Vanadium (V)
transition-metalSolid
Standard Atomic Weight
50.9415 uElectron configuration
[Ar] 4s2 3d3Melting point
1909.85 °C (2183 K)Boiling point
3406.85 °C (3680 K)Density
6000 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
1.63Ionization energy (1st)
Discovery year
1830Atomic radius
135 pmDetails
Vanadium is a hard early transition metal with variable oxidation states and strong affinity for oxygen, nitrogen, and carbon. It occurs mainly dispersed in minerals rather than as native metal. Its technological importance comes chiefly from alloying steel and from vanadium redox-flow batteries. Chemically it is notable for accessible +2, +3, +4, and +5 states, often producing distinctly colored ions and oxides.
Pure vanadium is a bright white metal, and is soft and ductile. It has good corrosion resistance to alkalis, sulfuric and hydrochloric acid, and salt water, but the metal oxidizes readily above 660°C.
The metal has good structural strength and a low fission neutron cross section, making it useful in nuclear applications.
The name derives from the Scandinavian goddess of love and beauty, Freyja Vanadis, because of its many beautiful multi-coloured compounds. Vanadium was discovered by the Swedish physician and chemist Nils-Gabriel Sefström in 1830.
Vanadium had originally been discovered by the Spanish mineralogist Andres Manuel del Rio y Fernandez in 1801, who named it erythronium, after the plant of that name whose flowers have many beautiful colours. Del Rio later decided that it was really chromium in his lead sample. Vanadium metal was first isolated by the English chemist Henry Enfield Roscoe in 1869.
Vanadium was discovered by Andrés Manuel del Rio, a Spanish chemist, in 1801. Rio sent samples of vanadium ore and a letter describing his methods to the Institute de France in Paris, France, for analysis and confirmation. Unfortunately for Rio, his letter was lost in a shipwreck and the Institute only received his samples, which contained a brief note describing how much this new element, which Rio had named erythronium, resembled chromium. Rio withdrew his claim when he received a letter from Paris disputing his discovery. Vanadium was rediscovered by Nils Gabriel Sefstrôm, a Swedish chemist, in 1830 while analyzing samples of iron from a mine in Sweden. Vanadium was isolated by Sir Henry Enfield Roscoe, an English chemist, in 1867 by combining vanadium trichloride (VCl3) with hydrogen gas (H2). Today, vanadium is primarily obtained from the minerals vanadinite (Pb5(VO)3Cl) and carnotite (K2(UO2)2VO4·1-3H2O) by heating crushed ore in the presence of carbon and chlorine to produce vanadium trichloride. The vanadium trichloride is then heated with magnesium in an argon atmosphere.
Named after Scandinavian goddess, Vanadis. Vanadium was first discovered by del Rio in 1801. Unfortunately, a French chemist incorrectly declared that del Rio's new element was only impure chromium. Del Rio thought himself to be mistaken and accepted the French chemists' statement.
The element was rediscovered in 1830 by Sefstrom, who named the element in honor of the Scandinavian goddess, Vanadis, because of its beautiful multicolored compounds. It was isolated in nearly pure form by Roscoe, who in 1867 reduced the chloride with hydrogen.
Vanadium of 99.3 to 99.8% purity was not produced until 1922.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
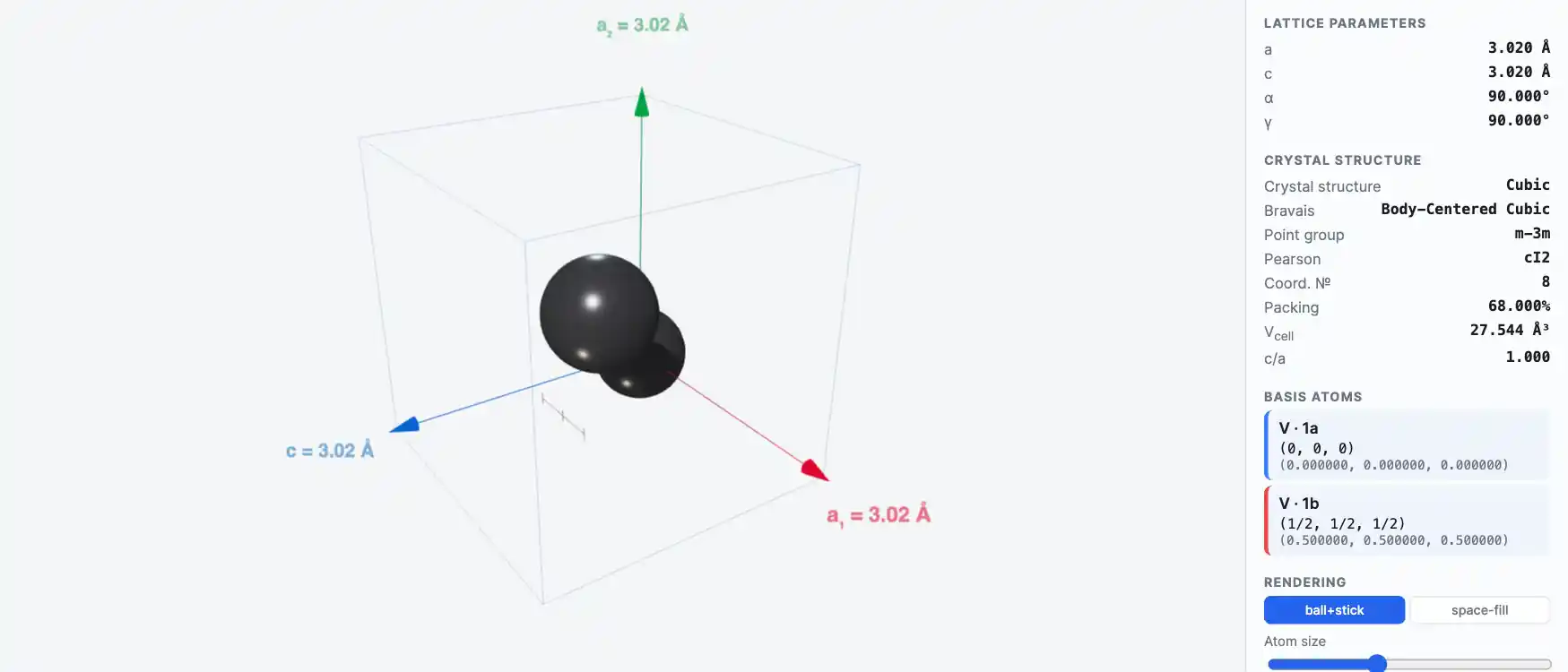
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
V: 3d³ 4s²[Ar] 3d³ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d³ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 51 Stable | 50.94395704 ± 0.00000094 | 99.7500% | Stable |
Phase / State
Reason: 1884.8 °C below melting point (1909.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 23 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| V I | 0 | 3985 | 1256 | 3985 |
| V II | +1 | 3568 | 1896 | 3568 |
| V III | +2 | 94 | 30 | 30 |
| V IV | +3 | 423 | 300 | 423 |
| V V | +4 | 164 | 10 | 164 |
| V VI | +5 | 175 | 4 | 175 |
| V VII | +6 | 39 | 9 | 39 |
| V VIII | +7 | 69 | 19 | 69 |
| V IX | +8 | 72 | 44 | 72 |
| V X | +9 | 69 | 45 | 69 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| V I | 0 | 550 |
| V II | +1 | 408 |
| V III | +2 | 300 |
| V IV | +3 | 100 |
| V V | +4 | 71 |
| V VI | +5 | 62 |
| V VII | +6 | 35 |
| V VIII | +7 | 52 |
| V IX | +8 | 39 |
| V X | +9 | 28 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 79 pm |
| +3 | 6 | N/A | 64 pm |
| +4 | 5 | N/A | 53 pm |
| +4 | 6 | N/A | 57.99999999999999 pm |
| +4 | 8 | N/A | 72 pm |
| +5 | 4 | N/A | 35.5 pm |
| +5 | 5 | N/A | 46 pm |
| +5 | 6 | N/A | 54 pm |
Compounds
Isotopes (1)
Natural vanadium is a mixture of two isotopes, 50V (0.24%) and 51V (99.76%). 50V is slightly radioactive, having a half-life of> 3.9 x 1017 years. Nine other unstable isotopes are recognized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 51 Stable | 50.94395704 ± 0.00000094 | 99.7500% ± 0.0040% | Stable | stable |
Spectral Lines
Showing 50 of 2461 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 437.92304 nm | 74000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 411.17788 nm | 53000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 438.4713 nm | 44000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 438.99793 nm | 30000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 440.85162 nm | 29000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 411.51768 nm | 25000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 439.52233 nm | 23000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 440.81958 nm | 23000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 385.58404 nm | 18000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4D* | Measured | NIST | |
| 412.80642 nm | 18000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 413.19909 nm | 18000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 409.97833 nm | 17000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 410.5157 nm | 17000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 440.76338 nm | 17000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 384.074941 nm | 16000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4D* | Measured | NIST | |
| 390.22531 nm | 14000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 410.97575 nm | 14000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 413.44835 nm | 14000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 440.66382 nm | 14000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 446.02914 nm | 13000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p z 6P* | Measured | NIST | |
| 412.34985 nm | 12000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 409.26831 nm | 11000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 411.64716 nm | 11000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6D* | Measured | NIST | |
| 382.855694 nm | 10000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4D* | Measured | NIST | |
| 387.507162 nm | 9000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 459.41158 nm | 8900000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4G* | Measured | NIST | |
| 440.05717 nm | 8800000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 609.02084 nm | 8100000 | V I | emission | 3d4.(5D).4s a 4D → 3d4.(5D).4p z 4P* | Measured | NIST | |
| 386.48561 nm | 7900000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 381.82414 nm | 7800000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4D* | Measured | NIST | |
| 569.85189 nm | 7200000 | V I | emission | 3d4.(5D).4s a 4D → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 435.28654 nm | 6600000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4F* | Measured | NIST | |
| 445.97536 nm | 6300000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p z 6P* | Measured | NIST | |
| 381.349106 nm | 6000000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4D* | Measured | NIST | |
| 458.6366 nm | 5700000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4G* | Measured | NIST | |
| 570.3575 nm | 5600000 | V I | emission | 3d4.(5D).4s a 4D → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 624.31073 nm | 5500000 | V I | emission | 3d4.(5D).4s a 6D → 3d3.(4F).4s.4p.(3P*) z 6D* | Measured | NIST | |
| 409.0568 nm | 5300000 | V I | emission | 3d4.(5D).4s a 4D → 3d3.(4F).4s.4p.(1P*) w 4F* | Measured | NIST | |
| 488.15569 nm | 5300000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4D* | Measured | NIST | |
| 444.168 nm | 5200000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p z 6P* | Measured | NIST | |
| 572.70445 nm | 5100000 | V I | emission | 3d4.(5D).4s a 4D → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 434.0998 nm | 5000000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4F* | Measured | NIST | |
| 458.03967 nm | 4400000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4G* | Measured | NIST | |
| 487.54859 nm | 4400000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 4D* | Measured | NIST | |
| 409.54749 nm | 4300000 | V I | emission | 3d4.(5D).4s a 4D → 3d3.(4F).4s.4p.(1P*) w 4F* | Measured | NIST | |
| 389.01792 nm | 4200000 | V I | emission | 3d3.4s2 a 4F → 3d3.(4F).4s.4p.(3P*) z 2G* | Measured | NIST | |
| 390.98572 nm | 4200000 | V I | emission | 3d3.4s2 a 4F → 3d4.(5D).4p y 4F* | Measured | NIST | |
| 441.64662 nm | 4000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 442.15674 nm | 4000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p y 6F* | Measured | NIST | |
| 443.78304 nm | 4000000 | V I | emission | 3d4.(5D).4s a 6D → 3d4.(5D).4p z 6P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2183.15 K |
| Boiling point | 3680.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.5744 |
| 2 | p | 3.9272 |
| 2 | s | 6.8186 |
| 3 | d | 14.0171 |
| 3 | p | 12.215 |
| 3 | s | 11.2907 |
| 4 | s | 18.0188 |
Crystal Radii Detail (8)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 93 | ||
| 3 | VI | 78 | from r^3 vs V plots, | |
| 4 | V | 67 | ||
| 4 | VI | 72 | from r^3 vs V plots, | |
| 4 | VIII | 86 | estimated, | |
| 5 | IV | 49.5 | from r^3 vs V plots, | |
| 5 | V | 60 | ||
| 5 | VI | 68 |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 39 | p | — |
| 40 | p | — |
| 41 | p | — |
| 42 | p | — |
| 43 | B+ | 100% |
| 43 | B+p | 2.5% |
| 44 | B+ | 100% |
| 44 | B+A | — |
| 44 | B+p | — |
| 45 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.06459 |
| 10.1617 | — | 1.11805 |
| 10.3261 | — | 1.17419 |
| 10.4931 | — | 1.23315 |
| 10.6628 | — | 1.29507 |
| 10.8353 | — | 1.3601 |
| 11.0106 | — | 1.42839 |
| 11.1886 | — | 1.50012 |
| 11.3696 | — | 1.57258 |
| 11.5535 | — | 1.6378 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.20×102 milligrams per kilogram
References (1)
- [5] Vanadium https://education.jlab.org/itselemental/ele023.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2.5×10-3 milligrams per liter
References (1)
- [5] Vanadium https://education.jlab.org/itselemental/ele023.html
Sources
Sources of this element.
Vanadium is found in about 65 different minerals among which are carnotite, roscoelite, vanadinite, and patronite, important sources of the metal. Vanadium is also found in phosphate rock and certain iron ores, and is present in some crude oils in the form of organic complexes. It is also found in small percentages in meteorites.
Commercial production from petroleum ash holds promise as an important source of the element. High-purity ductile vanadium can be obtained by reduction of vanadium trichloride with magnesium or with magnesium-sodium mixtures.
Much of the vanadium metal being produced is now made by calcium reduction of V2O5 in a pressure vessel, an adaption of a process developed by McKechnie and Seybair.
References (1)
- [6] Vanadium https://periodic.lanl.gov/23.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Vanadium.
The element property data was retrieved from publications.