Aluminum (Al)
post-transition-metalSolid
Standard Atomic Weight
26.981538 uElectron configuration
[Ne] 3s2 3p1Melting point
660.287 °C (933.437 K)Boiling point
2518.85 °C (2792 K)Density
2700 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3Electronegativity (Pauling)
1.61Ionization energy (1st)
Discovery year
1825Atomic radius
125 pmDetails
Aluminum is a light, silvery post-transition metal and the most abundant metal in Earth’s crust. It is highly reactive thermodynamically, but a thin, adherent oxide film protects the metal from rapid corrosion in air and water. Its low density, electrical conductivity, formability, and alloying behavior make it one of the major structural and engineering metals.
Pure aluminum, a silvery-white metal, possesses many desirable characteristics. It is light, it is nonmagnetic and nonsparking, stands second among metals in the scale of malleability, and sixth in ductility.
The name derives from the Latin, alum and alumen for "stringent" because the early Romans called any substance with a stringent taste alum. The element was known in prehistoric times. In 1825, the Danish physicist, Hans Christian Oersted, isolated impure aluminium. The pure metal was first isolated by the German chemist Friedrich Wöhler in 1827.
Although aluminum is the most abundant metal in the earth's crust, it is never found free in nature. All of the earth's aluminum has combined with other elements to form compounds. Two of the most common compounds are alum, such as potassium aluminum sulfate (KAl(SO4)2·12H2O), and aluminum oxide (Al2O3). About 8.2% of the earth's crust is composed of aluminum. Scientists suspected than an unknown metal existed in alum as early as 1787, but they did not have a way to extract it until 1825. Hans Christian Oersted, a Danish chemist, was the first to produce tiny amounts of aluminum. Two years later, Friedrich Wöhler, a German chemist, developed a different way to obtain aluminum. By 1845, he was able to produce samples large enough to determine some of aluminum's basic properties. Wöhler's method was improved in 1854 by Henri Étienne Sainte-Claire Deville, a French chemist. Deville's process allowed for the commercial production of aluminum. As a result, the price of aluminum dropped from around $1200 per kilogram in 1852 to around $40 per kilogram in 1859. Unfortunately, aluminum remained too expensive to be widely used.
From the Latin word alumen, alum. The ancient Greeks and Romans used alum as an astringent and as a mordant in dyeing. In 1761 de Morveau proposed the name alumine for the base in alum, and Lavoisier, in 1787, thought this to be the oxide of a still undiscovered metal.
Friedrich Wohler is generally credited with having isolated the metal in 1827, although an impure form was prepared by Oersted two years earlier. In 1807, Davy proposed the name aluminium for the metal, undiscovered at that time, and later agreed to change it to aluminum. Shortly thereafter, the name aluminum was adopted to conform with the "ium" ending of most elements.
Aluminium was also the accepted spelling in the U.S. until 1925, at which time the American Chemical Society decided to use the name aluminum thereafter in their publications. See the Wikipedia entry on Aluminium for additional discussion on the spelling of this element.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
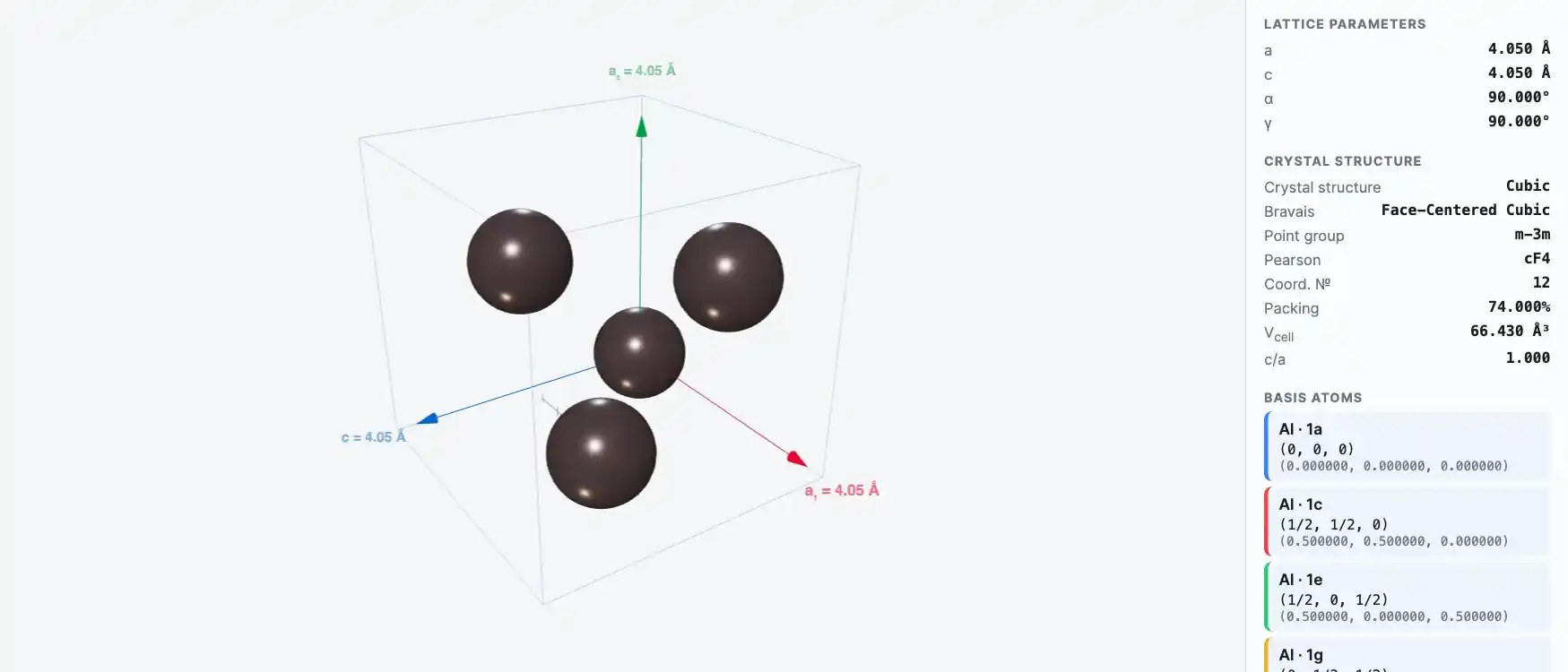
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Al: 3s² 3p¹[Ne] 3s² 3p¹1s² 2s² 2p⁶ 3s² 3p¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 27 Stable | 26.98153853 ± 0.00000011 | 100.0000% | Stable |
Phase / State
Reason: 635.3 °C below melting point (660.287 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 13 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Al I | 0 | 589 | 322 | 560 |
| Al II | +1 | 1429 | 986 | 1429 |
| Al III | +2 | 344 | 259 | 344 |
| Al IV | +3 | 409 | 123 | 409 |
| Al V | +4 | 600 | 444 | 600 |
| Al VI | +5 | 515 | 491 | 515 |
| Al VII | +6 | 350 | 339 | 350 |
| Al VIII | +7 | 440 | 418 | 438 |
| Al IX | +8 | 372 | 339 | 372 |
| Al X | +9 | 189 | 169 | 189 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Al I | 0 | 192 |
| Al II | +1 | 219 |
| Al III | +2 | 84 |
| Al IV | +3 | 121 |
| Al V | +4 | 158 |
| Al VI | +5 | 87 |
| Al VII | +6 | 73 |
| Al VIII | +7 | 95 |
| Al IX | +8 | 69 |
| Al X | +9 | 61 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 4 | N/A | 39 pm |
| +3 | 5 | N/A | 48 pm |
| +3 | 6 | N/A | 53.5 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 27 Stable | 26.98153853 ± 0.00000011 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 341 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 466.3046 nm | 1000 | Al II | emission | 3p2 1D → 3s.4p 1P* | Measured | NIST | |
| 559.33 nm | 800 | Al II | emission | 3s.4p 1P* → 3s.4d 1D | Measured | NIST | |
| 458.5818 nm | 500 | Al II | emission | 3s.4d 3D → 3s.7f 3F* | Measured | NIST | |
| 458.8199 nm | 400 | Al II | emission | 3s.4d 3D → 3s.7f 3F* | Measured | NIST | |
| 464.8609 nm | 400 | Al II | emission | 3s.4d 1D → 3s.10p 1P* | Measured | NIST | |
| 466.6799 nm | 400 | Al II | emission | 3s.5p 1P* → 3s.11s 1S | Measured | NIST | |
| 458.975 nm | 300 | Al II | emission | 3s.4d 3D → 3s.7f 3F* | Measured | NIST | |
| 444.7805 nm | 200 | Al II | emission | 3s.4d 1D → 3s.11p 1P* | Measured | NIST | |
| 458.968 nm | 200 | Al II | emission | 3s.4d 3D → 3s.7f 3F* | Measured | NIST | |
| 600.641 nm | 200 | Al II | emission | 3s.5p 3P* → 3s.7d 3D | Measured | NIST | |
| 390.0675 nm | 100 | Al II | emission | 3s.3p 1P* → 3p2 1D | Measured | NIST | |
| 528.3733 nm | 100 | Al II | emission | 3s.5p 3P* → 3s.8d 3D | Measured | NIST | |
| 561.329 nm | 100 | Al II | emission | 3s.4d 1D → 3s.7f 1F* | Measured | NIST | |
| 585.376 nm | 100 | Al II | emission | 3s.4d 3D → 3s.6f 3F* | Measured | NIST | |
| 624.337 nm | 100 | Al II | emission | 3s.4p 3P* → 3s.4d 3D | Measured | NIST | |
| 704.208 nm | 100 | Al II | emission | 3s.4s 3S → 3s.4p 3P* | Measured | NIST | |
| 747.141 nm | 90 | Al II | emission | 3s.3d 1D → 3s.4f 1F* | Measured | NIST | |
| 586.177 nm | 80 | Al II | emission | 3s.4d 3D → 3s.6f 3F* | Measured | NIST | |
| 597.197 nm | 80 | Al II | emission | 3s.5p 1P* → 3s.7d 1D | Measured | NIST | |
| 683.713 nm | 80 | Al II | emission | 3s.4p 3P* → 3s.5s 3S | Measured | NIST | |
| 623.175 nm | 75 | Al II | emission | 3s.4p 3P* → 3s.4d 3D | Measured | NIST | |
| 600.187 nm | 60 | Al II | emission | 3s.5p 3P* → 3s.7d 3D | Measured | NIST | |
| 422.6816 nm | 50 | Al II | emission | 3s.4d 3D → 3s.8f 3F* | Measured | NIST | |
| 422.7495 nm | 50 | Al II | emission | 3s.4d 3D → 3s.8f 3F* | Measured | NIST | |
| 422.7987 nm | 50 | Al II | emission | 3s.4d 3D → 3s.8f 3F* | Measured | NIST | |
| 586.79 nm | 50 | Al II | emission | 3s.4d 3D → 3s.6f 3F* | Measured | NIST | |
| 607.32 nm | 50 | Al II | emission | 3s.5p 3P* → 3s.8s 3S | Measured | NIST | |
| 622.619 nm | 50 | Al II | emission | 3s.4p 3P* → 3s.4d 3D | Measured | NIST | |
| 682.339 nm | 50 | Al II | emission | 3s.4p 3P* → 3s.5s 3S | Measured | NIST | |
| 705.671 nm | 50 | Al II | emission | 3s.4s 3S → 3s.4p 3P* | Measured | NIST | |
| 744.944 nm | 50 | Al II | emission | 3s.5p 1P* → 3s.6d 1D | Measured | NIST | |
| 399.5837 nm | 40 | Al II | emission | 3s.4d 3D → 3s.9f 3F* | Measured | NIST | |
| 450.371 nm | 40 | Al IV | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[5/2] | Measured | NIST | |
| 600.192 nm | 40 | Al II | emission | 3s.5p 3P* → 3s.7d 3D | Measured | NIST | |
| 399.6141 nm | 30 | Al II | emission | 3s.4d 3D → 3s.9f 3F* | Measured | NIST | |
| 450.237 nm | 30 | Al IV | emission | 2s2.2p5.(2P*<1/2>).4s 2[1/2]* → 2s2.2p5.(2P*<1/2>).4p 2[3/2] | Measured | NIST | |
| 463.576 nm | 30 | Al II | emission | 3s.5p 3P* → 3s.10d 3D | Measured | NIST | |
| 528.5838 nm | 30 | Al II | emission | 3s.5p 1P* → 3s.8d 1D | Measured | NIST | |
| 606.112 nm | 30 | Al II | emission | 3s.5p 1P* → 3s.8s 1S | Measured | NIST | |
| 633.571 nm | 30 | Al II | emission | 3s.3d 1D → 3s.5p 1P* | Measured | NIST | |
| 399.6368 nm | 20 | Al II | emission | 3s.4d 3D → 3s.9f 3F* | Measured | NIST | |
| 402.6318 nm | 20 | Al II | emission | 3s.3d 1D → 3s.6p 1P* | Measured | NIST | |
| 446.894 nm | 20 | Al IV | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[3/2] | Measured | NIST | |
| 569.66 nm | 17 | Al III | emission | 2p6.4s 2S → 2p6.4p 2P* | Measured | NIST | |
| 572.273 nm | 16 | Al III | emission | 2p6.4s 2S → 2p6.4p 2P* | Measured | NIST | |
| 462.038 nm | 15 | Al IV | emission | 2s2.2p5.(2P*<3/2>).4s 2[3/2]* → 2s2.2p5.(2P*<3/2>).4p 2[5/2] | Measured | NIST | |
| 531.6073 nm | 15 | Al II | emission | 3s.5p 3P* → 3s.9s 3S | Measured | NIST | |
| 452.919 nm | 14 | Al III | emission | 2p6.4p 2P* → 2p6.4d 2D | Measured | NIST | |
| 451.257 nm | 13 | Al III | emission | 2p6.4p 2P* → 2p6.4d 2D | Measured | NIST | |
| 669.6018 nm | 13 | Al I | emission | 3s2.4s 2S → 3s2.5p 2P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 933.47 K |
| Boiling point | 2792.15 K |
| Critical point (temperature) | 6700.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (5)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.409 |
| 2 | p | 4.0366 |
| 2 | s | 4.7864 |
| 3 | p | 8.9344 |
| 3 | s | 8.8828 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | IV | 53 | ||
| 3 | V | 62 | ||
| 3 | VI | 67.5 | from r^3 vs V plots, |
Isotope Decay Modes (51)
| Isotope | Mode | Intensity |
|---|---|---|
| 21 | p | — |
| 22 | B+ | 100% |
| 22 | B+p | 55% |
| 22 | 2p | 1.1% |
| 22 | B+A | 0% |
| 23 | B+ | 100% |
| 23 | B+p | 1.2% |
| 24 | B+ | 100% |
| 24 | B+A | 0% |
| 24 | B+p | 0% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.1199 |
| 10.1617 | — | 3.05822 |
| 10.3261 | — | 2.99776 |
| 10.4931 | — | 2.9385 |
| 10.6628 | — | 2.88041 |
| 10.8353 | — | 2.82347 |
| 11.0106 | — | 2.76766 |
| 11.1886 | — | 2.722 |
| 11.3696 | — | 2.69148 |
| 11.5535 | — | 2.66129 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8.23×104 milligrams per kilogram
References (1)
- [5] Aluminum https://education.jlab.org/itselemental/ele013.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-3 milligrams per liter
References (1)
- [5] Aluminum https://education.jlab.org/itselemental/ele013.html
Sources
Sources of this element.
The method of obtaining aluminum metal by the electrolysis of alumina dissolved in cryolite was discovered in 1886 by Hall in the U.S. and at about the same time by Heroult in France. Cryolite, a natural ore found in Greenland, is no longer widely used in commercial production, but has been replaced by an artificial mixture of sodium, aluminum, and calcium fluorides.
Aluminum can now be produced from clay, but the process is not economically feasible at present. Aluminum is the most abundant metal to be found in the earth's crust (8.1%), but is never found free in nature. In addition to the minerals mentioned above, it is also found in granite and in many other common minerals.
References (1)
- [6] Aluminum https://periodic.lanl.gov/13.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Aluminum.
The element property data was retrieved from publications.