Fluorine (F)
halogenGas
Standard Atomic Weight
18.998403 uElectron configuration
[He] 2s2 2p5Melting point
-219.62 °C (53.53 K)Boiling point
-188.12 °C (85.03 K)Density
1.696 kg/m³Oxidation states
-1Electronegativity (Pauling)
3.98Ionization energy (1st)
Discovery year
1886Atomic radius
50 pmDetails
Fluorine is the lightest halogen and the most electronegative chemical element. In elemental form it occurs as diatomic fluorine, F₂, a highly reactive oxidizing gas. Natural fluorine is almost entirely the stable isotope ¹⁹F and is found in minerals rather than as the free element. Its chemistry underlies fluoride minerals, uranium enrichment chemistry, fluoropolymers, refrigerants, many agrochemicals, and a large share of modern medicinal chemistry.
Fluorine is the most electronegative and reactive of all elements. It is a pale yellow, corrosive gas, which reacts with most organic and inorganic substances. Finely divided metals, glass, ceramics, carbon, and even water burn in fluorine with a bright flame.
Until World War II, there was no commercial production of elemental fluorine. The nuclear bomb project and nuclear energy applications, however, made it necessary to produce large quantities.
The name derives from the Latin fluere for "flow" or "flux" because fluorite (CaF2) was used as a flux in metallurgy owing to its low melting point. It was discovered in hydrofluoric acid by the Swedish pharmacist and chemist Carl-Wilhelm Scheele in 1771, but it was not isolated until 1886 by the French pharmacist and chemist Henri Moissan.
Fluorine is the most reactive of all elements and no chemical substance is capable of freeing fluorine from any of its compounds. For this reason, fluorine does not occur free in nature and was extremely difficult for scientists to isolate. The first recorded use of a fluorine compound dates to around 1670 to a set of instructions for etching glass that called for Bohemian emerald (CaF2). Chemists attempted to identify the material that was capable of etching glass and George Gore was able to produce a small amount of fluorine through an electrolytic process in 1869. Unknown to Gore, fluorine gas explosively combines with hydrogen gas. That is exactly what happened in Gore's experiment when the fluorine gas that formed on one electrode combined with the hydrogen gas that formed on the other electrode. Ferdinand Frederic Henri Moissan, a French chemist, was the first to successfully isolate fluorine in 1886. He did this through the electrolysis of potassium fluoride (KF) and hydrofluoric acid (HF). He also completely isolated the fluorine gas from the hydrogen gas and he built his electrolysis device completely from platinum. His work was so impressive that he was awarded the Nobel Prize for chemistry in 1906. Today, fluorine is still produced through the electrolysis of potassium fluoride and hydrofluoric acid as well as through the electrolysis of molten potassium acid fluoride (KHF2).
From the Latin and French fluere: flow or flux. In 1529, Georigius Agricola described the use of fluorspar as a flux, and as early as 1670 Schwandhard found that glass was etched when exposed to fluorspar treated with acid. Scheele and many later investigators, including Davy, Gay-Lussac, Lavoisier, and Thenard, experimented with hydrofluoric acid, some experiments ending tragically.
The element was finally isolated in 1866 by Moissan after nearly 74 years of continuous effort.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
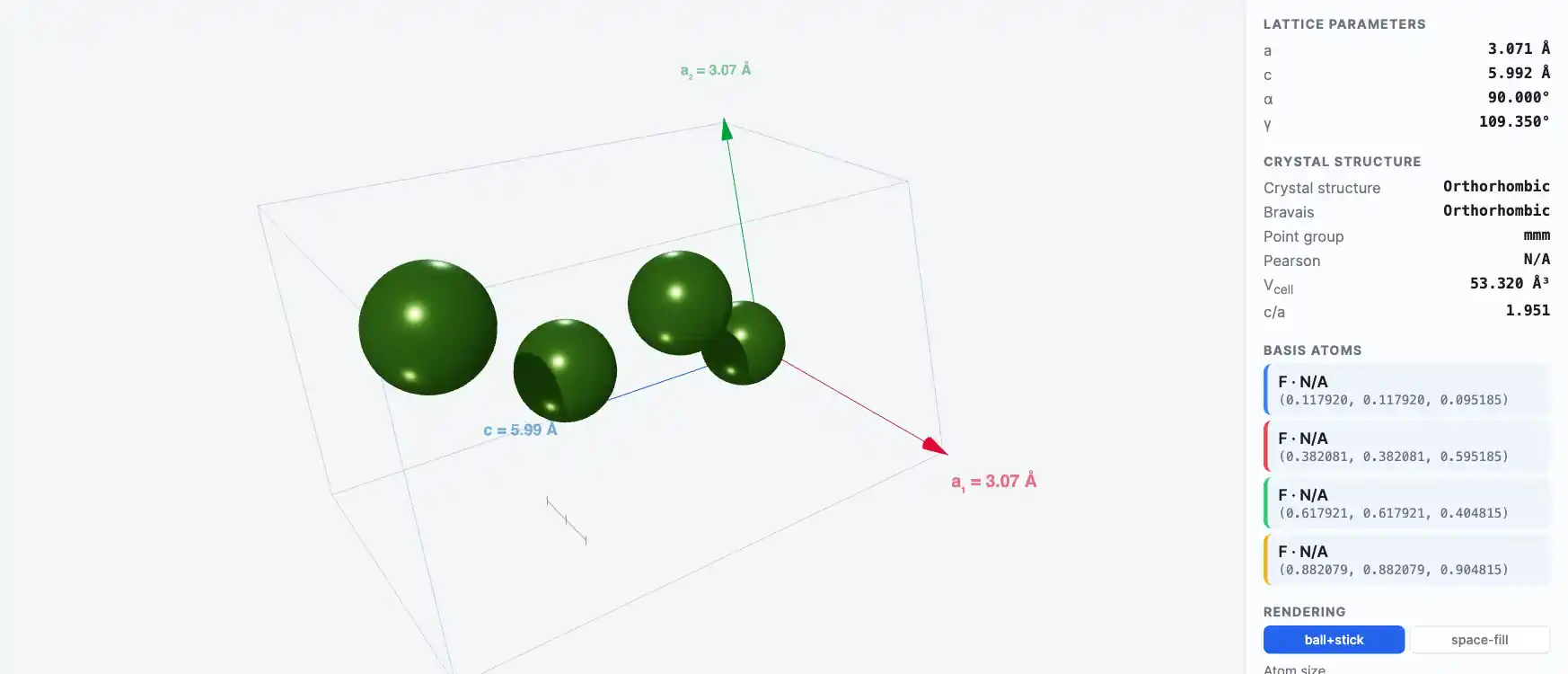
Crystal Structure
N/A
Electronic Structure
Identifiers
Electron Configuration Measured
F: 2s² 2p⁵[He] 2s² 2p⁵1s² 2s² 2p⁵Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 19 Stable | 18.99840316273 ± 0.00000000092 | 100.0000% | Stable |
Phase / State
Reason: 213.1 °C above boiling point (-188.12 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| F I | 0 | 162 | 120 | 162 |
| F II | +1 | 150 | 67 | 67 |
| F III | +2 | 141 | 34 | 34 |
| F IV | +3 | 75 | 30 | 30 |
| F V | +4 | 513 | 472 | 472 |
| F VI | +5 | 269 | 269 | 269 |
| F VII | +6 | 470 | 439 | 470 |
| F VIII | +7 | 128 | 128 | 128 |
| F IX | +8 | 137 | 137 | 137 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| F I | 0 | 303 |
| F II | +1 | 291 |
| F III | +2 | 278 |
| F IV | +3 | 170 |
| F V | +4 | 138 |
| F VI | +5 | 100 |
| F VII | +6 | 77 |
| F VIII | +7 | 151 |
| F IX | +8 | 149 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -1 | 2 | N/A | 128.5 pm |
| -1 | 3 | N/A | 130 pm |
| -1 | 4 | N/A | 131 pm |
| -1 | 6 | N/A | 133 pm |
| +7 | 6 | N/A | 8 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 19 Stable | 18.99840316273 ± 0.00000000092 | 100.0000% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 383.22 nm | N/A | F V | emission | 2s.2p.(3P*).4d 2F* → 2p2.(1D).3d 2D | Measured | NIST | |
| 384.7086 nm | 270 | F II | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).3p 5P | Measured | NIST | |
| 384.9985 nm | 260 | F II | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).3p 5P | Measured | NIST | |
| 385.1668 nm | 250 | F II | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).3p 5P | Measured | NIST | |
| 385.69 nm | N/A | F VI | emission | 1s2.2s.3s 1S → 1s2.2s.3p 3P* | Measured | NIST | |
| 385.712 nm | N/A | F V | emission | 2p2.(3P).3p 4S* → 2p2.(3P).3d 4P | Measured | NIST | |
| 387.086 nm | N/A | F V | emission | 2p2.(3P).3p 4S* → 2p2.(3P).3d 4P | Measured | NIST | |
| 388.508 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 388.6 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8g 2G | Measured | NIST | |
| 388.6 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8g 2G | Measured | NIST | |
| 388.6 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8g 2G | Measured | NIST | |
| 389.2 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8d 2D | Measured | NIST | |
| 389.2 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8d 2D | Measured | NIST | |
| 389.2 nm | N/A | F VII | emission | 1s2.7f 2F* → 1s2.8d 2D | Measured | NIST | |
| 390.229 nm | N/A | F V | emission | 2p2.(3P).3p 4S* → 2p2.(3P).3d 4P | Measured | NIST | |
| 390.45 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 394.51 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2F | Measured | NIST | |
| 394.51 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2F | Measured | NIST | |
| 394.51 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2F | Measured | NIST | |
| 394.518 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 394.736 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 396.08 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 396.113 nm | N/A | F IV | emission | 2s2.2p2 3P → 2s2.2p2 1D | Measured | NIST | |
| 399.6 nm | N/A | F VII | emission | 1s2.7d 2D → 1s2.8p 2P* | Measured | NIST | |
| 399.6 nm | N/A | F VII | emission | 1s2.7d 2D → 1s2.8p 2P* | Measured | NIST | |
| 399.6 nm | N/A | F VII | emission | 1s2.7d 2D → 1s2.8p 2P* | Measured | NIST | |
| 399.692 nm | N/A | F IV | emission | 2s2.2p2 3P → 2s2.2p2 1D | Measured | NIST | |
| 399.692 nm | N/A | F IV | emission | 2s2.2p2 3P → 2s2.2p2 1D | Measured | NIST | |
| 400.26 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 400.942 nm | N/A | F V | emission | 2p2.(3P).3s 4P → 2s.2p.(3P*).4s 4P* | Measured | NIST | |
| 402.4726 nm | 240 | F II | emission | 2s2.2p3.(4S*).3s 3S* → 2s2.2p3.(4S*).3p 3P | Measured | NIST | |
| 402.501 nm | 220 | F II | emission | 2s2.2p3.(4S*).3s 3S* → 2s2.2p3.(4S*).3p 3P | Measured | NIST | |
| 402.5491 nm | 230 | F II | emission | 2s2.2p3.(4S*).3s 3S* → 2s2.2p3.(4S*).3p 3P | Measured | NIST | |
| 405.99 nm | N/A | F IV | emission | 2s2.2p2 3P → 2s2.2p2 1D | Measured | NIST | |
| 405.99 nm | N/A | F IV | emission | 2s2.2p2 3P → 2s2.2p2 1D | Measured | NIST | |
| 410.3075 nm | 190 | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.3213 nm | 170 | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.3506 nm | 200 | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.3713 nm | 180 | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.387 nm | 170 | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.4008 nm | N/A | F II | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).3d 3D* | Measured | NIST | |
| 410.916 nm | 170 | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.03 nm | N/A | F VI | emission | 1s2.2s.3p 1P* → 1s2.2s.3d 3D | Measured | NIST | |
| 411.272 nm | N/A | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.2969 nm | N/A | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.44 nm | N/A | F VI | emission | 1s2.2s.3p 1P* → 1s2.2s.3d 3D | Measured | NIST | |
| 411.6535 nm | 160 | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.699 nm | N/A | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.8752 nm | N/A | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 411.9207 nm | 150 | F II | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2D*).3p 3D | Measured | NIST | |
| 415.775 nm | N/A | F II | emission | 2s2.2p4 1D → 2s2.2p4 1S | Measured | NIST | |
| 423.3 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 424.76 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 426.19 nm | N/A | F V | emission | 2s.2p.(3P*).3p 2D → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 426.28 nm | N/A | F VI | emission | 1s2.2s.3s 1S → 1s2.2s.3p 1P* | Measured | NIST | |
| 427.32 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 427.94 nm | N/A | F V | emission | 2s.2p.(3P*).3p 2D → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 429.9165 nm | 200 | F II | emission | 2s2.2p3.(2D*).3s 1D* → 2s2.2p3.(2D*).3p 1F | Measured | NIST | |
| 432.27 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 433.94 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 435.28 nm | N/A | F V | emission | 2s.2p.(3P*).3p 2D → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 437.11 nm | N/A | F V | emission | 2s.2p.(3P*).3p 2D → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 439.05 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3P* | Measured | NIST | |
| 444.6527 nm | 160 | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 444.6689 nm | N/A | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 444.6721 nm | 170 | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 444.7117 nm | N/A | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 444.7148 nm | N/A | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 444.7188 nm | 180 | F II | emission | 2s2.2p3.(4S*).3d 3D* → 2s2.2p3.(4S*).4f 3F | Measured | NIST | |
| 455.99 nm | N/A | F VI | emission | 1s2.2p.3p 1D → 1s2.2p.3d 1F* | Measured | NIST | |
| 456.45 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 457.45 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 457.96 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 459.81 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 460.57 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 461.08 nm | N/A | F VI | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 463.41 nm | N/A | F VI | emission | 1s2.2p.4p 1P → 1s2.2p.4d 1P* | Measured | NIST | |
| 478.945 nm | N/A | F II | emission | 2s2.2p4 3P → 2s2.2p4 1D | Measured | NIST | |
| 478.945 nm | N/A | F II | emission | 2s2.2p4 3P → 2s2.2p4 1D | Measured | NIST | |
| 486.899 nm | N/A | F II | emission | 2s2.2p4 3P → 2s2.2p4 1D | Measured | NIST | |
| 486.899 nm | N/A | F II | emission | 2s2.2p4 3P → 2s2.2p4 1D | Measured | NIST | |
| 490.456 nm | N/A | F II | emission | 2s2.2p4 3P → 2s2.2p4 1D | Measured | NIST | |
| 507.4 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 507.86 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 509.78 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 510.25 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 511.78 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 515.72 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 517.29 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 517.4 nm | N/A | F VIII | emission | 1s.3s 3S → 1s.3p 3P* | Measured | NIST | |
| 522.95 nm | N/A | F V | emission | 2s.2p.(3P*).4d 4D* → 2p2.(3P).3d 4P | Measured | NIST | |
| 525.1 nm | N/A | F VIII | emission | 1s.3s 3S → 1s.3p 3P* | Measured | NIST | |
| 528.03 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2D | Measured | NIST | |
| 528.03 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2D | Measured | NIST | |
| 528.03 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2D | Measured | NIST | |
| 528.03 nm | N/A | F V | emission | 2p2.(1D).3p 2D* → 2p2.(1D).3d 2D | Measured | NIST | |
| 533.07 nm | N/A | F VI | emission | 1s2.2p.3p 1S → 1s2.2p.3d 1P* | Measured | NIST | |
| 543.21 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 544 nm | N/A | F VII | emission | 1s2.8p 2P* → 1s2.9d 2D | Measured | NIST | |
| 544 nm | N/A | F VII | emission | 1s2.8p 2P* → 1s2.9d 2D | Measured | NIST | |
| 544 nm | N/A | F VII | emission | 1s2.8p 2P* → 1s2.9d 2D | Measured | NIST | |
| 545.91 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 549.84 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 549.99 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 556.76 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 560.85 nm | N/A | F VI | emission | 1s2.2p.3p 3P → 1s2.2p.3d 3D* | Measured | NIST | |
| 568.67 nm | N/A | F V | emission | 2s.2p.(3P*).3s 2P* → 2s.2p.(3P*).3p 2P | Measured | NIST | |
| 572.12 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 572.12 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 572.15 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 573.29 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 573.29 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 573.32 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 573.32 nm | N/A | F III | emission | 2s2.2p3 2D* → 2s2.2p3 2P* | Measured | NIST | |
| 576.14 nm | N/A | F V | emission | 2s.2p.(3P*).3s 2P* → 2s.2p.(3P*).3p 2P | Measured | NIST | |
| 585.63 nm | N/A | F V | emission | 2s.2p.(3P*).3s 2P* → 2s.2p.(3P*).3p 2P | Measured | NIST | |
| 593.55 nm | N/A | F V | emission | 2s.2p.(3P*).3s 2P* → 2s.2p.(3P*).3p 2P | Measured | NIST | |
| 604 nm | N/A | F VII | emission | 1s2.8d 2D → 1s2.9p 2P* | Measured | NIST | |
| 604 nm | N/A | F VII | emission | 1s2.8d 2D → 1s2.9p 2P* | Measured | NIST | |
| 604 nm | N/A | F VII | emission | 1s2.8d 2D → 1s2.9p 2P* | Measured | NIST | |
| 683 nm | N/A | F VII | emission | 1s2.9p 2P* → 1s2.10d 2D | Measured | NIST | |
| 683 nm | N/A | F VII | emission | 1s2.9p 2P* → 1s2.10d 2D | Measured | NIST | |
| 683 nm | N/A | F VII | emission | 1s2.9p 2P* → 1s2.10d 2D | Measured | NIST | |
| 713.8 nm | N/A | F V | emission | 2s.2p.(3P*).4p 2D → 2s.2p.(3P*).4d 2F* | Measured | NIST | |
| 719.4 nm | N/A | F V | emission | 2s.2p.(3P*).4p 2D → 2s.2p.(3P*).4d 2F* | Measured | NIST | |
| 723.4 nm | N/A | F VI | emission | 1s2.2p.4s 1P* → 1s2.2p.4p 1D | Measured | NIST | |
| 728.5 nm | N/A | F VIII | emission | 1s.3s 1S → 1s.3p 1P* | Measured | NIST | |
| 735.8 nm | N/A | F V | emission | 2s.2p.(3P*).4p 2D → 2s.2p.(3P*).4d 2F* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 53.48 K |
| Boiling point | 85.04 K |
| Critical point (temperature) | 144.41 K |
| Critical point (pressure) | 5.17 MPa |
| Triple point (temperature) | 53.48 K |
| Triple point (pressure) | 90 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3499 |
| 2 | p | 3.9 |
| 2 | s | 3.8724 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -1 | II | 114.5 | ||
| -1 | III | 116 | ||
| -1 | IV | 117 | ||
| -1 | VI | 119 | ||
| 7 | VI | 22 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (31)
| Isotope | Mode | Intensity |
|---|---|---|
| 13 | p | — |
| 14 | p | — |
| 15 | p | 100% |
| 16 | p | 100% |
| 17 | B+ | 100% |
| 18 | B+ | 100% |
| 20 | B- | 100% |
| 21 | B- | 100% |
| 22 | B- | 100% |
| 22 | B-n | 11% |
X‑ray Scattering Factors (502)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.05165 |
| 10.1617 | — | 0.05648 |
| 10.3261 | — | 0.06176 |
| 10.4931 | — | 0.06754 |
| 10.6628 | — | 0.07386 |
| 10.8353 | — | 0.08077 |
| 11.0106 | — | 0.08833 |
| 11.1886 | — | 0.09659 |
| 11.3696 | — | 0.10831 |
| 11.5535 | — | 0.12462 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5.85×102 milligrams per kilogram
References (1)
- [5] Fluorine https://education.jlab.org/itselemental/ele009.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.3 milligrams per liter
References (1)
- [5] Fluorine https://education.jlab.org/itselemental/ele009.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Fluorine.
The element property data was retrieved from publications.