Titanium (Ti)
transition-metalSolid
Standard Atomic Weight
47.867 uElectron configuration
[Ar] 4s2 3d2Melting point
1667.85 °C (1941 K)Boiling point
3286.85 °C (3560 K)Density
4500 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
1.54Ionization energy (1st)
Discovery year
1791Atomic radius
140 pmDetails
Titanium is a light, strong transition metal with a high melting point and exceptional resistance to corrosion in many natural and industrial environments. Its chemistry is dominated by the +4 oxidation state, although +3 and lower states occur in specialized compounds. The metal is abundant in Earth’s crust but is rarely found in concentrated metallic form because it bonds strongly to oxygen and nitrogen. Its combination of low density, strength, and passivation makes it important in aerospace, chemical equipment, pigments, and medical materials.
Titanium, when pure, is a lustrous, white metal. It has a low density, good strength, is easily fabricated, and has excellent corrosion resistance. It is ductile only when it is free of oxygen. The metal, which burns in air, is the only element that burns in nitrogen.
Titanium is resistant to dilute sulfuric and hydrochloric acid, most organic acids, most chlorine gas, and chloride solutions.
Natural titanium is reported to become very radioactive after bombardment with deuterons. The emitted radiations are mostly positrons and hard gamma rays. The metal is dimorphic. The hexagonal alpha form changes to the cubic beta form very slowly at about 880°C. The metal combines with oxygen at red heat, and with chlorine at 550°C.
Titanium metal is considered to be physiologically inert. When pure, titanium dioxide is relatively clear and has an extremely high index of refraction with an optical dispersion higher than diamond.
The name derives from the Latin titans, who were the mythological "first sons of the earth". It was originally discovered by the English clergyman William Gregor in the mineral ilmenite (FeTiO3) in 1791. He called this mineral menachanite and the element menachin, for the Menachan parish where it was found. It was rediscovered in 1795 by the German chemist Martin Heinrich Klaproth, who called it titanium because it had no characteristic properties to use as a name. Titanium metal was first isolated by the Swedish chemists Sven Otto Pettersson and Lars Fredrik Nilson.
Titanium was discovered in 1791 by the Reverend William Gregor, an English pastor. Pure titanium was first produced by Matthew A. Hunter, an American metallurgist, in 1910. Titanium is the ninth most abundant element in the earth's crust and is primarily found in the minerals rutile (TiO2), ilmenite (FeTiO3) and sphene (CaTiSiO5). Titanium makes up about 0.57% of the earth's crust.
From the Latin titans, the first sons of the Earth, Greek mythology.
Discovered by Gregor in 1791; named by Klaproth in 1795. Impure titanium was prepared by Nilson and Pettersson in 1887; however, the pure metal (99.9%) was not made until 1910 when Hunter heated TiCl4 with sodium in a steel bomb.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
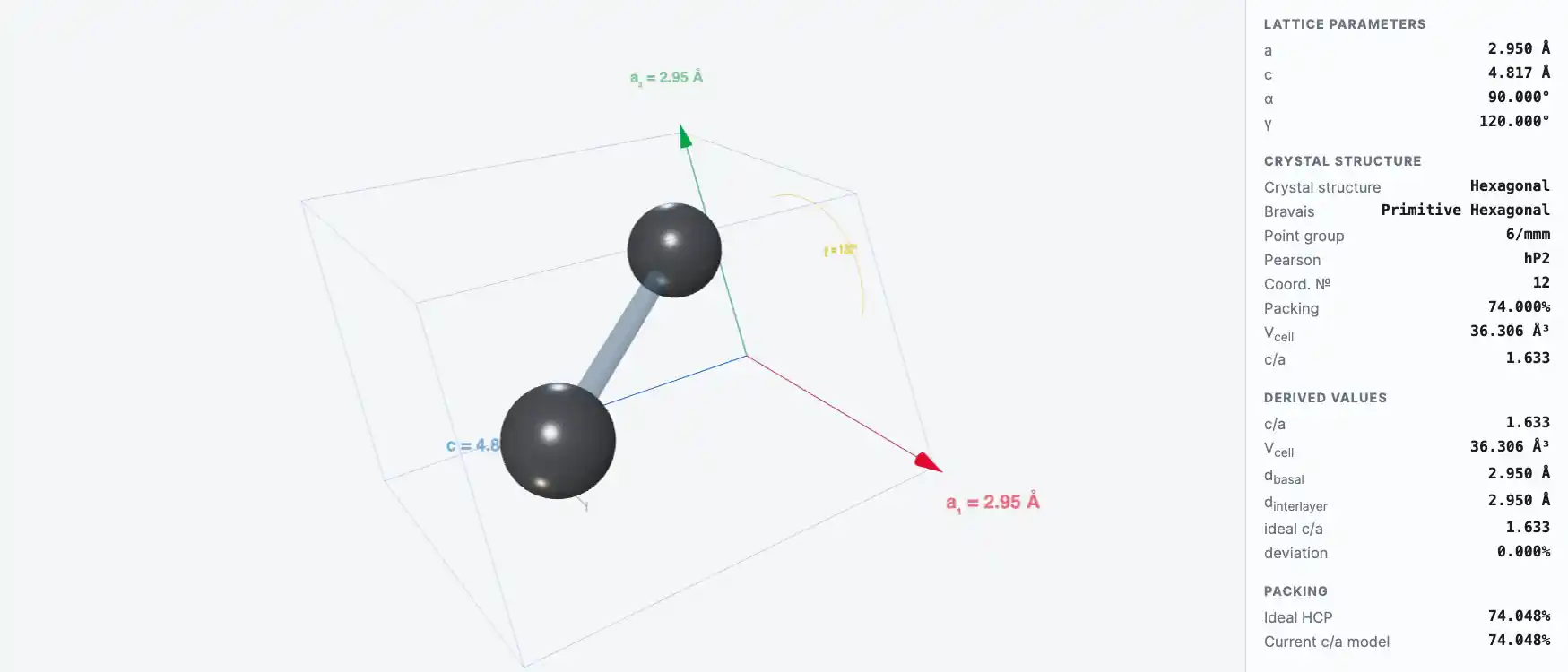
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ti: 3d² 4s²[Ar] 3d² 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d² 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 46 Stable | 45.95262772 ± 0.00000035 | 8.2500% | Stable |
| 47 Stable | 46.95175879 ± 0.00000038 | 7.4400% | Stable |
| 48 Stable | 47.94794198 ± 0.00000038 | 73.7200% | Stable |
| 49 Stable | 48.94786568 ± 0.00000039 | 5.4100% | Stable |
| 50 Stable | 49.94478689 ± 0.00000039 | 5.1800% | Stable |
Phase / State
Reason: 1642.8 °C below melting point (1667.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 22 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ti I | 0 | 4029 | 496 | 4029 |
| Ti II | +1 | 1872 | 470 | 1872 |
| Ti III | +2 | 819 | 297 | 819 |
| Ti IV | +3 | 86 | 39 | 86 |
| Ti V | +4 | 252 | 4 | 252 |
| Ti VI | +5 | 71 | 14 | 71 |
| Ti VII | +6 | 92 | 13 | 92 |
| Ti VIII | +7 | 85 | 37 | 85 |
| Ti IX | +8 | 85 | 50 | 85 |
| Ti X | +9 | 162 | 78 | 162 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ti I | 0 | 559 |
| Ti II | +1 | 253 |
| Ti III | +2 | 200 |
| Ti IV | +3 | 40 |
| Ti V | +4 | 66 |
| Ti VI | +5 | 59 |
| Ti VII | +6 | 62 |
| Ti VIII | +7 | 44 |
| Ti IX | +8 | 32 |
| Ti X | +9 | 83 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 86 pm |
| +3 | 6 | N/A | 67 pm |
| +4 | 4 | N/A | 42 pm |
| +4 | 5 | N/A | 51 pm |
| +4 | 6 | N/A | 60.5 pm |
| +4 | 8 | N/A | 74 pm |
Compounds
Isotopes (5)
Natural titanium consists of five isotopes with atomic masses from 46 to 50. All are stable. Eight other unstable isotopes are known.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 46 Stable | 45.95262772 ± 0.00000035 | 8.2500% ± 0.0300% | Stable | stable | |
| 47 Stable | 46.95175879 ± 0.00000038 | 7.4400% ± 0.0200% | Stable | stable | |
| 48 Stable | 47.94794198 ± 0.00000038 | 73.7200% ± 0.0300% | Stable | stable | |
| 49 Stable | 48.94786568 ± 0.00000039 | 5.4100% ± 0.0200% | Stable | stable | |
| 50 Stable | 49.94478689 ± 0.00000039 | 5.1800% ± 0.0200% | Stable | stable |
Spectral Lines
Showing 50 of 1717 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 521.03843 nm | 21000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3F* | Measured | NIST | |
| 506.46526 nm | 17000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 519.29686 nm | 17000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3F* | Measured | NIST | |
| 517.37431 nm | 15000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3F* | Measured | NIST | |
| 498.17305 nm | 14000 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 503.99574 nm | 14000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 468.19089 nm | 13000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3G* | Measured | NIST | |
| 499.1066 nm | 13000 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 499.9503 nm | 12000 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 501.41861 nm | 11000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 399.86363 nm | 10000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(1P*) y 3F* | Measured | NIST | |
| 466.75845 nm | 10000 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3G* | Measured | NIST | |
| 500.72093 nm | 10000 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 453.32394 nm | 9200 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 398.17616 nm | 8800 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(1P*) y 3F* | Measured | NIST | |
| 398.97582 nm | 8800 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(1P*) y 3F* | Measured | NIST | |
| 501.42762 nm | 8700 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 395.82055 nm | 8600 | Ti I | emission | 3d2.4s2 a 3F → 3d3.(4F).4p y 3D* | Measured | NIST | |
| 465.64693 nm | 8400 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(3P*) z 3G* | Measured | NIST | |
| 395.63338 nm | 8000 | Ti I | emission | 3d2.4s2 a 3F → 3d3.(4F).4p y 3D* | Measured | NIST | |
| 453.47761 nm | 7900 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 394.86705 nm | 7000 | Ti I | emission | 3d2.4s2 a 3F → 3d3.(4F).4p y 3D* | Measured | NIST | |
| 484.08737 nm | 6600 | Ti I | emission | 3d2.4s2 a 1D → 3d2.(1D).4s.4p.(1P*) y 1D* | Measured | NIST | |
| 430.59074 nm | 6400 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p x 5D* | Measured | NIST | |
| 453.55686 nm | 6100 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 394.77683 nm | 5700 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(1D).4s.4p.(3P*) 3P* | Measured | NIST | |
| 502.00263 nm | 5100 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 430.10787 nm | 4900 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p x 5D* | Measured | NIST | |
| 503.5903 nm | 4900 | Ti I | emission | 3d3.(4F).4s b 3F → 3d3.(4F).4p w 3G* | Measured | NIST | |
| 502.28679 nm | 4800 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 453.59176 nm | 4700 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 430.05538 nm | 4400 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p x 5D* | Measured | NIST | |
| 453.60403 nm | 4000 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 503.64639 nm | 4000 | Ti I | emission | 3d3.(4F).4s b 3F → 3d3.(4F).4p w 3G* | Measured | NIST | |
| 501.61609 nm | 3800 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 451.8022 nm | 3700 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 488.50794 nm | 3700 | Ti I | emission | 3d3.(2G).4s a 3G → 3d3.(2G).4p y 3H* | Measured | NIST | |
| 392.45264 nm | 3600 | Ti I | emission | 3d2.4s2 a 3F → 3d3.(4F).4p y 3D* | Measured | NIST | |
| 402.45711 nm | 3600 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(1P*) y 3F* | Measured | NIST | |
| 390.47826 nm | 3500 | Ti I | emission | 3d2.4s2 a 1D → 3d2.(1D).4s.4p.(1P*) y 1F* | Measured | NIST | |
| 452.2797 nm | 3500 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 502.48444 nm | 3500 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5G* | Measured | NIST | |
| 398.24811 nm | 3400 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3P).4s.4p.(3P*) z 5S* | Measured | NIST | |
| 454.87635 nm | 3400 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 455.24533 nm | 3400 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p y 5F* | Measured | NIST | |
| 400.89274 nm | 3300 | Ti I | emission | 3d2.4s2 a 3F → 3d2.(3F).4s.4p.(1P*) y 3F* | Measured | NIST | |
| 503.83979 nm | 3300 | Ti I | emission | 3d3.(4F).4s b 3F → 3d3.(4F).4p w 3G* | Measured | NIST | |
| 392.98737 nm | 3200 | Ti I | emission | 3d2.4s2 a 3F → 3d3.(4F).4p y 3D* | Measured | NIST | |
| 429.86657 nm | 3200 | Ti I | emission | 3d3.(4F).4s a 5F → 3d3.(4F).4p x 5D* | Measured | NIST | |
| 489.99088 nm | 3200 | Ti I | emission | 3d3.(2G).4s a 3G → 3d3.(2G).4p y 3H* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1943.15 K |
| Boiling point | 3560.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.5591 |
| 2 | p | 3.9352 |
| 2 | s | 6.6234 |
| 3 | d | 13.8586 |
| 3 | p | 11.8963 |
| 3 | s | 10.9669 |
| 4 | s | 17.1832 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 100 | estimated, | |
| 3 | VI | 81 | from r^3 vs V plots, | |
| 4 | IV | 56 | calculated, | |
| 4 | V | 65 | calculated, | |
| 4 | VI | 74.5 | from r^3 vs V plots, | |
| 4 | VIII | 88 | calculated, |
Isotope Decay Modes (47)
| Isotope | Mode | Intensity |
|---|---|---|
| 37 | p | — |
| 38 | 2p | — |
| 39 | B+ | 100% |
| 39 | B+p | 93.7% |
| 39 | 2p | — |
| 40 | B+ | 100% |
| 40 | B+p | 95.8% |
| 41 | B+ | 100% |
| 41 | B+p | 91.1% |
| 42 | B+ | 100% |
X‑ray Scattering Factors (530)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.51668 |
| 10.1428 | — | 1.54246 |
| 10.3068 | — | 1.57217 |
| 10.4735 | — | 1.60245 |
| 10.6429 | — | 1.63331 |
| 10.8151 | — | 1.66477 |
| 10.99 | — | 1.70636 |
| 11.1677 | — | 1.75257 |
| 11.3484 | — | 1.80003 |
| 11.5319 | — | 1.84878 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5.65×103 milligrams per kilogram
References (1)
- [5] Titanium https://education.jlab.org/itselemental/ele022.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1×10-3 milligrams per liter
References (1)
- [5] Titanium https://education.jlab.org/itselemental/ele022.html
Sources
Sources of this element.
Titanium is present in meteorites and the sun. Rocks obtained during the Apollo 17 lunar mission showed presence of 12.1% TiO2; rocks obtained during earlier Apollo missions show lower percentages.
Titanium oxide bands are prominent in the spectra of M-type stars. The element is the ninth most abundant in the crust of the earth. Titanium is almost always present in igneous rocks and in the sediments derived from them.
It occurs in the minerals rutile, ilmenite, and sphene, and is present in titanates and in many iron ores. Titanium is present in ash of coal, in plants, and in human body.
The metal was a laboratory curiosity until Kroll, in 1946, showed that titanium could be produced commercially by reducing titanium tetrachloride with magnesium. This method is still largely used for producing the metal. The metal can be purified by decomposing the iodide.
References (1)
- [6] Titanium https://periodic.lanl.gov/22.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Titanium.
The element property data was retrieved from publications.