Magnesium (Mg)
alkaline-earth-metalSolid
Standard Atomic Weight
24.305 u [24.304, 24.307]Electron configuration
[Ne] 3s2Melting point
649.85 °C (923 K)Boiling point
1089.85 °C (1363 K)Density
1740 kg/m³Oxidation states
0, +1, +2Electronegativity (Pauling)
1.31Ionization energy (1st)
Discovery year
1755Atomic radius
150 pmDetails
Magnesium is a light alkaline earth metal and a major rock-forming element. It occurs in silicate minerals, carbonates, evaporites, seawater, and brines, almost entirely as Mg²⁺ rather than as native metal. Its low density, ready formation of stable salts, and high affinity for oxygen shape both its metallurgy and its geochemistry. Magnesium is also essential in biology, where it stabilizes phosphate chemistry and is central to chlorophyll.
Magnesium is a light, silvery-white, and fairly tough metal. It tarnishes slightly in air, and finely divided magnesium readily ignites upon heating in air and burns with a dazzling white flame.
The name derives from Magnesia, a district in the north-eastern region of Greece called Thessalia. The Scottish chemist Joseph Black recognized it as a separate element in 1755. In 1808, the English chemist Humphry Davy obtained the impure metal, and in 1831 the French pharmacist and chemist Antoine- Alexandre Brutus Bussy isolated the metal in the pure state.
Although it is the eighth most abundant element in the universe and the seventh most abundant element in the earth's crust, magnesium is never found free in nature. Magnesium was first isolated by Sir Humphry Davy, an English chemist, through the electrolysis of a mixture of magnesium oxide (MgO) and mercuric oxide (HgO) in 1808. Today, magnesium can be extracted from the minerals dolomite (CaCO3·MgCO3) and carnallite (KCl·MgCl2·6H2O), but is most often obtained from seawater. Every cubic kilometer of seawater contains about 1.3 billion kilograms of magnesium (12 billion pounds per cubic mile).
From Magnesia, district in Thessaly. Compounds of magnesium have long been known. Black recognized magnesium as an element in 1755. Davy isolated it in 1808 and Bussy prepared it in coherent form in 1831. Magnesium is the eighth most abundant element in the earth's crust. It does not occur uncombined, but is found in large deposits in the form of magnesite, dolomite, and other minerals.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
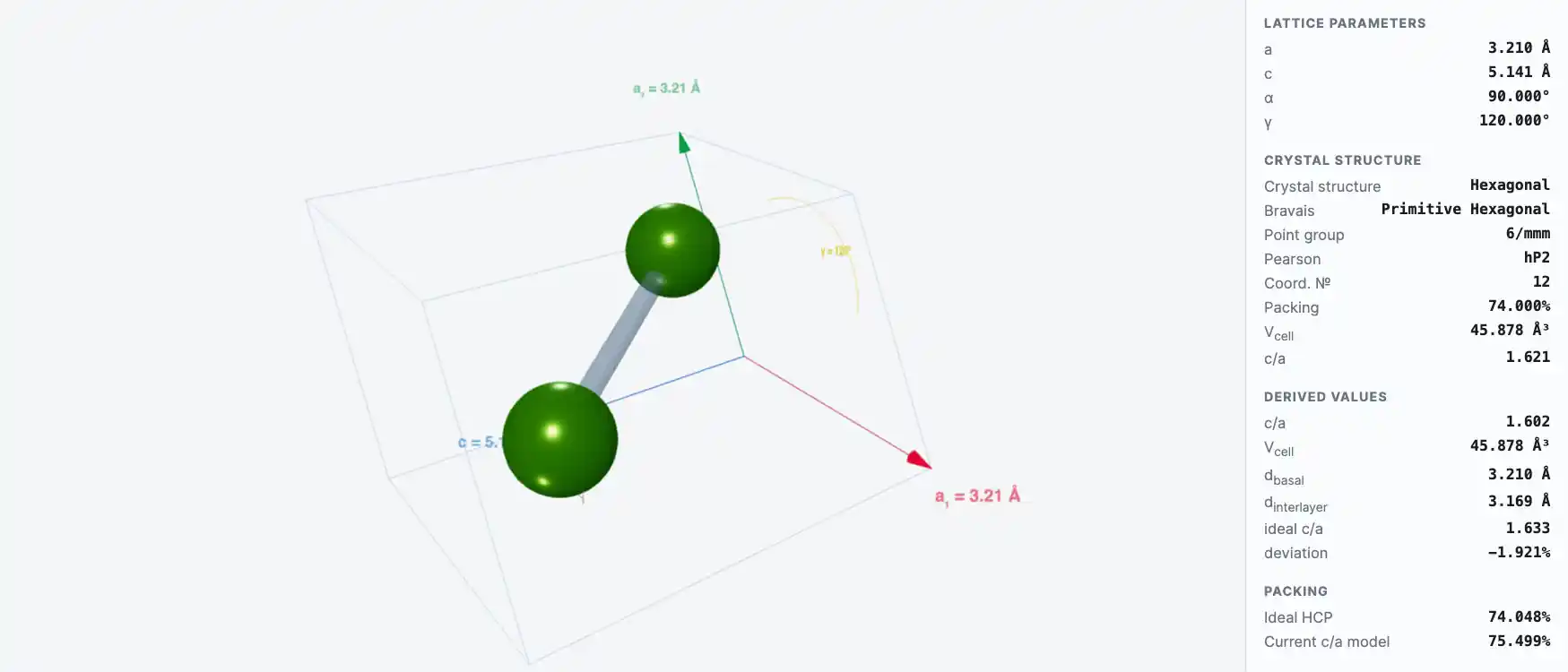
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Mg: 3s²[Ne] 3s²1s² 2s² 2p⁶ 3s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 24 Stable | 23.985041697 ± 0.000000014 | 78.9900% | Stable |
| 25 Stable | 24.985836976 ± 0.00000005 | 10.0000% | Stable |
| 26 Stable | 25.982592968 ± 0.000000031 | 11.0100% | Stable |
Phase / State
Reason: 624.9 °C below melting point (649.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 12 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Mg I | 0 | 1342 | 1090 | 1342 |
| Mg II | +1 | 601 | 482 | 601 |
| Mg III | +2 | 452 | 149 | 452 |
| Mg IV | +3 | 821 | 625 | 821 |
| Mg V | +4 | 518 | 513 | 515 |
| Mg VI | +5 | 890 | 883 | 890 |
| Mg VII | +6 | 379 | 344 | 379 |
| Mg VIII | +7 | 944 | 941 | 944 |
| Mg IX | +8 | 461 | 444 | 461 |
| Mg X | +9 | 223 | 184 | 223 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Mg I | 0 | 323 |
| Mg II | +1 | 149 |
| Mg III | +2 | 114 |
| Mg IV | +3 | 173 |
| Mg V | +4 | 104 |
| Mg VI | +5 | 120 |
| Mg VII | +6 | 104 |
| Mg VIII | +7 | 113 |
| Mg IX | +8 | 94 |
| Mg X | +9 | 60 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 56.99999999999999 pm |
| +2 | 5 | N/A | 66 pm |
| +2 | 6 | N/A | 72 pm |
| +2 | 8 | N/A | 89 pm |
Compounds
Isotopes (3)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 24 Stable | 23.985041697 ± 0.000000014 | 78.9900% ± 0.0400% | Stable | stable | |
| 25 Stable | 24.985836976 ± 0.00000005 | 10.0000% ± 0.0100% | Stable | stable | |
| 26 Stable | 25.982592968 ± 0.000000031 | 11.0100% ± 0.0300% | Stable | stable |
Spectral Lines
Showing 50 of 399 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 518.36043 nm | 45 | Mg I | emission | 3s.3p 3P* → 3s.4s 3S | Measured | NIST | |
| 517.26844 nm | 44 | Mg I | emission | 3s.3p 3P* → 3s.4s 3S | Measured | NIST | |
| 516.73213 nm | 42 | Mg I | emission | 3s.3p 3P* → 3s.4s 3S | Measured | NIST | |
| 383.82919 nm | 40 | Mg I | emission | 3s.3p 3P* → 3s.3d 3D | Measured | NIST | |
| 552.84047 nm | 40 | Mg I | emission | 3s.3p 1P* → 3s.4d 1D | Measured | NIST | |
| 383.23039 nm | 38 | Mg I | emission | 3s.3p 3P* → 3s.3d 3D | Measured | NIST | |
| 382.93547 nm | 36 | Mg I | emission | 3s.3p 3P* → 3s.3d 3D | Measured | NIST | |
| 470.29908 nm | 30 | Mg I | emission | 3s.3p 1P* → 3s.5d 1D | Measured | NIST | |
| 571.1088 nm | 30 | Mg I | emission | 3s.3p 1P* → 3s.5s 1S | Measured | NIST | |
| 435.19057 nm | 20 | Mg I | emission | 3s.3p 1P* → 3s.6d 1D | Measured | NIST | |
| 416.72713 nm | 15 | Mg I | emission | 3s.3p 1P* → 3s.7d 1D | Measured | NIST | |
| 625.6757 nm | 15 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[5/2] | Measured | NIST | |
| 448.1126 nm | 14 | Mg II | emission | 2p6.3d 2D → 2p6.4f 2F* | Measured | NIST | |
| 448.1325 nm | 13 | Mg II | emission | 2p6.3d 2D → 2p6.4f 2F* | Measured | NIST | |
| 738.7689 nm | 12 | Mg I | emission | 3s.3d 1D → 3s.8f 1F* | Measured | NIST | |
| 405.75052 nm | 10 | Mg I | emission | 3s.3p 1P* → 3s.8d 1D | Measured | NIST | |
| 439.0572 nm | 10 | Mg II | emission | 2p6.4p 2P* → 2p6.5d 2D | Measured | NIST | |
| 473.00286 nm | 10 | Mg I | emission | 3s.3p 1P* → 3s.6s 1S | Measured | NIST | |
| 491.5991 nm | 10 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[5/2] → 2s2.2p5.(2P*<3/2>).4d 2[7/2]* | Measured | NIST | |
| 583.981 nm | 10 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[3/2] | Measured | NIST | |
| 631.8717 nm | 10 | Mg I | emission | 3s.4s 3S → 3s.6p 3P* | Measured | NIST | |
| 634.6742 nm | 10 | Mg II | emission | 2p6.4d 2D → 2p6.6f 2F* | Measured | NIST | |
| 719.3184 nm | 10 | Mg I | emission | 3s.3d 1D → 3s.9f 1F* | Measured | NIST | |
| 729.1055 nm | 10 | Mg I | emission | 3s.4s 1S → 3s.6p 1P* | Measured | NIST | |
| 438.4637 nm | 9 | Mg II | emission | 2p6.4p 2P* → 2p6.5d 2D | Measured | NIST | |
| 443.3988 nm | 9 | Mg II | emission | 2p6.4p 2P* → 2p6.6s 2S | Measured | NIST | |
| 452.6219 nm | 9 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[3/2]* | Measured | NIST | |
| 459.6921 nm | 9 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[1/2]* | Measured | NIST | |
| 496.041 nm | 9 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[5/2] → 2s2.2p5.(2P*<3/2>).4d 2[7/2]* | Measured | NIST | |
| 631.9237 nm | 9 | Mg I | emission | 3s.4s 3S → 3s.6p 3P* | Measured | NIST | |
| 634.6964 nm | 9 | Mg II | emission | 2p6.4d 2D → 2p6.6f 2F* | Measured | NIST | |
| 384.8211 nm | 8 | Mg II | emission | 2p6.3d 2D → 2p6.5p 2P* | Measured | NIST | |
| 398.67533 nm | 8 | Mg I | emission | 3s.3p 1P* → 3s.9d 1D | Measured | NIST | |
| 442.7994 nm | 8 | Mg II | emission | 2p6.4p 2P* → 2p6.6s 2S | Measured | NIST | |
| 467.3315 nm | 8 | Mg III | emission | 2s2.2p5.(2P*<1/2>).4s 2[1/2]* → 2s2.2p5.(2P*<1/2>).4p 2[1/2] | Measured | NIST | |
| 498.1469 nm | 8 | Mg III | emission | 2s2.2p5.(2P*<1/2>).4p 2[3/2] → 2s2.2p5.(2P*<1/2>).4d 2[5/2]* | Measured | NIST | |
| 526.422 nm | 8 | Mg II | emission | 2p6.4d 2D → 2p6.7f 2F* | Measured | NIST | |
| 640.6637 nm | 8 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[5/2] | Measured | NIST | |
| 678.7855 nm | 8 | Mg II | emission | 2p6.5p 2P* → 2p6.7d 2D | Measured | NIST | |
| 681.927 nm | 8 | Mg II | emission | 2p6.5p 2P* → 2p6.8s 2S | Measured | NIST | |
| 706.0414 nm | 8 | Mg I | emission | 3s.3d 1D → 3s.10f 1F* | Measured | NIST | |
| 385.0386 nm | 7 | Mg II | emission | 2p6.3d 2D → 2p6.5p 2P* | Measured | NIST | |
| 423.9473 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<1/2>).4p 2[1/2] | Measured | NIST | |
| 463.2537 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[1/2]* | Measured | NIST | |
| 480.2585 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[3/2] → 2s2.2p5.(2P*<3/2>).4d 2[3/2]* | Measured | NIST | |
| 491.5363 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<1/2>).4p 2[3/2] → 2s2.2p5.(2P*<1/2>).4d 2[5/2]* | Measured | NIST | |
| 497.0497 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[3/2] → 2s2.2p5.(2P*<3/2>).4d 2[5/2]* | Measured | NIST | |
| 502.3674 nm | 7 | Mg III | emission | 2s2.2p5.(2P*<3/2>).4p 2[3/2] → 2s2.2p5.(2P*<3/2>).4d 2[5/2]* | Measured | NIST | |
| 526.4364 nm | 7 | Mg II | emission | 2p6.4d 2D → 2p6.7f 2F* | Measured | NIST | |
| 591.6431 nm | 7 | Mg II | emission | 2p6.4d 2D → 2p6.7p 2P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 923.15 K |
| Boiling point | 1363.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (4)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3911 |
| 2 | p | 4.1742 |
| 2 | s | 4.608 |
| 3 | s | 8.6925 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | 71 | ||
| 2 | V | 80 | ||
| 2 | VI | 86 | ||
| 2 | VIII | 103 | calculated, |
Isotope Decay Modes (43)
| Isotope | Mode | Intensity |
|---|---|---|
| 19 | 2p | 100% |
| 20 | B+ | 100% |
| 20 | B+p | 30.3% |
| 21 | B+ | 100% |
| 21 | B+p | 20.1% |
| 21 | B+A | 0.1% |
| 21 | B+pA | 0% |
| 22 | B+ | 100% |
| 23 | B+ | 100% |
| 27 | B- | 100% |
X‑ray Scattering Factors (755)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 0.5 | 0.117 | 0.14592 |
| 0.5079 | 0.1168 | 0.14896 |
| 0.516 | 0.1165 | 0.15206 |
| 0.5242 | 0.1165 | 0.15522 |
| 0.5325 | 0.1162 | 0.15845 |
| 0.5409 | 0.1162 | 0.16175 |
| 0.5495 | 0.1162 | 0.16511 |
| 0.5582 | 0.1161 | 0.16855 |
| 0.5671 | 0.1163 | 0.17205 |
| 0.5761 | 0.1167 | 0.17558 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.33×104 milligrams per kilogram
References (1)
- [5] Magnesium https://education.jlab.org/itselemental/ele012.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.29×103 milligrams per liter
References (1)
- [5] Magnesium https://education.jlab.org/itselemental/ele012.html
Sources
Sources of this element.
The metal is now principally obtained in the U.S. by electrolysis of fused magnesium chloride derived from brines, wells, and sea water.
References (1)
- [6] Magnesium https://periodic.lanl.gov/12.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Magnesium.
The element property data was retrieved from publications.