Helium (He)
noble-gasGas
Standard Atomic Weight
4.002602 uElectron configuration
1s2Melting point
-272.2 °C (0.95 K)Boiling point
-268.93 °C (4.22 K)Density
0.1785 kg/m³Oxidation states
0Electronegativity (Pauling)
N/AIonization energy (1st)
Discovery year
1868Atomic radius
120 pmDetails
Helium is a noble gas and the second element in the periodic table. It is chemically inert under ordinary conditions because its 1s electron shell is filled, and it exists as monatomic He rather than as a molecule. Its low density, very low boiling point, high thermal conductivity, and nonflammability make it technologically important. On Earth it is uncommon in the atmosphere but can accumulate in some natural gas reservoirs through radioactive decay of uranium and thorium.
Helium has the lowest melting point of any element and is widely used in cryogenic research because its boiling point is close to absolute zero. Also, the element is vital in the study of super conductivity.
Using liquid helium, Kurti, co-workers and others have succeeded in obtaining temperatures of a few microkelvins by the adiabatic demagnetization of copper nuclei.
Helium has other peculiar properties: It is the only liquid that cannot be solidified by lowering the temperature. It remains liquid down to absolute zero at ordinary pressures, but will readily solidify by increasing the pressure. Solid 3He and 4He are unusual in that both can be changed in volume by more than 30% by applying pressure.
The specific heat of helium gas is unusually high. The density of helium vapor at the normal boiling point is also very high, with the vapor expanding greatly when heated to room temperature. Containers filled with helium gas at 5 to 10 K should be treated as though they contained liquid helium due to the large increase in pressure resulting from warming the gas to room temperature.
While helium normally has a 0 valence, it seems to have a weak tendency to combine with certain other elements. Means of preparing helium difluoride have been studied, and species such as HeNe and the molecular ions He+ and He++ have been investigated.
The name derives from the Greek helios for "sun". The element was discovered by spectroscopy during a solar eclipse in the sun's chromosphere by the French astronomer Pierre-Jules-Cesar Janssen in 1868. It was independently discovered and named helium by the English astronomer Joseph Norman Lockyer.
Helium was thought to be only a solar constituent until it was later found to be identical to the helium in the uranium ore cleveite by the Scottish chemist William Ramsay in 1895. The Swedish chemists Per Theodore Cleve and Nils Abraham Langet independently found helium in cleveite at about the same time.
Helium, the second most abundant element in the universe, was discovered on the sun before it was found on the earth. Pierre-Jules-César Janssen, a French astronomer, noticed a yellow line in the sun's spectrum while studying a total solar eclipse in 1868. Sir Norman Lockyer, an English astronomer, realized that this line, with a wavelength of 587.49 nanometers, could not be produced by any element known at the time. It was hypothesized that a new element on the sun was responsible for this mysterious yellow emission. This unknown element was named helium by Lockyer. The hunt to find helium on earth ended in 1895. Sir William Ramsay, a Scottish chemist, conducted an experiment with a mineral containing uranium called clevite. He exposed the clevite to mineral acids and collected the gases that were produced. He then sent a sample of these gases to two scientists, Lockyer and Sir William Crookes, who were able to identify the helium within it. Two Swedish chemists, Nils Langlet and Per Theodor Cleve, independently found helium in clevite at about the same time as Ramsay.
Helium makes up about 0.0005% of the earth's atmosphere. This trace amount of helium is not gravitationally bound to the earth and is constantly lost to space. The earth's atmospheric helium is replaced by the decay of radioactive elements in the earth's crust. Alpha decay, one type of radioactive decay, produces particles called alpha particles. An alpha particle can become a helium atom once it captures two electrons from its surroundings. This newly formed helium can eventually work its way to the atmosphere through cracks in the crust.
From the Greek word helios, the sun. Janssen obtained the first evidence of helium during the solar eclipse of 1868 when he detected a new line in the solar spectrum. Lockyer and Frankland suggested the name helium for the new element. In 1895 Ramsay discovered helium in the uranium mineral cleveite while it was independently discovered in cleveite by the Swedish chemists Cleve and Langlet at about the same time. Rutherford and Royds in 1907 demonstrated that alpha particles are helium nuclei.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
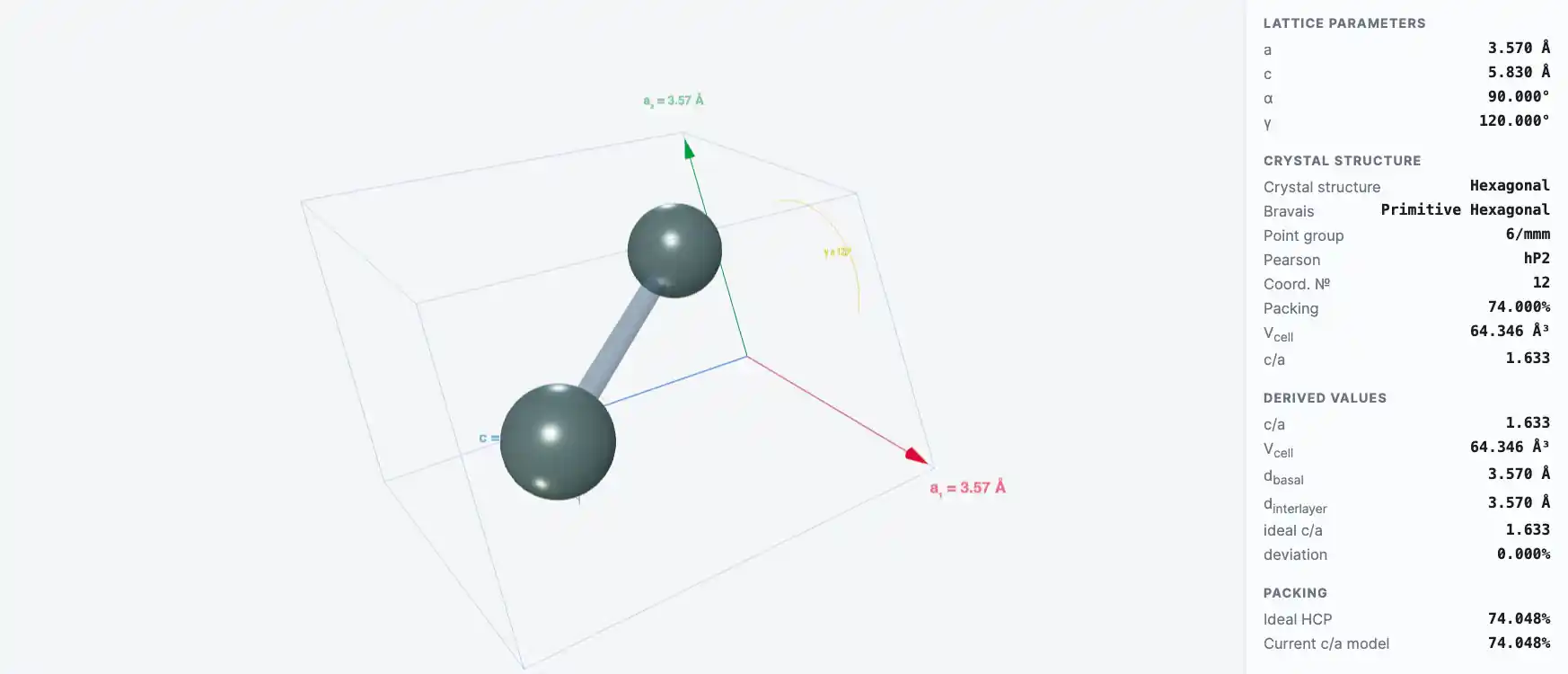
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
He: 1s²1s²1s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 3 Stable | 3.0160293201 ± 0.0000000025 | 0.0001% | Stable |
| 4 Stable | 4.00260325413 ± 0.00000000006 | 99.9999% | Stable |
Phase / State
Reason: 293.9 °C above boiling point (-268.93 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| 3He I Isotope | 0 | 2289 | 0 | 2289 |
| He I | 0 | 2300 | 2289 | 2300 |
| He II | +1 | 140 | 140 | 140 |
| 3He II Isotope | +1 | 140 | 140 | 140 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| 3He I Isotope | 0 | 188 |
| He I | 0 | 843 |
| He II | +1 | 149 |
| 3He II Isotope | +1 | 149 |
Compounds
Isotopes (2)
Seven isotopes of helium are known: Liquid helium (He-4) exists in two forms: He-4I and He-4II, with a sharp transition point at 2.174K. He-4I (above this temperature) is a normal liquid, but He-4II (below it) is unlike any other known substance. It expands on cooling, its conductivity for heat is enormous, and neither its heat conduction nor viscosity obeys normal rules.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 3 Stable | 3.0160293201 ± 0.0000000025 | 0.0001% ± 0.0000% | Stable | stable | |
| 4 Stable | 4.00260325413 ± 0.00000000006 | 99.9999% ± 0.0000% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 381.9601975 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 381.9602773 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 381.9602828 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 381.9613129 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 381.9613927 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 381.975731 nm | 1 | He I | emission | 1s.2p 3P* → 1s.6d 3D | Measured | NIST | |
| 383.3548713 nm | 0 | He I | emission | 1s.2p 1P* → 1s.10d 1D | Measured | NIST | |
| 383.8100125 nm | N/A | He I | emission | 1s.2p 1P* → 1s.10s 1S | Measured | NIST | |
| 386.7472343 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6s 3S | Measured | NIST | |
| 386.7483778 nm | N/A | He I | emission | 1s.2p 3P* → 1s.6s 3S | Measured | NIST | |
| 386.7631595 nm | 1 | He I | emission | 1s.2p 3P* → 1s.6s 3S | Measured | NIST | |
| 387.1786406 nm | 1 | He I | emission | 1s.2p 1P* → 1s.9d 1D | Measured | NIST | |
| 387.8176858 nm | N/A | He I | emission | 1s.2p 1P* → 1s.9s 1S | Measured | NIST | |
| 388.8604644 nm | N/A | He I | emission | 1s.2s 3S → 1s.3p 3P* | Measured | NIST | |
| 388.864559 nm | N/A | He I | emission | 1s.2s 3S → 1s.3p 3P* | Measured | NIST | |
| 388.8648915 nm | N/A | He I | emission | 1s.2s 3S → 1s.3p 3P* | Measured | NIST | |
| 392.6544387 nm | 1 | He I | emission | 1s.2p 1P* → 1s.8d 1D | Measured | NIST | |
| 393.5945223 nm | 0 | He I | emission | 1s.2p 1P* → 1s.8s 1S | Measured | NIST | |
| 396.4728829 nm | 20 | He I | emission | 1s.2s 1S → 1s.4p 1P* | Measured | NIST | |
| 397.2015454 nm | N/A | He I | emission | 1s.2s 1S → 1s.4d 1D | Measured | NIST | |
| 400.9256516 nm | 1 | He I | emission | 1s.2p 1P* → 1s.7d 1D | Measured | NIST | |
| 402.3979795 nm | 1 | He I | emission | 1s.2p 1P* → 1s.7s 1S | Measured | NIST | |
| 402.6184368 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 402.6185901 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 402.6186005 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 402.619676 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 402.6198294 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 402.6356959 nm | 5 | He I | emission | 1s.2p 3P* → 1s.5d 3D | Measured | NIST | |
| 412.0810765 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5s 3S | Measured | NIST | |
| 412.0823747 nm | N/A | He I | emission | 1s.2p 3P* → 1s.5s 3S | Measured | NIST | |
| 412.0991564 nm | 2 | He I | emission | 1s.2p 3P* → 1s.5s 3S | Measured | NIST | |
| 414.1332157 nm | N/A | He I | emission | 1s.2p 1P* → 1s.6p 1P* | Measured | NIST | |
| 414.3759059 nm | 3 | He I | emission | 1s.2p 1P* → 1s.6d 1D | Measured | NIST | |
| 416.8971512 nm | 1 | He I | emission | 1s.2p 1P* → 1s.6s 1S | Measured | NIST | |
| 438.3278555 nm | N/A | He I | emission | 1s.2p 1P* → 1s.5p 1P* | Measured | NIST | |
| 438.7929143 nm | 10 | He I | emission | 1s.2p 1P* → 1s.5d 1D | Measured | NIST | |
| 443.7553428 nm | 3 | He I | emission | 1s.2p 1P* → 1s.5s 1S | Measured | NIST | |
| 447.1470373 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 447.1474077 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 447.1474317 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 447.1485658 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 447.1489362 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 447.1683251 nm | 25 | He I | emission | 1s.2p 3P* → 1s.4d 3D | Measured | NIST | |
| 468.537685 nm | N/A | He II | emission | 3p 2P* → 4d 2D | Measured | NIST | |
| 468.5407226 nm | N/A | He II | emission | 3s 2S → 4p 2P* | Measured | NIST | |
| 468.5524404 nm | N/A | He II | emission | 3p 2P* → 4s 2S | Measured | NIST | |
| 468.5568006 nm | N/A | He II | emission | 3s 2S → 4p 2P* | Measured | NIST | |
| 468.570385 nm | N/A | He II | emission | 3d 2D → 4f 2F* | Measured | NIST | |
| 468.570438 nm | N/A | He II | emission | 3p 2P* → 4d 2D | Measured | NIST | |
| 468.575708 nm | N/A | He II | emission | 3d 2D → 4p 2P* | Measured | NIST | |
| 468.5757975 nm | N/A | He II | emission | 3p 2P* → 4d 2D | Measured | NIST | |
| 468.5804092 nm | N/A | He II | emission | 3d 2D → 4f 2F* | Measured | NIST | |
| 468.583089 nm | N/A | He II | emission | 3d 2D → 4f 2F* | Measured | NIST | |
| 468.5884123 nm | N/A | He II | emission | 3d 2D → 4p 2P* | Measured | NIST | |
| 468.5905553 nm | N/A | He II | emission | 3p 2P* → 4s 2S | Measured | NIST | |
| 468.5917885 nm | N/A | He II | emission | 3d 2D → 4p 2P* | Measured | NIST | |
| 471.3139173 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4s 3S | Measured | NIST | |
| 471.3156155 nm | N/A | He I | emission | 1s.2p 3P* → 1s.4s 3S | Measured | NIST | |
| 471.3375684 nm | 4 | He I | emission | 1s.2p 3P* → 1s.4s 3S | Measured | NIST | |
| 491.074748 nm | N/A | He I | emission | 1s.2p 1P* → 1s.4p 1P* | Measured | NIST | |
| 492.0612726 nm | N/A | He I | emission | 1s.2p 1P* → 1s.4f 1F* | Measured | NIST | |
| 492.1931036 nm | 20 | He I | emission | 1s.2p 1P* → 1s.4d 1D | Measured | NIST | |
| 501.567801 nm | 100 | He I | emission | 1s.2s 1S → 1s.3p 1P* | Measured | NIST | |
| 504.208749 nm | N/A | He I | emission | 1s.2s 1S → 1s.3d 1D | Measured | NIST | |
| 504.773857 nm | 10 | He I | emission | 1s.2p 1P* → 1s.4s 1S | Measured | NIST | |
| 587.443388 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 1D | Measured | NIST | |
| 587.446026 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 1D | Measured | NIST | |
| 587.559871 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 587.561397 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 587.561484 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 587.56251 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 587.564036 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 587.596628 nm | 100 | He I | emission | 1s.2p 3P* → 1s.3d 3D | Measured | NIST | |
| 655.976872 nm | N/A | He II | emission | 4p 2P* → 6d 2D | Measured | NIST | |
| 655.979395 nm | N/A | He II | emission | 4s 2S → 6p 2P* | Measured | NIST | |
| 655.98544 nm | N/A | He II | emission | 4p 2P* → 6s 2S | Measured | NIST | |
| 655.988733 nm | N/A | He II | emission | 4s 2S → 6p 2P* | Measured | NIST | |
| 656.005227 nm | N/A | He II | emission | 4d 2D → 6f 2F* | Measured | NIST | |
| 656.005274 nm | N/A | He II | emission | 4p 2P* → 6d 2D | Measured | NIST | |
| 656.008318 nm | N/A | He II | emission | 4d 2D → 6p 2P* | Measured | NIST | |
| 656.008387 nm | N/A | He II | emission | 4p 2P* → 6d 2D | Measured | NIST | |
| 656.01416 nm | N/A | He II | emission | 4f 2F* → 6g 2G | Measured | NIST | |
| 656.014176 nm | N/A | He II | emission | 4d 2D → 6f 2F* | Measured | NIST | |
| 656.015708 nm | N/A | He II | emission | 4f 2F* → 6d 2D | Measured | NIST | |
| 656.015732 nm | N/A | He II | emission | 4d 2D → 6f 2F* | Measured | NIST | |
| 656.016955 nm | N/A | He II | emission | 4p 2P* → 6s 2S | Measured | NIST | |
| 656.017657 nm | N/A | He II | emission | 4d 2D → 6p 2P* | Measured | NIST | |
| 656.018478 nm | N/A | He II | emission | 4f 2F* → 6g 2G | Measured | NIST | |
| 656.01882 nm | N/A | He II | emission | 4f 2F* → 6d 2D | Measured | NIST | |
| 656.018823 nm | N/A | He II | emission | 4d 2D → 6p 2P* | Measured | NIST | |
| 656.019412 nm | N/A | He II | emission | 4f 2F* → 6g 2G | Measured | NIST | |
| 656.02096 nm | N/A | He II | emission | 4f 2F* → 6d 2D | Measured | NIST | |
| 663.190187 nm | N/A | He I | emission | 1s.2p 1P* → 1s.3p 1P* | Measured | NIST | |
| 667.815174 nm | 100 | He I | emission | 1s.2p 1P* → 1s.3d 1D | Measured | NIST | |
| 667.967687 nm | N/A | He I | emission | 1s.2p 1P* → 1s.3d 3D | Measured | NIST | |
| 706.517716 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3s 3S | Measured | NIST | |
| 706.521532 nm | N/A | He I | emission | 1s.2p 3P* → 1s.3s 3S | Measured | NIST | |
| 706.570863 nm | 30 | He I | emission | 1s.2p 3P* → 1s.3s 3S | Measured | NIST | |
| 716.055563 nm | N/A | He I | emission | 1s.3s 3S → 1s.10p 3P* | Measured | NIST | |
| 716.055907 nm | N/A | He I | emission | 1s.3s 3S → 1s.10p 3P* | Measured | NIST | |
| 716.055935 nm | N/A | He I | emission | 1s.3s 3S → 1s.10p 3P* | Measured | NIST | |
| 728.13508 nm | 50 | He I | emission | 1s.2p 1P* → 1s.3s 1S | Measured | NIST | |
| 729.803204 nm | N/A | He I | emission | 1s.3s 3S → 1s.9p 3P* | Measured | NIST | |
| 729.803696 nm | N/A | He I | emission | 1s.3s 3S → 1s.9p 3P* | Measured | NIST | |
| 729.803736 nm | N/A | He I | emission | 1s.3s 3S → 1s.9p 3P* | Measured | NIST | |
| 749.984714 nm | N/A | He I | emission | 1s.3s 3S → 1s.8p 3P* | Measured | NIST | |
| 749.985457 nm | N/A | He I | emission | 1s.3s 3S → 1s.8p 3P* | Measured | NIST | |
| 749.985518 nm | N/A | He I | emission | 1s.3s 3S → 1s.8p 3P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Supply Risk & Economics
Noble Gas Properties
Phase Transitions & Allotropes
| Boiling point | 4.22 K |
| Critical point (temperature) | 5.19 K |
| Critical point (pressure) | 0.23 MPa |
| Triple point (temperature) | 2.18 K |
| Triple point (pressure) | 5.04 kPa |
Advanced Reference Data
Screening Constants (1)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3125 |
Isotope Decay Modes (9)
| Isotope | Mode | Intensity |
|---|---|---|
| 5 | n | 100% |
| 6 | B- | 100% |
| 6 | B-d | 0% |
| 7 | n | 100% |
| 8 | B- | 100% |
| 8 | B-n | 16% |
| 8 | B-t | 0.9% |
| 9 | n | 100% |
| 10 | 2n | 100% |
X‑ray Scattering Factors (501)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0 |
| 10.1617 | — | 0 |
| 10.3261 | — | 0 |
| 10.4931 | — | 0 |
| 10.6628 | — | 0 |
| 10.8353 | — | 0 |
| 11.0106 | — | 0 |
| 11.1886 | — | 0 |
| 11.3696 | — | 0 |
| 11.5535 | — | 0 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
7×10-6 milligrams per liter
References (1)
Sources
Sources of this element.
Except for hydrogen, helium is the most abundant element found in the universe. Helium is extracted from natural gas. In fact, all natural gas contains at least trace quantities of helium.
It has been detected spectroscopically in great abundance, especially in the hotter stars, and it is an important component in both the proton-proton reaction and the carbon cycle, which account for the energy of the sun and stars.
The helium content of the atmosphere is about 1 part in 200,000. While it is present in various radioactive minerals as a decay product, the bulk of the Free World's supply is obtained from wells in Texas, Oklahoma, and Kansas. Outside the United States, the only known helium extraction plants, in 1984 were in Eastern Europe (Poland), the USSR, and a few in India.
References (1)
- [6] Helium https://periodic.lanl.gov/2.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Helium.
The element property data was retrieved from publications.