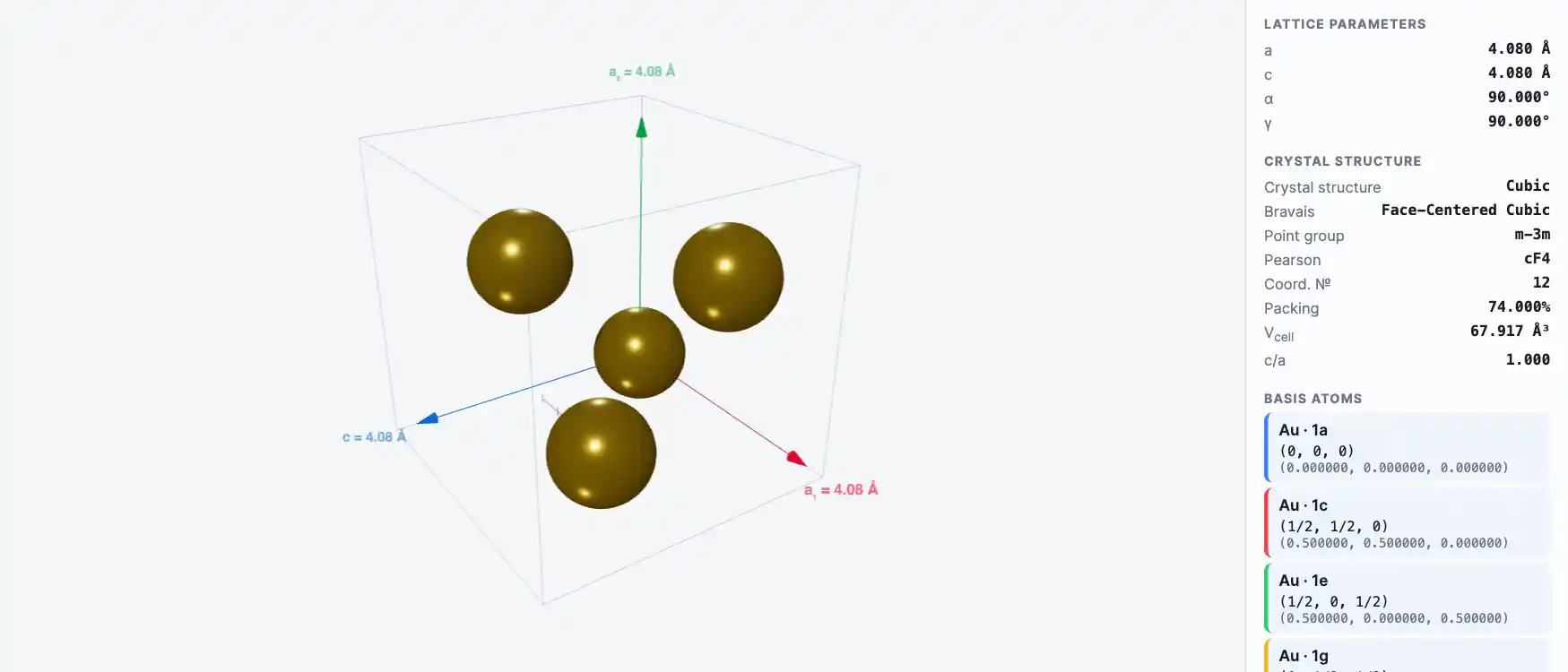
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Au: 4f¹⁴ 5d¹⁰ 6s¹[Xe] 4f¹⁴ 5d¹⁰ 6s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 197 Stable | 196.96656879 ± 0.00000071 | 100.0000% | Stable |
Phase / State
Reason: 1039.2 °C below melting point (1064.18 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 79 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Au I | 0 | 191 | 20 | 90 |
| Au II | +1 | 111 | 0 | 10 |
| Au III | +2 | 150 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Au I | 0 | 75 |
| Au II | +1 | 48 |
| Au III | +2 | 2 |
| Au IV | +3 | 2 |
| Au V | +4 | 2 |
| Au VI | +5 | 2 |
| Au VII | +6 | 2 |
| Au VIII | +7 | 2 |
| Au IX | +8 | 2 |
| Au X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 6 | N/A | 137 pm |
| +3 | 4 | N/A | 68 pm |
| +3 | 6 | N/A | 85 pm |
| +5 | 6 | N/A | 56.99999999999999 pm |
Compounds
Isotopes (1)
The most common gold compounds are auric chloride and chlorauric acid, the latter being used in photography for toning the silver image. Gold has 18 isotopes; 198Au, with a half-life of 2.7 days, is used for treating cancer and other diseases. Disodium aurothiomalate is administered intramuscularly as a treatment for arthritis. A mixture of one part nitric acid with three of hydrochloric acid is called aqua regia (because it dissolved gold, the King of Metals). Gold is available commercially with a purity of 99.999+%. For many years the temperature assigned to the freezing point of gold has been 1063.0C; this has served as a calibration point for the International Temperature Scales (ITS-27 and ITS-48) and the International Practical Temperature Scale (IPTS-48). In 1968, a new International Practical Temperature Scale (IPTS-68) was adopted, which demands that the freezing point of gold be changed to 1064.43C. The specific gravity of gold has been found to vary considerably depending on temperature, how the metal is precipitated, and cold-worked.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 197 Stable | 196.96656879 ± 0.00000071 | 100.0000% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1337.33 K |
| Boiling point | 3109.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.5239 |
| 2 | p | 4.4868 |
| 2 | s | 20.6302 |
| 3 | d | 13.4917 |
| 3 | p | 22.297 |
| 3 | s | 23.2372 |
| 4 | d | 37.472 |
| 4 | f | 38.3504 |
| 4 | p | 35.4532 |
| 4 | s | 34.5868 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | VI | 151 | Ahrens (1952) ionic radius, | |
| 3 | IVSQ | 82 | ||
| 3 | VI | 99 | Ahrens (1952) ionic radius, | |
| 5 | VI | 71 |
Isotope Decay Modes (71)
| Isotope | Mode | Intensity |
|---|---|---|
| 168 | p | — |
| 169 | p | — |
| 169 | A | — |
| 169 | B+ | — |
| 170 | p | 89% |
| 170 | A | 11% |
| 171 | p | 100% |
| 171 | A | — |
| 172 | A | 100% |
| 172 | p | — |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.73645 |
| 10.1617 | — | 1.81425 |
| 10.3261 | — | 1.89553 |
| 10.4931 | — | 1.98045 |
| 10.6628 | — | 2.06919 |
| 10.8353 | — | 2.16029 |
| 11.0106 | — | 2.25522 |
| 11.1886 | — | 2.35433 |
| 11.3696 | — | 2.45698 |
| 11.5535 | — | 2.56237 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
4×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4×10-6 milligrams per liter
References (1)
Sources
Sources of this element.
It occurs in veins and alluvial deposits, and is often separated from rocks and other minerals by mining and panning operations. About two thirds of the world's gold output comes from South Africa, and about two thirds of the total U.S. production comes from South Dakota and Nevada. The metal is recovered from its ores by cyaniding, amalgamating, and smelting processes. Refining is also frequently done by electrolysis. Gold occurs in sea water to the extent of 0.1 to 2 mg/ton, depending on the location where the sample is taken. As yet, no method has been found for recovering gold from sea water profitably.
References (1)
- [6] Gold https://periodic.lanl.gov/79.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Gold.
The element property data was retrieved from publications.