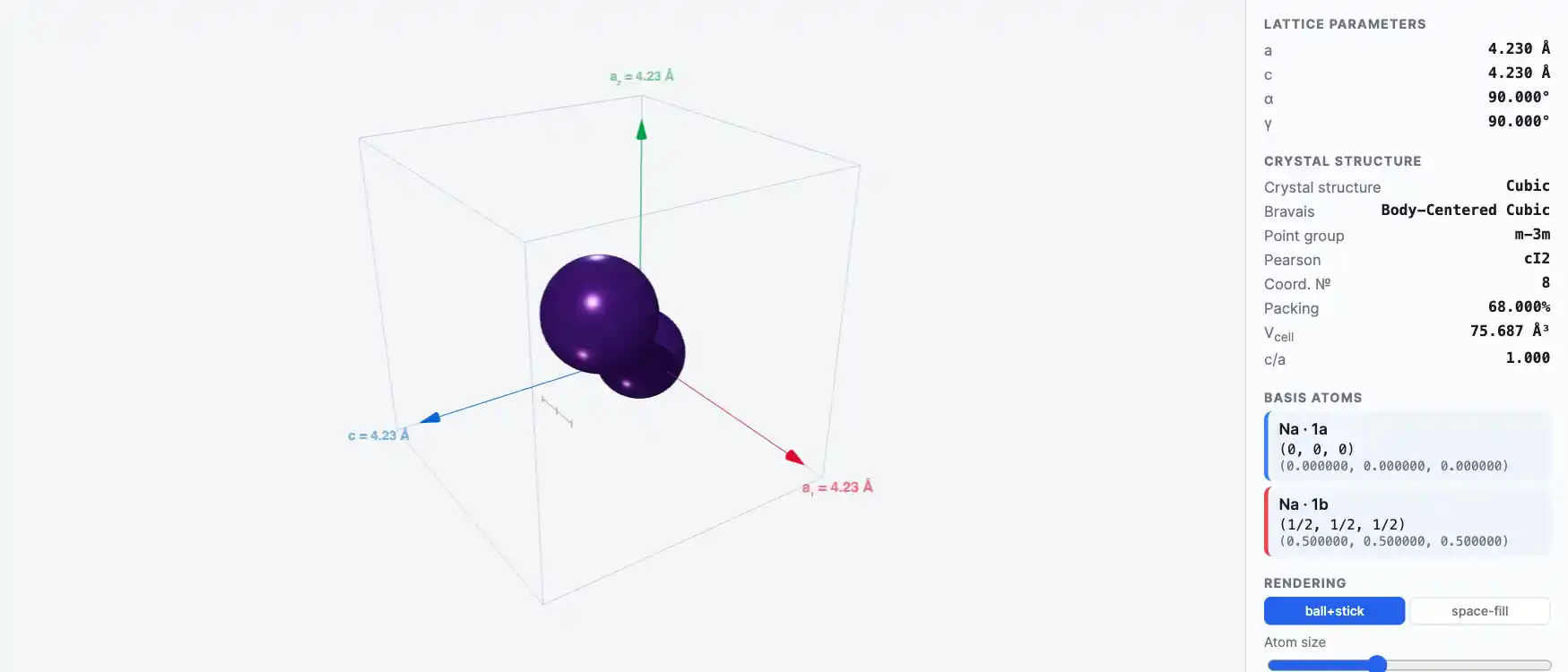
Sodium (Na)
alkali-metalSolid
Standard Atomic Weight
22.989769 uElectron configuration
[Ne] 3s1Melting point
97.8 °C (370.95 K)Boiling point
882.85 °C (1156 K)Density
970 kg/m³Oxidation states
−1, 0, +1Electronegativity (Pauling)
0.93Ionization energy (1st)
Discovery year
1807Atomic radius
180 pmDetails
Sodium is a soft, highly reactive alkali metal in group 1. It has one valence electron and almost always forms Na⁺ in ordinary compounds. The element is abundant in seawater, evaporite deposits, and silicate minerals, but it is never found free in nature because it reacts readily with water, oxygen, and many nonmetals. Its salts are central to physiology, glassmaking, detergents, and bulk chemical manufacture.
Sodium, like every reactive element, is never found free in nature. Sodium is a soft, bright, silvery metal which floats on water. Decomposition in water results in the evolution of hydrogen and the formation of the hydroxide. It may or may not ignite spontaneously on water, depending on the amount of oxide and metal exposed to the water. It normally does not ignite in air at temperatures below 115°C.
The name derives from the English soda and Latin sodanum for "headache remedy". The symbol Na derives from the Latin natrium for "natron" (soda in English). Sodium was discovered in 1807 by the English chemist Humphry Davy from electrolysis of caustic soda (NaOH).
Although sodium is the sixth most abundant element on earth and comprises about 2.6% of the earth's crust, it is a very reactive element and is never found free in nature. Pure sodium was first isolated by Sir Humphry Davy in 1807 through the electrolysis of caustic soda (NaOH). Since sodium can ignite on contact with water, it must be stored in a moisture free environment.
From the English word, soda; Medieval Latin, sodanum: a headache remedy. Long recognized in compounds, sodium was first isolated by Davy in 1807 by electrolysis of caustic soda.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Na: 3s¹[Ne] 3s¹1s² 2s² 2p⁶ 3s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 23 Stable | 22.989769282 ± 0.0000000019 | 100.0000% | Stable |
Phase / State
Reason: 72.8 °C below melting point (97.8 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 11 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Na I | 0 | 869 | 523 | 858 |
| Na II | +1 | 1345 | 176 | 605 |
| Na III | +2 | 560 | 417 | 560 |
| Na IV | +3 | 687 | 671 | 687 |
| Na V | +4 | 529 | 503 | 527 |
| Na VI | +5 | 657 | 594 | 641 |
| Na VII | +6 | 1374 | 1369 | 1374 |
| Na VIII | +7 | 464 | 456 | 464 |
| Na IX | +8 | 172 | 138 | 172 |
| Na X | +9 | 594 | 586 | 594 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Na I | 0 | 430 |
| Na II | +1 | 165 |
| Na III | +2 | 120 |
| Na IV | +3 | 104 |
| Na V | +4 | 102 |
| Na VI | +5 | 116 |
| Na VII | +6 | 142 |
| Na VIII | +7 | 89 |
| Na IX | +8 | 46 |
| Na X | +9 | 111 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 4 | N/A | 99 pm |
| +1 | 5 | N/A | 100 pm |
| +1 | 6 | N/A | 102 pm |
| +1 | 7 | N/A | 112.00000000000001 pm |
| +1 | 8 | N/A | 118 pm |
| +1 | 9 | N/A | 124 pm |
| +1 | 12 | N/A | 139 pm |
Compounds
Isotopes (1)
Thirteen isotopes of sodium are recognized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 23 Stable | 22.989769282 ± 0.0000000019 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 480 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 588.995094 nm | 80000 | Na I | emission | 2p6.3s 2S → 2p6.3p 2P* | Measured | NIST | |
| 589.592424 nm | 40000 | Na I | emission | 2p6.3s 2S → 2p6.3p 2P* | Measured | NIST | |
| 388.181 nm | 420 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4D → 2p5.3s.(3P*).3d 4F* | Measured | NIST | |
| 443.234 nm | 310 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4S → 2p5.3s.(3P*).4s 4P* | Measured | NIST | |
| 411.3703 nm | 300 | Na II | emission | 2s2.2p5.3p 1S → 2s2.2p5.(2P*<1/2>).3d 2[3/2]* | Measured | NIST | |
| 507.12 nm | 270 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4D → 2p5.3s.(3P*).4s 4P* | Measured | NIST | |
| 412.3069 nm | 250 | Na II | emission | 2s2.2p5.3p 1S → 2s2.2p5.(2P*<1/2>).4s 2[1/2]* | Measured | NIST | |
| 423.335 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 424.09 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 429.249 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[1/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 429.287 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[1/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 430.882 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[1/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 430.904 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[1/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 432.091 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 433.729 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 434.412 nm | 250 | Na II | emission | 2s2.2p5.3p 1S → 2s2.2p5.(2P*<3/2>).4s 2[3/2]* | Measured | NIST | |
| 439.281 nm | 250 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[9/2] | Measured | NIST | |
| 408.1372 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[3/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 436.859 nm | 200 | Na II | emission | 2s2.2p5.3p 1S → 2s2.2p5.(2P*<3/2>).3d 2[3/2]* | Measured | NIST | |
| 437.522 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 438.748 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 440.512 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[9/2] | Measured | NIST | |
| 444.669 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 444.741 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 445.473 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 445.523 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 445.72 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 447.463 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 447.88 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[3/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 448.167 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 449.015 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 449.088 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 449.961 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 450.697 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 451.92 nm | 200 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[3/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 452.497 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 453.331 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 455.152 nm | 200 | Na II | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 391.803 nm | 160 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4D → 2p5.3s.(3P*).3d 4F* | Measured | NIST | |
| 438.42 nm | 160 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 439.63 nm | 160 | Na II | emission | 2s2.2p5.(2P*<3/2>).3d 2[7/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 459.094 nm | 160 | Na II | emission | 2s2.2p5.3p 1S → 2s2.2p5.(2P*<3/2>).3d 2[1/2]* | Measured | NIST | |
| 472.23 nm | 160 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 473.113 nm | 160 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 474.163 nm | 160 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[5/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 476.892 nm | 160 | Na II | emission | 2s2.2p5.(2P*<1/2>).3d 2[3/2]* → 2s2.2p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 418.546 nm | 150 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4P → 2p5.3s.(3P*).3d 4D* | Measured | NIST | |
| 386.543 nm | 130 | Na I | emission | 2p5.(2P*).3s.3p.(3P*) 4D → 2p5.3s.(3P*).3d 4F* | Measured | NIST | |
| 408.7593 nm | 130 | Na II | emission | 2s2.2p5.3s 1P* → 2s2.2p5.3p 3S | Measured | NIST | |
| 420.2759 nm | 130 | Na II | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 370.94 K |
| Boiling point | 1156.09 K |
| Critical point (temperature) | 2573.15 K |
| Critical point (pressure) | 35 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (4)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3741 |
| 2 | p | 4.1982 |
| 2 | s | 4.4286 |
| 3 | s | 8.4926 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | IV | 113 | ||
| 1 | V | 114 | ||
| 1 | VI | 116 | ||
| 1 | VII | 126 | ||
| 1 | VIII | 132 | ||
| 1 | IX | 138 | calculated, | |
| 1 | XII | 153 |
Isotope Decay Modes (47)
| Isotope | Mode | Intensity |
|---|---|---|
| 17 | p | 100% |
| 18 | p | — |
| 19 | p | 100% |
| 20 | B+ | 100% |
| 20 | B+A | 25% |
| 21 | B+ | 100% |
| 22 | B+ | 100% |
| 22 | e+ | 90.6% |
| 22 | EC | 9.4% |
| 24 | B- | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.01475 |
| 10.1617 | — | 0.01517 |
| 10.3261 | — | 0.01559 |
| 10.4931 | — | 0.01603 |
| 10.6628 | — | 0.01648 |
| 10.8353 | — | 0.01695 |
| 11.0106 | — | 0.01743 |
| 11.1886 | — | 0.01792 |
| 11.3696 | — | 0.01842 |
| 11.5535 | — | 0.01894 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.36×104 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.08×104 milligrams per liter
References (1)
Sources
Sources of this element.
Sodium is present in fair abundance in the sun and stars. The D lines of sodium are among the most prominent in the solar spectrum. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; it is the most abundant of the alkali group of metals.
It is now obtained commercially by the electrolysis of absolutely dry fused sodium chloride. This method is much cheaper than that of electrolyzing sodium hydroxide, as was used several years ago.
References (1)
- [6] Sodium https://periodic.lanl.gov/11.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Sodium.
The element property data was retrieved from publications.