Chlorine (Cl)
halogenGas
Standard Atomic Weight
35.45 u [35.446, 35.457]Electron configuration
[Ne] 3s2 3p5Melting point
-101.5 °C (171.65 K)Boiling point
-34.04 °C (239.11 K)Density
3.214 kg/m³Oxidation states
−1, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
3.16Ionization energy (1st)
Discovery year
1774Atomic radius
100 pmDetails
Chlorine is a reactive halogen and a yellow-green diatomic gas, Cl₂, under ordinary conditions. It is a strong oxidizing agent and occurs naturally mainly as chloride salts, especially in seawater and evaporite minerals. Chlorine chemistry is central to water disinfection, inorganic chlorides, chlorinated solvents, polymers, and many industrial oxidation and substitution processes. The element has two stable isotopes, ³⁵Cl and ³⁷Cl.
It is a member of the halogen (salt-forming) group of elements and is obtained from chlorides by the action of oxidizing agents and more often by electrolysis; it is a greenish-yellow gas, combining directly with nearly all elements. At 10°C one volume of water dissolves 3.10 volumes of chlorine, at 30°C only 1.77 volumes.
The name derives from the Greek chloros for "pale green" or "greenish yellow" colour of the element. It was discovered by the Swedish pharmacist and chemist Carl-Wilhelm Scheele in 1774. In 1810, the English chemist Humphry Davy proved it was an element.
Since it combines directly with nearly every element, chlorine is never found free in nature. Chlorine was first produced by Carl Wilhelm Scheele, a Swedish chemist, when he combined the mineral pyrolusite (MnO2) with hydrochloric acid (HCl) in 1774. Although Scheele thought the gas produced in his experiment contained oxygen, Sir Humphry Davy proved in 1810 that it was actually a distinct element. Today, most chlorine is produced through the electrolysis of aqueous sodium chloride (NaCl).
From the Greek word chloro, greenish yellow. Discovered in 1774 by Scheele, who thought it contained oxygen. Chlorine was named in 1810 by Davy, who insisted it was an element.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
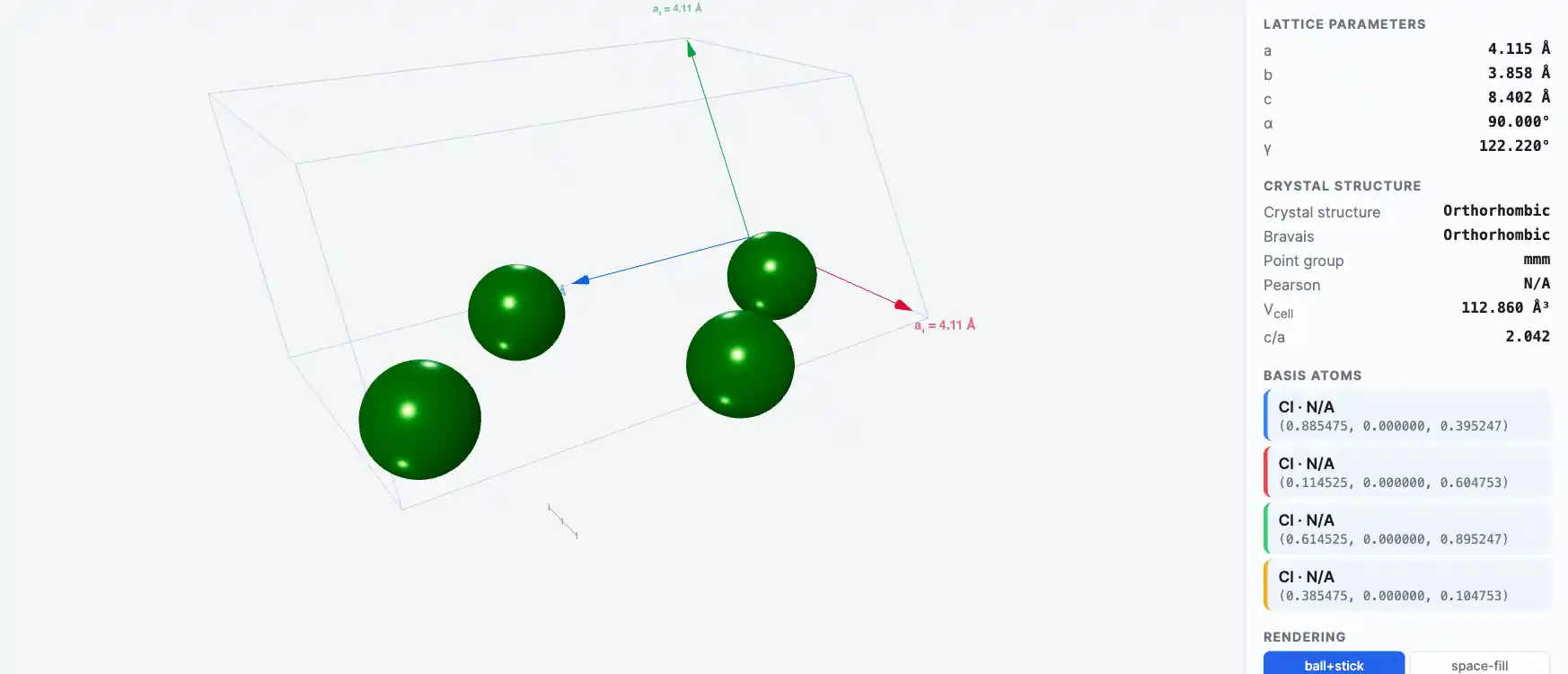
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Cl: 3s² 3p⁵[Ne] 3s² 3p⁵1s² 2s² 2p⁶ 3s² 3p⁵Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 35 Stable | 34.968852682 ± 0.000000037 | 75.7600% | Stable |
| 37 Stable | 36.965902602 ± 0.000000055 | 24.2400% | Stable |
Phase / State
Reason: 59.0 °C above boiling point (-34.04 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Showing 10 of 17 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Cl I | 0 | 221 | 99 | 100 |
| Cl II | +1 | 292 | 221 | 221 |
| Cl III | +2 | 192 | 166 | 166 |
| Cl IV | +3 | 73 | 42 | 45 |
| Cl V | +4 | 27 | 6 | 6 |
| Cl VI | +5 | 1 | 1 | 1 |
| Cl VII | +6 | 28 | 28 | 28 |
| Cl VIII | +7 | 5 | 5 | 5 |
| Cl IX | +8 | 3 | 3 | 3 |
| Cl X | +9 | 11 | 11 | 11 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Cl I | 0 | 379 |
| Cl II | +1 | 275 |
| Cl III | +2 | 83 |
| Cl IV | +3 | 41 |
| Cl V | +4 | 29 |
| Cl VI | +5 | 67 |
| Cl VII | +6 | 47 |
| Cl VIII | +7 | 29 |
| Cl IX | +8 | 57 |
| Cl X | +9 | 15 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -1 | 6 | N/A | 181 pm |
| +5 | 3 | N/A | 12 pm |
| +7 | 4 | N/A | 8 pm |
| +7 | 6 | N/A | 27 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 35 Stable | 34.968852682 ± 0.000000037 | 75.7600% ± 0.1000% | Stable | stable | |
| 37 Stable | 36.965902602 ± 0.000000055 | 24.2400% ± 0.1000% | Stable | stable |
Spectral Lines
Showing 50 of 207 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 479.4556 nm | 99000 | Cl II | emission | 3s2.3p3.(4S*).4s 5S* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 542.3257 nm | 99000 | Cl II | emission | 3s2.3p3.(4S*).3d 5D* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 489.6783 nm | 81000 | Cl II | emission | 3s2.3p3.(2D*).4s 3D* → 3s2.3p3.(2D*).4p 3F | Measured | NIST | |
| 521.7945 nm | 56000 | Cl II | emission | 3s2.3p3.(4S*).4s 3S* → 3s2.3p3.(4S*).4p 3P | Measured | NIST | |
| 490.4776 nm | 47000 | Cl II | emission | 3s2.3p3.(2D*).4s 3D* → 3s2.3p3.(2D*).4p 3F | Measured | NIST | |
| 481.007 nm | 29000 | Cl II | emission | 3s2.3p3.(4S*).4s 5S* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 491.773 nm | 26000 | Cl II | emission | 3s2.3p3.(2D*).4s 3D* → 3s2.3p3.(2D*).4p 3F | Measured | NIST | |
| 507.8267 nm | 26000 | Cl II | emission | 3s2.3p3.(2D*).4s 3D* → 3s2.3p3.(2D*).4p 3D | Measured | NIST | |
| 386.0828 nm | 25000 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 522.1362 nm | 23000 | Cl II | emission | 3s2.3p3.(4S*).4s 3S* → 3s2.3p3.(4S*).4p 3P | Measured | NIST | |
| 544.3375 nm | 19000 | Cl II | emission | 3s2.3p3.(4S*).3d 5D* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 481.948 nm | 16000 | Cl II | emission | 3s2.3p3.(4S*).4s 5S* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 539.2125 nm | 15000 | Cl II | emission | 3s2.3p3.(2D*).4s 1D* → 3s2.3p3.(2D*).4p 1F | Measured | NIST | |
| 478.132 nm | 13000 | Cl II | emission | 3s2.3p3.(2P*).4s 3P* → 3s2.3p3.(2P*).4p 3D | Measured | NIST | |
| 385.0988 nm | 10000 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 499.5473 nm | 10000 | Cl II | emission | 3s2.3p3.(2D*).3d 3F* → 3s2.3p3.(2D*).4p 3D | Measured | NIST | |
| 542.3516 nm | 10000 | Cl II | emission | 3s2.3p3.(4S*).3d 5D* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 544.4217 nm | 10000 | Cl II | emission | 3s2.3p3.(4S*).3d 5D* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 385.1374 nm | 7900 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 725.6618 nm | 7500 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4S* | Measured | NIST | |
| 545.7037 nm | 5600 | Cl II | emission | 3s2.3p3.(4S*).3d 5D* → 3s2.3p3.(4S*).4p 5P | Measured | NIST | |
| 741.4118 nm | 5000 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 2P* | Measured | NIST | |
| 386.099 nm | 4400 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 476.8651 nm | 4300 | Cl II | emission | 3s2.3p3.(2P*).4s 3P* → 3s2.3p3.(2P*).4p 3D | Measured | NIST | |
| 384.5639 nm | 3900 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 384.5362 nm | 3100 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 380.5174 nm | 1900 | Cl II | emission | 3s2.3p3.(2D*).4p 3D → 3s2.3p3.(2D*).4d 3F* | Measured | NIST | |
| 609.468 nm | 1900 | Cl II | emission | 3s2.3p3.(2D*).4s 1D* → 3s2.3p3.(2D*).4p 1P | Measured | NIST | |
| 384.5788 nm | 1500 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 391.3866 nm | 1500 | Cl II | emission | 3s2.3p3.(2D*).4p 3F → 3s2.3p3.(2D*).4d 3F* | Measured | NIST | |
| 380.9459 nm | 1300 | Cl II | emission | 3s2.3p3.(2D*).4p 3D → 3s2.3p3.(2D*).4d 3F* | Measured | NIST | |
| 385.1651 nm | 1200 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 391.6632 nm | 1100 | Cl II | emission | 3s2.3p3.(2D*).4p 3F → 3s2.3p3.(2D*).4d 3F* | Measured | NIST | |
| 386.1378 nm | 1000 | Cl II | emission | 3s2.3p3.(4S*).4p 5P → 3s2.3p3.(4S*).4d 5D* | Measured | NIST | |
| 399.1367 nm | 700 | Cl III | emission | 3s2.3p2.(3P).3d 4P → 3s2.3p2.(3P).4p 4P* | Measured | NIST | |
| 401.8351 nm | 600 | Cl III | emission | 3s2.3p2.(3P).3d 4P → 3s2.3p2.(3P).4p 4P* | Measured | NIST | |
| 405.893 nm | 600 | Cl III | emission | 3s2.3p2.(3P).3d 4P → 3s2.3p2.(3P).4p 4P* | Measured | NIST | |
| 410.4082 nm | 500 | Cl III | emission | 3s2.3p2.(3P).3d 4P → 3s2.3p2.(3P).4p 4P* | Measured | NIST | |
| 410.6764 nm | 500 | Cl III | emission | 3s2.3p2.(3P).3d 4P → 3s2.3p2.(3P).4p 4P* | Measured | NIST | |
| 436.3268 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4D* | Measured | NIST | |
| 436.9498 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 2D* | Measured | NIST | |
| 437.9896 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4D* | Measured | NIST | |
| 438.9751 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4D* | Measured | NIST | |
| 443.8488 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4P* | Measured | NIST | |
| 452.6182 nm | 100 | Cl I | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).5p 2P* | Measured | NIST | |
| 439.0403 nm | 90 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4D* | Measured | NIST | |
| 440.302 nm | 90 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 4P* | Measured | NIST | |
| 447.5304 nm | 90 | Cl I | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).5p 2D* | Measured | NIST | |
| 460.0977 nm | 80 | Cl I | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).5p 2P* | Measured | NIST | |
| 466.1208 nm | 80 | Cl I | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).5p 2P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 171.65 K |
| Boiling point | 239.11 K |
| Critical point (temperature) | 417.05 K |
| Critical point (pressure) | 7.99 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (5)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.4761 |
| 2 | p | 4.0068 |
| 2 | s | 5.5696 |
| 3 | p | 10.8839 |
| 3 | s | 9.9317 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -1 | VI | 167 | Pauling's (1960) crystal radius, | |
| 5 | IIIPY | 26 | ||
| 7 | IV | 22 | ||
| 7 | VI | 41 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (45)
| Isotope | Mode | Intensity |
|---|---|---|
| 28 | p | 100% |
| 29 | p | 100% |
| 30 | p | 100% |
| 31 | B+ | 100% |
| 31 | B+p | 2.4% |
| 32 | B+ | 100% |
| 32 | B+A | 0.1% |
| 32 | B+p | 0% |
| 33 | B+ | 100% |
| 34 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.46938 |
| 10.1617 | — | 1.6922 |
| 10.3261 | — | 1.9488 |
| 10.4931 | — | 2.24432 |
| 10.6628 | — | 2.51303 |
| 10.8353 | — | 2.73153 |
| 11.0106 | — | 2.95549 |
| 11.1886 | — | 3.19416 |
| 11.3696 | — | 3.48881 |
| 11.5535 | — | 3.87618 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.45×102 milligrams per kilogram
References (1)
- [5] Chlorine https://education.jlab.org/itselemental/ele017.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.94×104 milligrams per liter
References (1)
- [5] Chlorine https://education.jlab.org/itselemental/ele017.html
Sources
Sources of this element.
In nature it is found in the combined state only, chiefly with sodium as common salt (NaCl), carnallite, and sylvite.
References (1)
- [6] Chlorine https://periodic.lanl.gov/17.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Analyses of chlorine isotopes and other environmental tracers can help to identify whether an environmental contaminant is of anthropogenic origin or naturally occurring. For example, perchlorate (ClO4 -) can be of anthropogenic origin and is also found naturally. Perchlorate is a widespread groundwater contaminant that can interfere with hormone production in the thyroid gland by displacing iodide. Both the stable chlorine isotope-amount ratio n(37Cl)/n(35Cl) and the mole fraction of 36Cl, n(36Cl)/n(Cl), can provide useful information about origins of perchlorate in the environment (Fig. IUPAC.17.2). Such information may be important for legal reasons and for remediation of contaminated areas [152] [152] M. A. Stewart, A. J. Spivack. Rev. Mineral. Geochem.55, 231 (2004).[152] M. A. Stewart, A. J. Spivack. Rev. Mineral. Geochem.55, 231 (2004).[152] M. A. Stewart, A. J. Spivack. Rev. Mineral. Geochem.55, 231 (2004).[152] M. A. Stewart, A. J. Spivack. Rev. Mineral. Geochem.55, 231 (2004)., [153] [153] J. K. Böhlke, N. C. Sturchio, B. Gu, J. Horita, G. M. Brown, W. A. Jackson, J. R. Batista, P. B. Hatzinger. Anal. Chem.77, 7838 (2005).[153] J. K. Böhlke, N. C. Sturchio, B. Gu, J. Horita, G. M. Brown, W. A. Jackson, J. R. Batista, P. B. Hatzinger. Anal. Chem.77, 7838 (2005)..
References (4)
- [152] M. A. Stewart, A. J. Spivack. Rev. Mineral. Geochem.55, 231 (2004).
- [153] J. K. Böhlke, N. C. Sturchio, B. Gu, J. Horita, G. M. Brown, W. A. Jackson, J. R. Batista, P. B. Hatzinger. Anal. Chem.77, 7838 (2005).
- [154] J. K. Böhlke, P. Hatzinger, N. C. Sturchio, B. Gu, I. J. Abbene, S. J. Mroczkowski. Environ. Sci. Technol.43, 5619 (2009).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Chlorine.
The element property data was retrieved from publications.