Thorium (Th)
actinideSolid
Standard Atomic Weight
232.0377 uElectron configuration
[Rn] 7s2 6d2Melting point
1749.85 °C (2023 K)Boiling point
4787.85 °C (5061 K)Density
1.172000e+4 kg/m³Oxidation states
−1, +1, +2, +3, +4Electronegativity (Pauling)
1.3Ionization energy (1st)
Discovery year
1828Atomic radius
180 pmDetails
Thorium is a naturally occurring actinide metal with atomic number 90. It is weakly radioactive and is found mainly as ²³²Th, an isotope with a very long half-life. Chemically it is dominated by the +4 oxidation state and often resembles the tetravalent lanthanides more than uranium or plutonium. Its main technological interest is as a fertile nuclear material that can be converted to fissile ²³³U in reactors.
When pure, thorium is a silvery-white metal that is air-stable and retains its luster for several months. When contaminated with the oxide, thorium slowly tarnishes in air, becoming gray and finally black. The physical properties of thorium are greatly influenced by the degree of contamination with the oxide. The purest specimens often contain several tenths of a percent of the oxide. High-purity thorium has been made. Pure thorium is soft, very ductile, and can be cold-rolled, swaged, and drawn. Thorium is dimorphic, changing at 1400°C from a cubic to a body-centered cubic structure. Thorium oxide has a melting point of 3300°C, which is the highest of all oxides. Only a few elements, such as tungsten, and a few compounds, such as tantalum carbide, have higher melting points. Thorium is slowly attacked by water, but does not dissolve readily in most common acids, except hydrochloric. Powdered thorium metal is often pyrophoric and should be handled carefully. When heated in air, thorium turnings ignite and burn brilliantly with a white light.
The name derives from Thor, the Scandinavian god of thunder. It was discovered in the mineral thorite (ThSiO4) by the Swedish chemist Jöns Jacob Berzelius in 1828. Thorium was first isolated by the chemists D. Lely, Jr. and L. Hamburger in 1914.
Thorium was discovered by Jöns Jacob Berzelius, a Swedish chemist, in 1828. He discovered it in a sample of a mineral that was given to him by the Reverend Has Morten Thrane Esmark, who suspected that it contained an unknown substance. Esmark's mineral is now known as thorite (ThSiO4). Thorium makes up about 0.0007% of the earth's crust and is primarily obtained from thorite, thorianite (ThO2) and monazite ((Ce, La, Th, Nd, Y)PO4).
Morten Esmark found a black mineral on Løvøya island, Norway and gave a sample to his father Jens Esmark, a noted mineralogist. The elder Esmark was not able to identify it and sent a sample to Swedish chemist Jöns Jakob Berzelius for examination in 1828. In 1829 Berzelius determined that it contained a new element, which he named thorium after Thor, the Norse god of thunder. The metal had no practical uses until 1885 when Carl Auer von Welsbach invented the gas mantle. Thorium was first observed to be radioactive in 1898, independently, by Polish-French physicist Marie Curie and German chemist Gerhard Carl Schmidt. Between 1900 and 1903, Ernest Rutherford and Frederick Soddy showed how thorium decayed at a fixed rate over time into a series of other elements. This observation led to the identification of half-life as one of the outcomes of the alpha particle experiments that led to their disintegration theory of radioactivity. The crystal bar process (or "iodide process") was discovered by Anton Eduard van Arkel and Jan Hendrik de Boer in 1925 to produce high-purity metallic thorium. Because of health concerns, the thorium in classic lantern mantles has been replaced by rare-earth elements that also produce intense light without the radioactivity.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
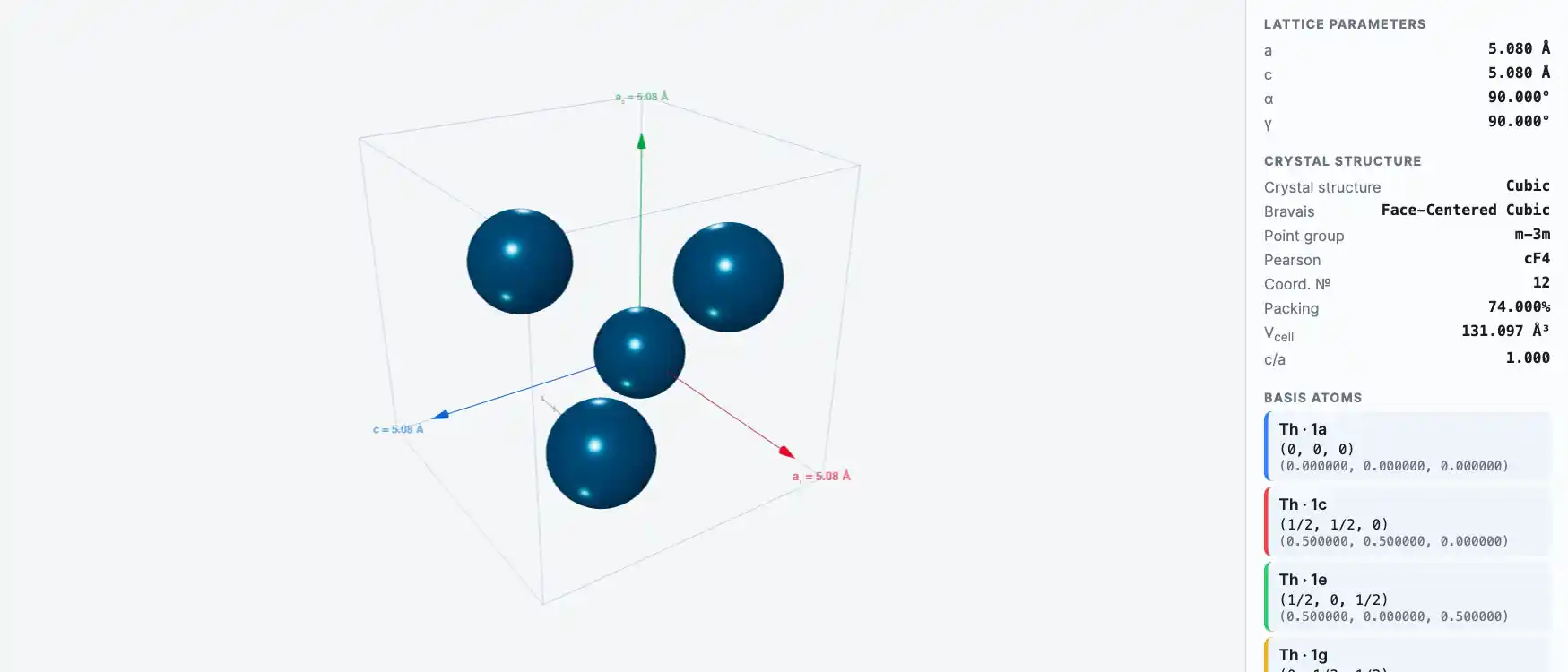
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Th: 6d² 7s²[Rn] 6d² 7s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p⁶ 6d² 7s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 223 Radioactive | 223.0208119 ± 0.0000099 | N/A | 600 ms |
| 217 Radioactive | 217.013117 ± 0.000022 | N/A | 248 us |
| 213 Radioactive | 213.013009 ± 0.000076 | N/A | 144 ms |
| 218 Radioactive | 218.013276 ± 0.000011 | N/A | 122 ns |
| 214 Radioactive | 214.0115 ± 0.000017 | N/A | 87 ms |
Phase / State
Reason: 1724.8 °C below melting point (1749.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 90 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Th I | 0 | 13387 | 0 | 13387 |
| Th II | +1 | 6502 | 0 | 6502 |
| Th III | +2 | 227 | 0 | 227 |
| Th IV | +3 | 27 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Th I | 0 | 788 |
| Th II | +1 | 517 |
| Th III | +2 | 176 |
| Th IV | +3 | 2 |
| Th V | +4 | 2 |
| Th VI | +5 | 2 |
| Th VII | +6 | 2 |
| Th VIII | +7 | 2 |
| Th IX | +8 | 2 |
| Th X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 9 | N/A | 120.9 pm |
| +4 | 6 | N/A | 94 pm |
| +4 | 8 | N/A | 105 pm |
| +4 | 9 | N/A | 109.00000000000001 pm |
| +4 | 10 | N/A | 112.99999999999999 pm |
| +4 | 11 | N/A | 118 pm |
| +4 | 12 | N/A | 121 pm |
Compounds
Isotopes (5)
Twenty-seven thorium radioisotopes have been characterized, with a range in atomic weight from 210 to 236. All are unstable with the most stable being 232Th with a half-life of 14.05 billion years. Thorium-232 represents all but a trace of naturally occurring thorium. It is an alpha emitter and goes through six alpha and four beta decay steps before becoming the stable isotope 208Pb. 232Th is sufficiently radioactive to expose a photographic plate in a few hours. Other isotopes of thorium are short-lived intermediates in the decay chains of higher elements, and only found in trace amounts. The longer-lived of these trace isotopes include: 230Th with a half-life of 75,380 years which is a daughter product of 238U decay; 229Th with a half-life of 7340 years and 228Th with a half-life of 1.92 years. All of the remaining radioactive isotopes have half-lives that are less than thirty days and the majority of these have half-lives less than ten minutes. Much of the internal heat the earth produces has been attributed to thorium and uranium.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 223 Radioactive | 223.0208119 ± 0.0000099 | N/A | 600 ms | α =100% | |
| 217 Radioactive | 217.013117 ± 0.000022 | N/A | 248 us | α =100% | |
| 213 Radioactive | 213.013009 ± 0.000076 | N/A | 144 ms | α ≈100%β+ ? | |
| 218 Radioactive | 218.013276 ± 0.000011 | N/A | 122 ns | α =100% | |
| 214 Radioactive | 214.0115 ± 0.000017 | N/A | 87 ms | α ≈100%β+ ? |
Spectral Lines
Showing 50 of 9955 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 576.055056 nm | 53000 | Th I | emission | 6d2.7s2 3F → 5f.6d.7s2 3D* | Measured | NIST | |
| 645.728238 nm | 44000 | Th I | emission | 5f.6d.7s2 3H* → 5f.6d.7s.7p 5I | Measured | NIST | |
| 395.039509 nm | 42000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 618.262159 nm | 38000 | Th I | emission | 6d2.7s2 3F → 6d.7s2.7p 3P* | Measured | NIST | |
| 658.390575 nm | 32000 | Th I | emission | 6d2.7s2 3F → 6d.7s2.7p 3F* | Measured | NIST | |
| 572.018265 nm | 31000 | Th I | emission | 6d2.7s2 → 5f.6d.7s2 3D* | Measured | NIST | |
| 698.965521 nm | 31000 | Th I | emission | 5f.6d.7s2 3H* → 5f.6d.7s.7p 5I | Measured | NIST | |
| 716.889496 nm | 30000 | Th I | emission | 6d2.7s2 3F → 6d.7s2.7p 3F* | Measured | NIST | |
| 449.3333668 nm | 28000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 658.853947 nm | 26000 | Th I | emission | 6d2.7s2 3P → 6d.7s2.7p 3P* | Measured | NIST | |
| 691.12262 nm | 25000 | Th I | emission | 6d2.7s2 3F → 6d2.7s.7p 5G* | Measured | NIST | |
| 380.307494 nm | 24000 | Th I | emission | 6d2.7s2 3F → 6d2.7s.7p 3D* | Measured | NIST | |
| 403.6047645 nm | 24000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 659.148431 nm | 21000 | Th I | emission | 6d2.7s2 3F → 6d2.7s.7p 5G* | Measured | NIST | |
| 401.912876 nm | 20000 | Th II | emission | 6d.7s2 2D → 6d.7s.(3D).7p * | Measured | NIST | |
| 558.702644 nm | 20000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 411.2754309 nm | 18000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 470.398977 nm | 18000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 580.414105 nm | 18000 | Th I | emission | 6d2.7s2 3F → 6d2.7s.7p 5F* | Measured | NIST | |
| 616.982198 nm | 18000 | Th I | emission | 6d2.7s2 3F → 5f.6d.7s2 3D* | Measured | NIST | |
| 597.366467 nm | 17000 | Th I | emission | 6d2.7s2 → 6d.7s2.7p 3P* | Measured | NIST | |
| 653.134169 nm | 17000 | Th I | emission | 6d3.(4F).7s 5F → 6d2.7s.7p 5P* | Measured | NIST | |
| 383.969505 nm | 16000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 421.092303 nm | 16000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 515.86042 nm | 16000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 620.349239 nm | 16000 | Th I | emission | 6d2.7s2 3F → 6d2.7s.7p 5F* | Measured | NIST | |
| 666.22685 nm | 16000 | Th I | emission | 6d3.(4F).7s 5F → 6d2.7s.7p 5P* | Measured | NIST | |
| 394.8030341 nm | 15000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 403.0842224 nm | 15000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 410.0341193 nm | 15000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 467.366094 nm | 15000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 506.797381 nm | 15000 | Th I | emission | 5f.6d.7s2 3H* | Measured | NIST | |
| 523.115956 nm | 15000 | Th I | emission | 6d2.7s2 3P → 6d2.7s.7p 5P* | Measured | NIST | |
| 634.285945 nm | 15000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 382.838452 nm | 14000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 472.3438197 nm | 14000 | Th I | emission | 6d2.7s2 3F → 5f.6d.7s2 3D* | Measured | NIST | |
| 553.926178 nm | 14000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 694.361046 nm | 14000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 720.80062 nm | 14000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 425.0314489 nm | 13000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 489.495493 nm | 13000 | Th I | emission | 6d2.7s2 3F → 6d.7s2.7p 3P* | Measured | NIST | |
| 387.382217 nm | 12000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 423.5463454 nm | 12000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 480.81337 nm | 12000 | Th I | emission | 6d2.7s2 3F → * | Measured | NIST | |
| 541.748576 nm | 12000 | Th I | emission | 6d2.7s2 → * | Measured | NIST | |
| 599.412865 nm | 12000 | Th I | emission | 6d2.7s2 3P → 6d.7s2.7p 3P* | Measured | NIST | |
| 659.393903 nm | 12000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 708.416896 nm | 12000 | Th I | emission | 6d3.(4F).7s 5F → * | Measured | NIST | |
| 738.550045 nm | 12000 | Th I | emission | 6d2.7s2 → 6d2.7s.7p 5F* | Measured | NIST | |
| 392.440084 nm | 11000 | Th I | emission | 6d2.7s2 → * | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2023.15 K |
| Boiling point | 5058.15 K |
Oxidation State Categories
Advanced Reference Data
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | VI | 108 | calculated, | |
| 4 | VIII | 119 | from r^3 vs V plots, calculated, | |
| 4 | IX | 123 | ||
| 4 | X | 127 | estimated, | |
| 4 | XI | 132 | calculated, | |
| 4 | XII | 135 | calculated, | |
| 3 | IX | — | 134.9 |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 208 | A | 100% |
| 209 | A | — |
| 209 | B+ | — |
| 210 | A | 100% |
| 210 | B+ | — |
| 211 | A | 100% |
| 211 | B+ | — |
| 212 | A | 100% |
| 212 | B+ | — |
| 213 | A | 100% |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.14769 |
| 10.1617 | — | 3.1007 |
| 10.3261 | — | 3.05442 |
| 10.4931 | — | 3.00883 |
| 10.6628 | — | 2.96392 |
| 10.8353 | — | 2.91968 |
| 11.0106 | — | 2.87611 |
| 11.1886 | — | 2.83318 |
| 11.3696 | — | 2.79089 |
| 11.5535 | — | 2.74024 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
9.6 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1×10-6 milligrams per liter
References (1)
Sources
Sources of this element.
Thorium-232 is a primordial nuclide, having existed in its current form for over 4.5 billion years, a half-life is comparable to the age of the Universe and thus predating the formation of the Earth. Thorium was forged in the cores of dying stars through the r-process and scattered across the galaxy by supernovas. Thorium is found in small amounts in most rocks and soils. Soil commonly contains an average of around 6 parts per million (ppm) of thorium. Thorium occurs in several minerals including thorite (ThSiO4), thorianite (ThO2 + UO2) and monazite. Thorianite is a rare mineral and may contain up to about 12% thorium oxide. Monazite contains 2.5% thorium, allanite has 0.1 to 2% thorium and zircon can have up to 0.4% thorium.[66] Thorium-containing minerals occur on all continents. Thorium is now thought to be about three times as abundant as uranium and about as abundant as lead or molybdenum. Thorium is recovered commercially from the mineral monazite, which contains from 3 to 9% ThO2 along with rare-earth minerals.
References (1)
- [6] Thorium https://periodic.lanl.gov/90.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Production
Several methods are available for producing thorium metal; it can be obtained by reducing thorium oxide with calcium, by electrolysis of anhydrous thorium chloride in a fused mixture of sodium and potassium chlorides, by calcium reduction of thorium tetrachloride mixed with anhydrous zinc chloride, and by reduction of thorium tetrachloride with an alkali metal.
References (1)
- [6] Thorium https://periodic.lanl.gov/90.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Thorium.
The element property data was retrieved from publications.