Lithium (Li)
alkali-metalSolid
Standard Atomic Weight
6.94 u [6.938, 6.997]Electron configuration
[He] 2s1Melting point
180.5 °C (453.65 K)Boiling point
1341.85 °C (1615 K)Density
534 kg/m³Oxidation states
+1Electronegativity (Pauling)
0.98Ionization energy (1st)
Discovery year
1817Atomic radius
145 pmDetails
Lithium is the lightest metal and the first alkali metal. It forms Li⁺ very readily, yet its small ion gives lithium chemistry a distinctive hardness, strong hydration, and extensive organometallic chemistry. In nature it occurs only in compounds, mainly in brines, pegmatite minerals, and some clays. Its low atomic mass, high electrochemical potential, and ability to move reversibly through host materials make it central to rechargeable batteries.
Socket silvery metal. First member of group 1 of the periodic table. Lithium salts are used in psychomedicine.
The name derives from the Latin lithos for "stone" because lithium was thought to exist only in minerals at that time. It was discovered by the Swedish mineralogist Johan August Arfwedson in 1818 in the mineral petalite LiAl(Si2O5)2. Lithium was isolated in 1855 by the German chemists Robert Wilhelm Bunsen and Augustus Matthiessen.
Lithium was discovered in the mineral petalite (LiAl(Si2O5)2) by Johann August Arfvedson in 1817. It was first isolated by William Thomas Brande and Sir Humphrey Davy through the electrolysis of lithium oxide (Li2O). Today, larger amounts of the metal are obtained through the electrolysis of lithium chloride (LiCl). Lithium is not found free in nature and makes up only 0.0007% of the earth's crust.
From the Greek word lithos, stone. Discovered by Arfvedson in 1817. Lithium is the lightest of all metals, with a density only about half that of water.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
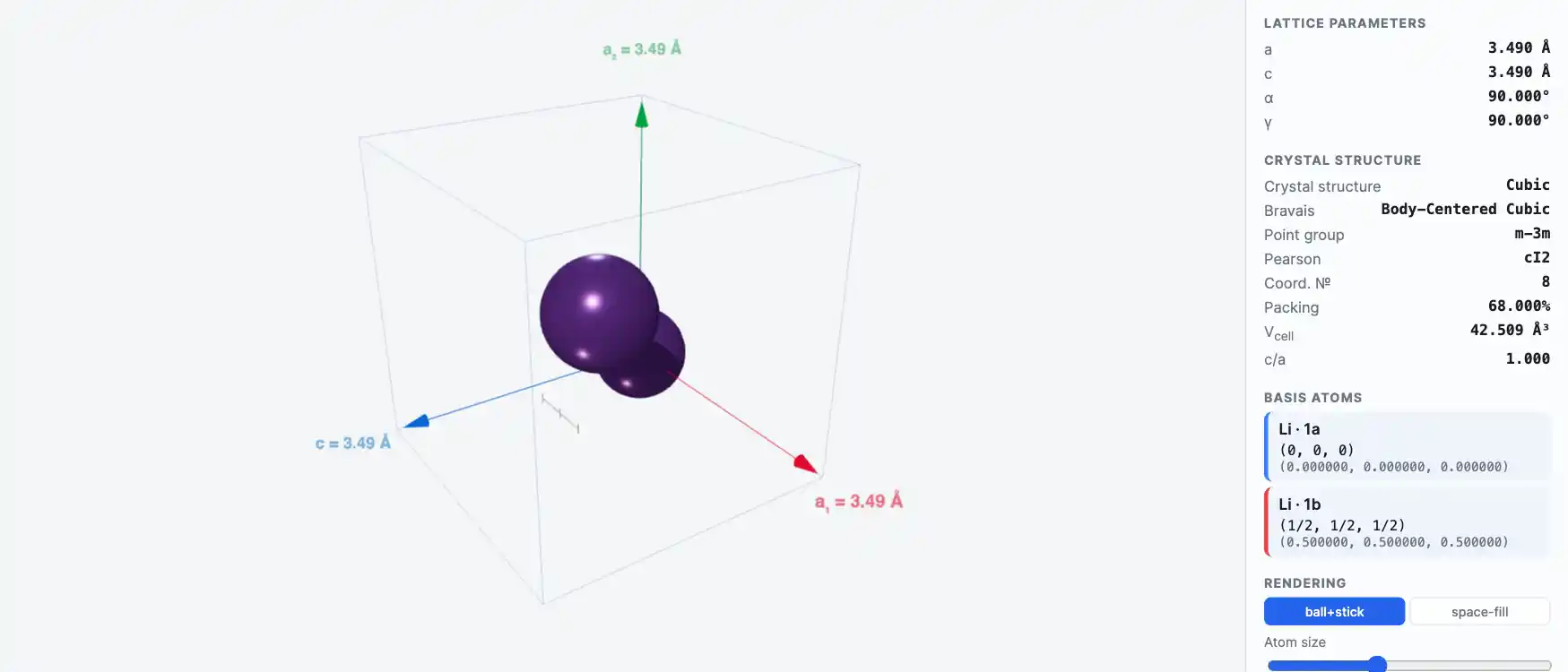
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Li: 2s¹[He] 2s¹1s² 2s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 6 Stable | 6.0151228874 ± 0.0000000016 | 7.5900% | Stable |
| 7 Stable | 7.0160034366 ± 0.0000000045 | 92.4100% | Stable |
Phase / State
Reason: 155.5 °C below melting point (180.5 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Li I | 0 | 344 | 257 | 328 |
| Li II | +1 | 663 | 564 | 630 |
| Li III | +2 | 144 | 144 | 144 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 4 | N/A | 59 pm |
| +1 | 6 | N/A | 76 pm |
| +1 | 8 | N/A | 92 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 6 Stable | 6.0151228874 ± 0.0000000016 | 7.5900% ± 0.0400% | Stable | stable | |
| 7 Stable | 7.0160034366 ± 0.0000000045 | 92.4100% ± 0.0400% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 383.559 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.7s 2S | Measured | NIST | |
| 383.564 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.7s 2S | Measured | NIST | |
| 387.8838 nm | N/A | Li II | emission | 1s.2s 3S → 1s.2p 1P* | Measured | NIST | |
| 391.5292 nm | 20 | Li I | emission | 1s2.2p 2P* → 1s2.6d 2D | Measured | NIST | |
| 391.5342 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.6d 2D | Measured | NIST | |
| 391.5344 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.6d 2D | Measured | NIST | |
| 398.5481 nm | 10 | Li I | emission | 1s2.2p 2P* → 1s2.6s 2S | Measured | NIST | |
| 398.5535 nm | 10 | Li I | emission | 1s2.2p 2P* → 1s2.6s 2S | Measured | NIST | |
| 413.2557 nm | 40 | Li I | emission | 1s2.2p 2P* → 1s2.5d 2D | Measured | NIST | |
| 413.2613 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.5d 2D | Measured | NIST | |
| 413.2615 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.5d 2D | Measured | NIST | |
| 415.519 nm | N/A | Li II | emission | 1s.3s 1S → 1s.4p 1P* | Measured | NIST | |
| 419.115 nm | N/A | Li II | emission | 1s.3s 1S → 1s.4d 1D | Measured | NIST | |
| 427.306 nm | 20 | Li I | emission | 1s2.2p 2P* → 1s2.5s 2S | Measured | NIST | |
| 427.312 nm | 20 | Li I | emission | 1s2.2p 2P* → 1s2.5s 2S | Measured | NIST | |
| 432.21 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 1D | Measured | NIST | |
| 432.226 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 1D | Measured | NIST | |
| 432.53 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 432.54 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 432.542 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 432.554 nm | 5 | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 432.562 nm | 1 | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 432.578 nm | N/A | Li II | emission | 1s.3p 3P* → 1s.4d 3D | Measured | NIST | |
| 449.8225057 nm | N/A | Li III | emission | 4p 2P* → 5d 2D | Measured | NIST | |
| 449.8277799 nm | N/A | Li III | emission | 4s 2S → 5p 2P* | Measured | NIST | |
| 449.8581249 nm | N/A | Li III | emission | 4p 2P* → 5s 2S | Measured | NIST | |
| 449.866202 nm | N/A | Li III | emission | 4s 2S → 5p 2P* | Measured | NIST | |
| 449.8846443 nm | N/A | Li III | emission | 4d 2D → 5f 2F* | Measured | NIST | |
| 449.8847466 nm | N/A | Li III | emission | 4p 2P* → 5d 2D | Measured | NIST | |
| 449.897364 nm | N/A | Li III | emission | 4d 2D → 5p 2P* | Measured | NIST | |
| 449.8975539 nm | N/A | Li III | emission | 4p 2P* → 5d 2D | Measured | NIST | |
| 449.9032229 nm | N/A | Li III | emission | 4f 2F* → 5g 2G | Measured | NIST | |
| 449.9032561 nm | N/A | Li III | emission | 4d 2D → 5f 2F* | Measured | NIST | |
| 449.9095915 nm | N/A | Li III | emission | 4f 2F* → 5d 2D | Measured | NIST | |
| 449.90966 nm | N/A | Li III | emission | 4d 2D → 5f 2F* | Measured | NIST | |
| 449.9118883 nm | N/A | Li III | emission | 4f 2F* → 5g 2G | Measured | NIST | |
| 449.9157307 nm | N/A | Li III | emission | 4f 2F* → 5g 2G | Measured | NIST | |
| 449.9220996 nm | N/A | Li III | emission | 4f 2F* → 5d 2D | Measured | NIST | |
| 449.9223809 nm | N/A | Li III | emission | 4d 2D → 5p 2P* | Measured | NIST | |
| 449.9224003 nm | N/A | Li III | emission | 4f 2F* → 5d 2D | Measured | NIST | |
| 449.933185 nm | N/A | Li III | emission | 4p 2P* → 5s 2S | Measured | NIST | |
| 449.9357979 nm | N/A | Li III | emission | 4d 2D → 5p 2P* | Measured | NIST | |
| 460.282 nm | 13 | Li I | emission | 1s2.2p 2P* → 1s2.4d 2D | Measured | NIST | |
| 460.289 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.4d 2D | Measured | NIST | |
| 460.289 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.4d 2D | Measured | NIST | |
| 463.61 nm | N/A | Li II | emission | 1s.3d 1D → 1s.4p 1P* | Measured | NIST | |
| 467.14 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 1F* | Measured | NIST | |
| 467.153 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 1F* | Measured | NIST | |
| 467.163 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.163 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.176 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.176 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.176 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.188 nm | 2 | Li II | emission | 1s.3d 3D → 1s.4f 3F* | Measured | NIST | |
| 467.806 nm | 3 | Li II | emission | 1s.3d 1D → 1s.4f 1F* | Measured | NIST | |
| 467.829 nm | N/A | Li II | emission | 1s.3d 1D → 1s.4f 3F* | Measured | NIST | |
| 467.829 nm | 1 | Li II | emission | 1s.3d 1D → 1s.4f 3F* | Measured | NIST | |
| 474.15 nm | N/A | Li II | emission | 1s.3p 1P* → 1s.4p 1P* | Measured | NIST | |
| 478.836 nm | N/A | Li II | emission | 1s.3p 1P* → 1s.4d 1D | Measured | NIST | |
| 479.239 nm | N/A | Li II | emission | 1s.3p 1P* → 1s.4d 3D | Measured | NIST | |
| 484.278 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 484.292 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 484.294 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 484.304 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 484.321 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 484.331 nm | N/A | Li II | emission | 1s.3d 3D → 1s.4p 3P* | Measured | NIST | |
| 488.12 nm | 4 | Li II | emission | 1s.3p 3P* → 1s.4s 3S | Measured | NIST | |
| 488.147 nm | 4 | Li II | emission | 1s.3p 3P* → 1s.4s 3S | Measured | NIST | |
| 488.169 nm | 1 | Li II | emission | 1s.3p 3P* → 1s.4s 3S | Measured | NIST | |
| 491.912 nm | N/A | Li II | emission | 1s.3d 1D → 1s.4s 1S | Measured | NIST | |
| 497.166 nm | 8 | Li I | emission | 1s2.2p 2P* → 1s2.4s 2S | Measured | NIST | |
| 497.174 nm | 8 | Li I | emission | 1s2.2p 2P* → 1s2.4s 2S | Measured | NIST | |
| 503.791 nm | N/A | Li II | emission | 1s.3p 1P* → 1s.4s 1S | Measured | NIST | |
| 510.8 nm | N/A | Li II | emission | 1s.4s 1S → 1s.7p 1P* | Measured | NIST | |
| 519.917 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 519.917 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 519.919 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 519.928 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 519.937 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 519.947 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7d 3D | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 527 nm | N/A | Li I | emission | 1s.2s.3d 4D → 1s.2p.3d 4D* | Measured | NIST | |
| 532.949 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7s 3S | Measured | NIST | |
| 532.96 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7s 3S | Measured | NIST | |
| 532.98 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.7s 3S | Measured | NIST | |
| 539.3 nm | N/A | Li II | emission | 1s.4d 1D → 1s.7p 1P* | Measured | NIST | |
| 540.153 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 1F* | Measured | NIST | |
| 540.172 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 1F* | Measured | NIST | |
| 540.175 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.186 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.186 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.205 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.205 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.205 nm | N/A | Li II | emission | 1s.4d 3D → 1s.7f 3F* | Measured | NIST | |
| 540.665 nm | N/A | Li II | emission | 1s.4d 1D → 1s.7f 1F* | Measured | NIST | |
| 540.698 nm | N/A | Li II | emission | 1s.4d 1D → 1s.7f 3F* | Measured | NIST | |
| 540.698 nm | N/A | Li II | emission | 1s.4d 1D → 1s.7f 3F* | Measured | NIST | |
| 541.091 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 1D | Measured | NIST | |
| 541.122 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.7d 1D | Measured | NIST | |
| 541.205 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.205 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.205 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.225 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.225 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.236 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.7d 3D | Measured | NIST | |
| 541.237 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.7d 3D | Measured | NIST | |
| 541.256 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.7d 3D | Measured | NIST | |
| 546.84 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.7d 1D | Measured | NIST | |
| 548.346 nm | N/A | Li II | emission | 1s.2s 3S → 1s.2p 3P* | Measured | NIST | |
| 548.44 nm | N/A | Li II | emission | 1s.2s 3S → 1s.2p 3P* | Measured | NIST | |
| 548.509 nm | N/A | Li II | emission | 1s.2s 3S → 1s.2p 3P* | Measured | NIST | |
| 552.54 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.7s 1S | Measured | NIST | |
| 565.388 nm | N/A | Li II | emission | 1s.4s 3S → 1s.6p 3P* | Measured | NIST | |
| 565.409 nm | N/A | Li II | emission | 1s.4s 3S → 1s.6p 3P* | Measured | NIST | |
| 565.421 nm | N/A | Li II | emission | 1s.4s 3S → 1s.6p 3P* | Measured | NIST | |
| 610.353 nm | 320 | Li I | emission | 1s2.2p 2P* → 1s2.3d 2D | Measured | NIST | |
| 610.364 nm | N/A | Li I | emission | 1s2.2p 2P* → 1s2.3d 2D | Measured | NIST | |
| 610.366 nm | 320 | Li I | emission | 1s2.2p 2P* → 1s2.3d 2D | Measured | NIST | |
| 611.81 nm | N/A | Li II | emission | 1s.4s 1S → 1s.6p 1P* | Measured | NIST | |
| 613.864 nm | N/A | Li II | emission | 1s.4s 1S → 1s.6d 1D | Measured | NIST | |
| 625.219 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 625.219 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 625.222 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 625.235 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 625.248 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 625.263 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6d 3D | Measured | NIST | |
| 653.14 nm | N/A | Li II | emission | 1s.4d 1D → 1s.6p 1P* | Measured | NIST | |
| 654.566 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 1F* | Measured | NIST | |
| 654.595 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 1F* | Measured | NIST | |
| 654.595 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 654.611 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 654.611 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 654.64 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 654.64 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 654.64 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6f 3F* | Measured | NIST | |
| 655.319 nm | N/A | Li II | emission | 1s.4d 1D → 1s.6f 1F* | Measured | NIST | |
| 655.364 nm | N/A | Li II | emission | 1s.4d 1D → 1s.6f 3F* | Measured | NIST | |
| 655.364 nm | N/A | Li II | emission | 1s.4d 1D → 1s.6f 3F* | Measured | NIST | |
| 656.006 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 1D | Measured | NIST | |
| 656.052 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.6d 1D | Measured | NIST | |
| 656.143 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6s 3S | Measured | NIST | |
| 656.16 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6s 3S | Measured | NIST | |
| 656.191 nm | N/A | Li II | emission | 1s.4p 3P* → 1s.6s 3S | Measured | NIST | |
| 656.261 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.261 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.261 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.29 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.29 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.306 nm | N/A | Li II | emission | 1s.4f 3F* → 1s.6d 3D | Measured | NIST | |
| 656.307 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.6d 3D | Measured | NIST | |
| 656.336 nm | N/A | Li II | emission | 1s.4f 1F* → 1s.6d 3D | Measured | NIST | |
| 662.07 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.6p 1P* | Measured | NIST | |
| 664.252 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.269 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.281 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.298 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.298 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.298 nm | N/A | Li II | emission | 1s.4d 3D → 1s.6p 3P* | Measured | NIST | |
| 664.48 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.6d 1D | Measured | NIST | |
| 664.77 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.6d 3D | Measured | NIST | |
| 668.73 nm | N/A | Li II | emission | 1s.4d 1D → 1s.6s 1S | Measured | NIST | |
| 670.776 nm | 3600 | Li I | emission | 1s2.2s 2S → 1s2.2p 2P* | Measured | NIST | |
| 670.791 nm | 3600 | Li I | emission | 1s2.2s 2S → 1s2.2p 2P* | Measured | NIST | |
| 678.09 nm | N/A | Li II | emission | 1s.4p 1P* → 1s.6s 1S | Measured | NIST | |
| 687.308 nm | N/A | Li I | emission | 1s2.3s 2S → 1s2.8p 2P* | Measured | NIST | |
| 687.308 nm | N/A | Li I | emission | 1s2.3s 2S → 1s2.8p 2P* | Measured | NIST | |
| 713.517 nm | N/A | Li I | emission | 1s2.3s 2S → 1s2.7p 2P* | Measured | NIST | |
| 713.517 nm | N/A | Li I | emission | 1s2.3s 2S → 1s2.7p 2P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 453.65 K |
| Boiling point | 1615.15 K |
| Critical point (temperature) | 3223.15 K |
| Critical point (pressure) | 67 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (2)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3094 |
| 2 | s | 1.7208 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | IV | 73 | ||
| 1 | VI | 90 | ||
| 1 | VIII | 106 | calculated, |
Isotope Decay Modes (17)
| Isotope | Mode | Intensity |
|---|---|---|
| 3 | p | — |
| 4 | p | 100% |
| 5 | p | 100% |
| 8 | B- | 100% |
| 8 | B-A | 100% |
| 9 | B- | 100% |
| 9 | B-n | 50.5% |
| 10 | n | 100% |
| 11 | B- | 100% |
| 11 | B-n | 86.3% |
X‑ray Scattering Factors (501)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.11642 |
| 10.1617 | — | 0.11743 |
| 10.3261 | — | 0.11844 |
| 10.4931 | — | 0.11947 |
| 10.6628 | — | 0.12051 |
| 10.8353 | — | 0.12155 |
| 11.0106 | — | 0.12261 |
| 11.1886 | — | 0.12367 |
| 11.3696 | — | 0.12444 |
| 11.5535 | — | 0.12502 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.0×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.8×10-1 milligrams per liter
References (1)
Sources
Sources of this element.
It does not occur freely in nature; combined, it is found in small units in nearly all igneous rocks and in many mineral springs. Lepidolite, spodumene, petalite, and amblygonite are the more important minerals containing it.
Lithium is presently being recovered from brines of Searles Lake, in California, and from those in Nevada. Large deposits of quadramene are found in North Carolina. The metal is produced electrolytically from the fused chloride. Lithium is silvery in appearance, much like Na, K, and other members of the alkali metal series. It reacts with water, but not as vigorously as sodium. Lithium imparts a beautiful crimson color to a flame, but when the metal burns strongly, the flame is a dazzling white.
References (1)
- [6] Lithium https://periodic.lanl.gov/3.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Lithium.
The element property data was retrieved from publications.