Argon (Ar)
noble-gasGas
Standard Atomic Weight
39.948 u [39.792, 39.963]Electron configuration
[Ne] 3s2 3p6Melting point
-189.35 °C (83.8 K)Boiling point
-185.85 °C (87.3 K)Density
1.7837 kg/m³Oxidation states
0Electronegativity (Pauling)
N/AIonization energy (1st)
Discovery year
1894Atomic radius
71 pmDetails
Argon is a colorless noble gas and the third most abundant gas in Earth’s atmosphere, after nitrogen and oxygen. Its closed-shell electron configuration makes it chemically very inert under ordinary conditions. Most terrestrial argon is ⁴⁰Ar, produced by the radioactive decay of ⁴⁰K in rocks. The element is valued mainly as a dense, nonreactive atmosphere for industrial, analytical, and lighting applications.
Argon is two and one half times as soluble in water as nitrogen, having about the same solubility as oxygen. Argon is colorless and odorless, both as a gas and liquid. Argon is considered to be a very inert gas and is not known to form true chemical compounds, as do krypton, xenon, and radon.
The name derives from the Greek argos for "lazy" or "inactive" because it does not combine with other elements. It was discovered in 1894 by the Scottish chemist William Ramsay and the English physicist Robert John Strutt (Lord Rayleigh) in liquefied air. Rayleigh's initial interest derived from a problem posed by the English physicist Henry Cavendish in 1785, i.e., when oxygen and nitrogen were removed from air, there was an unknown residual gas remaining.
Argon was discovered by Sir William Ramsay, a Scottish chemist, and Lord Rayleigh, an English chemist, in 1894. Argon makes up 0.93% of the earth's atmosphere, making it the third most abundant gas. Argon is obtained from the air as a byproduct of the production of oxygen and nitrogen.
From the Greek argos, inactive. Its presence in air was suspected by Cavendish in 1785, discovered by Lord Raleigh and Sir William Ramsay in 1894.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
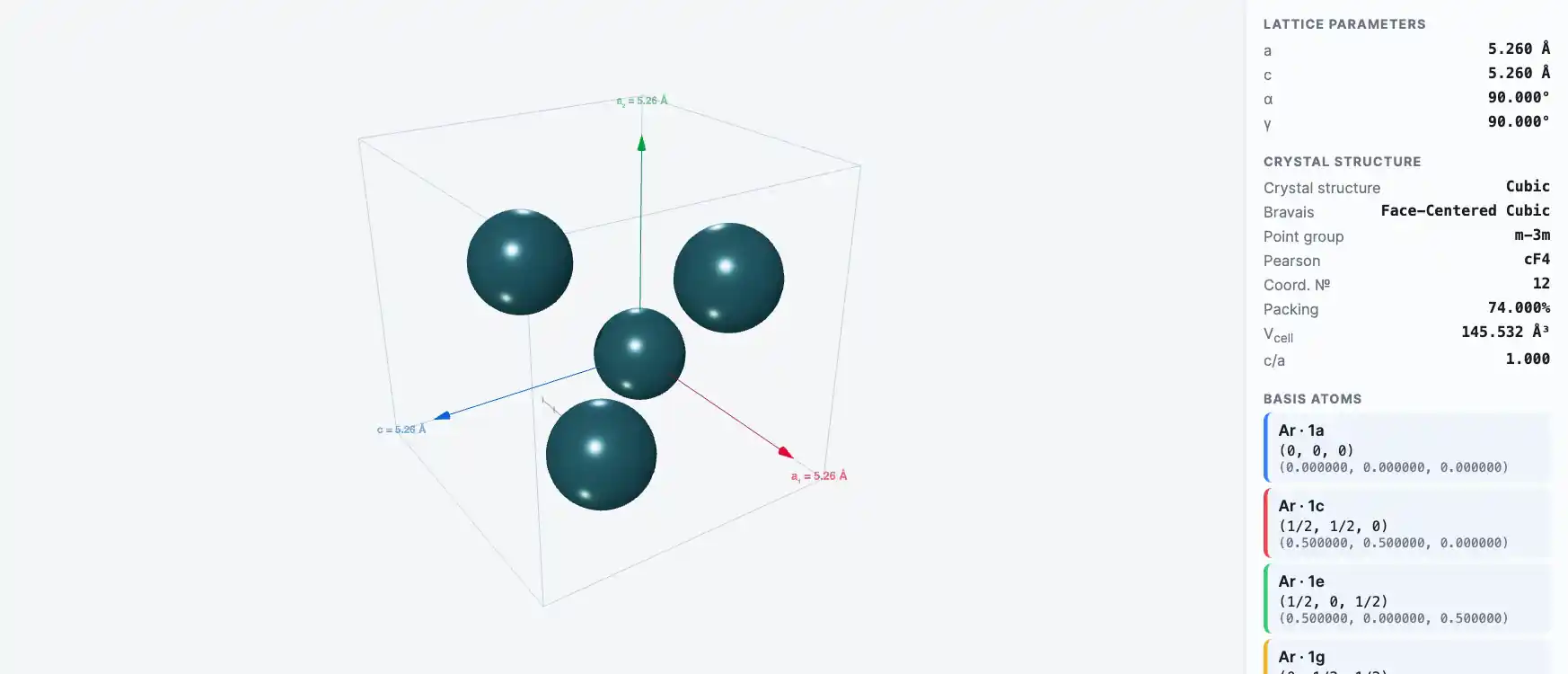
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ar: 3s² 3p⁶[Ne] 3s² 3p⁶1s² 2s² 2p⁶ 3s² 3p⁶Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 36 Stable | 35.967545105 ± 0.000000028 | 0.3336% | Stable |
| 38 Stable | 37.96273211 ± 0.00000021 | 0.0629% | Stable |
| 40 Stable | 39.9623831237 ± 0.0000000024 | 99.6035% | Stable |
Phase / State
Reason: 210.8 °C above boiling point (-185.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Showing 10 of 18 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ar I | 0 | 461 | 428 | 429 |
| Ar II | +1 | 2858 | 307 | 2858 |
| Ar III | +2 | 509 | 80 | 509 |
| Ar IV | +3 | 256 | 42 | 256 |
| Ar V | +4 | 111 | 18 | 111 |
| Ar VI | +5 | 104 | 6 | 104 |
| Ar VII | +6 | 218 | 21 | 218 |
| Ar VIII | +7 | 141 | 27 | 141 |
| Ar IX | +8 | 178 | 2 | 178 |
| Ar X | +9 | 92 | 1 | 92 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ar I | 0 | 504 |
| Ar II | +1 | 419 |
| Ar III | +2 | 125 |
| Ar IV | +3 | 58 |
| Ar V | +4 | 49 |
| Ar VI | +5 | 44 |
| Ar VII | +6 | 95 |
| Ar VIII | +7 | 72 |
| Ar IX | +8 | 98 |
| Ar X | +9 | 71 |
Compounds
Isotopes (3)
Naturally occurring argon is a mixture of three isotopes. Twelve other radioactive isotopes are known to exist.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 36 Stable | 35.967545105 ± 0.000000028 | 0.3336% ± 0.0021% | Stable | stable | |
| 38 Stable | 37.96273211 ± 0.00000021 | 0.0629% ± 0.0007% | Stable | stable | |
| 40 Stable | 39.9623831237 ± 0.0000000024 | 99.6035% ± 0.0025% | Stable | stable |
Spectral Lines
Showing 50 of 1065 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 458.989759 nm | 25704 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2F* | Measured | NIST | |
| 472.686807 nm | 23442 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2D* | Measured | NIST | |
| 696.543 nm | 10000 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<1/2>).4p 2[1/2] | Measured | NIST | |
| 706.72175 nm | 10000 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<1/2>).4p 2[3/2] | Measured | NIST | |
| 738.39801 nm | 10000 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<1/2>).4p 2[3/2] | Measured | NIST | |
| 440.098598 nm | 8710 | Ar II | emission | 3s2.3p4.(3P).3d 4D → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 501.716264 nm | 7413 | Ar II | emission | 3s2.3p4.(3P).3d 2D → 3s2.3p4.(1D).4p 2F* | Measured | NIST | |
| 476.486444 nm | 2344 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2P* | Measured | NIST | |
| 460.956692 nm | 2291 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2F* | Measured | NIST | |
| 487.986345 nm | 2239 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2D* | Measured | NIST | |
| 727.29354 nm | 2000 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<1/2>).4p 2[1/2] | Measured | NIST | |
| 427.752786 nm | 1995 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2P* | Measured | NIST | |
| 434.806354 nm | 1995 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 480.602014 nm | 1820 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 454.505166 nm | 1738 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2P* | Measured | NIST | |
| 442.60008 nm | 1514 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 465.79009 nm | 1445 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2P* | Measured | NIST | |
| 473.590548 nm | 1000 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 714.7041 nm | 1000 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<1/2>).4p 2[3/2] | Measured | NIST | |
| 413.172327 nm | 891 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2P* | Measured | NIST | |
| 496.507942 nm | 891 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2D* | Measured | NIST | |
| 457.934934 nm | 871 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2S* | Measured | NIST | |
| 484.780955 nm | 832 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 437.988058 nm | 794 | Ar II | emission | 3s2.3p4.(3P).4p 2S* → 3s2.3p4.(3P).5s 2P | Measured | NIST | |
| 407.200431 nm | 708 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2D* | Measured | NIST | |
| 443.018862 nm | 661 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 448.181045 nm | 646 | Ar II | emission | 3s2.3p4.(3P).3d 2D → 3s2.3p4.(1D).4p 2D* | Measured | NIST | |
| 437.075295 nm | 617 | Ar II | emission | 3s2.3p4.(3P).3d 2D → 3s2.3p4.(1D).4p 2D* | Measured | NIST | |
| 433.119915 nm | 603 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 437.966649 nm | 550 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 611.492319 nm | 537 | Ar II | emission | 3s2.3p4.(1D).3d 2G → 3s2.3p4.(1D).4p 2F* | Measured | NIST | |
| 410.391181 nm | 447 | Ar II | emission | 3s2.3p4.(3P).4p 4D* → 3s2.3p4.(3P).5s 4P | Measured | NIST | |
| 617.227751 nm | 407 | Ar II | emission | 3s2.3p4.(1D).3d 2G → 3s2.3p4.(1D).4p 2F* | Measured | NIST | |
| 415.85907 nm | 400 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 420.067472 nm | 400 | Ar I | emission | 3s2.3p5.(2P*<3/2>).4s 2[3/2]* → 3s2.3p5.(2P*<3/2>).5p 2[5/2] | Measured | NIST | |
| 506.203703 nm | 398 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 437.132854 nm | 355 | Ar II | emission | 3s2.3p4.(3P).3d 4D → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 500.93342 nm | 355 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 447.475916 nm | 347 | Ar II | emission | 3s2.3p4.(3P).3d 2D → 3s2.3p4.(1D).4p 2P* | Measured | NIST | |
| 422.815775 nm | 331 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 2D* | Measured | NIST | |
| 404.289342 nm | 288 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2D* | Measured | NIST | |
| 426.652661 nm | 288 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 488.904194 nm | 288 | Ar II | emission | 3s2.3p4.(3P).4s 2P → 3s2.3p4.(3P).4p 2P* | Measured | NIST | |
| 423.721956 nm | 269 | Ar II | emission | 3s2.3p4.(1D).4s 2D → 3s2.3p4.(1D).4p 2P* | Measured | NIST | |
| 664.369734 nm | 269 | Ar II | emission | 3s2.3p4.(3P).3d 4F → 3s2.3p4.(3P).4p 4D* | Measured | NIST | |
| 385.058079 nm | 263 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4S* | Measured | NIST | |
| 440.009637 nm | 257 | Ar II | emission | 3s2.3p4.(3P).3d 4D → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 443.099589 nm | 251 | Ar II | emission | 3s2.3p4.(3P).3d 4D → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 493.320891 nm | 251 | Ar II | emission | 3s2.3p4.(3P).4s 4P → 3s2.3p4.(3P).4p 4P* | Measured | NIST | |
| 514.17826 nm | 224 | Ar II | emission | 3s2.3p4.(3P).3d 2D → 3s2.3p4.(1D).4p 2F* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Noble Gas Properties
Phase Transitions & Allotropes
| Melting point | 83.81 K |
| Boiling point | 87.3 K |
| Critical point (temperature) | 150.69 K |
| Critical point (pressure) | 4.86 MPa |
| Triple point (temperature) | 83.81 K |
| Triple point (pressure) | 68.89 kPa |
Advanced Reference Data
Screening Constants (5)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.4925 |
| 2 | p | 3.9918 |
| 2 | s | 5.7696 |
| 3 | p | 11.2359 |
| 3 | s | 10.2432 |
Isotope Decay Modes (46)
| Isotope | Mode | Intensity |
|---|---|---|
| 29 | 2p | 100% |
| 30 | 2p | 100% |
| 31 | B+ | 100% |
| 31 | B+p | 68.3% |
| 31 | 2p | 9% |
| 31 | B+pA | 0.4% |
| 31 | 3p | 0.1% |
| 31 | B+A | 0% |
| 31 | 2p | 0% |
| 32 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0 |
| 10.1617 | — | 0 |
| 10.3261 | — | 0 |
| 10.4931 | — | 0 |
| 10.6628 | — | 0 |
| 10.8353 | — | 0 |
| 11.0106 | — | 0 |
| 11.1886 | — | 0 |
| 11.3696 | — | 0 |
| 11.5535 | — | 0 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.5 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4.5×10-1 milligrams per liter
References (1)
Sources
Sources of this element.
The gas is prepared by fractionation of liquid air because the atmosphere contains 0.94% argon. The atmosphere of Mars contains 1.6% of 40Ar and 5 ppm of 36Ar.
References (1)
- [6] Argon https://periodic.lanl.gov/18.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Argon.
The element property data was retrieved from publications.