Chromium (Cr)
transition-metalSolid
Standard Atomic Weight
51.9961 uElectron configuration
[Ar] 3d5 4s1Melting point
1906.85 °C (2180 K)Boiling point
2670.85 °C (2944 K)Density
7150 kg/m³Oxidation states
−4, −2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
1.66Ionization energy (1st)
Discovery year
1797Atomic radius
140 pmDetails
Chromium is a hard transition metal best known for forming adherent, protective oxide films and for giving many minerals and compounds strong colors. It occurs mainly in chromite ores and is an important alloying element in stainless and heat-resisting steels. Its chemistry spans several oxidation states, especially +3 and +6, with a sharp contrast between relatively stable Cr(III) compounds and strongly oxidizing, often toxic Cr(VI) species.
Chromium is used extensively in automobile trim as chromium metal because of its shiny finish and corrosion resistance.
The name derives from the Greek chroma for "colour", from the many coloured compounds of chromium. It was discovered in 1797 by the French chemist and pharmacist Nicolas-Louis Vauquelin, who also isolated chromium in 1798.
Chromium was discovered by Louis-Nicholas Vauquelin while experimenting with a material known as Siberian red lead, also known as the mineral crocoite (PbCrO4), in 1797. He produced chromium oxide (CrO3) by mixing crocoite with hydrochloric acid (HCl). Although he believed a method for isolating chromium didn't yet exist, Vauquelin was pleasantly surprised in 1798 to discover that he was able to obtain metallic chromium by simply heating chromium oxide in a charcoal oven. Today, chromium is primarily obtained by heating the mineral chromite (FeCr2O4) in the presence of aluminum or silicon.
From the Greek word chroma, color. Chromium is a steel-gray, lustrous, hard metal that takes a high polish. Discovered in 1797 by the Frenchman Louis Nicolas Vauquelin.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
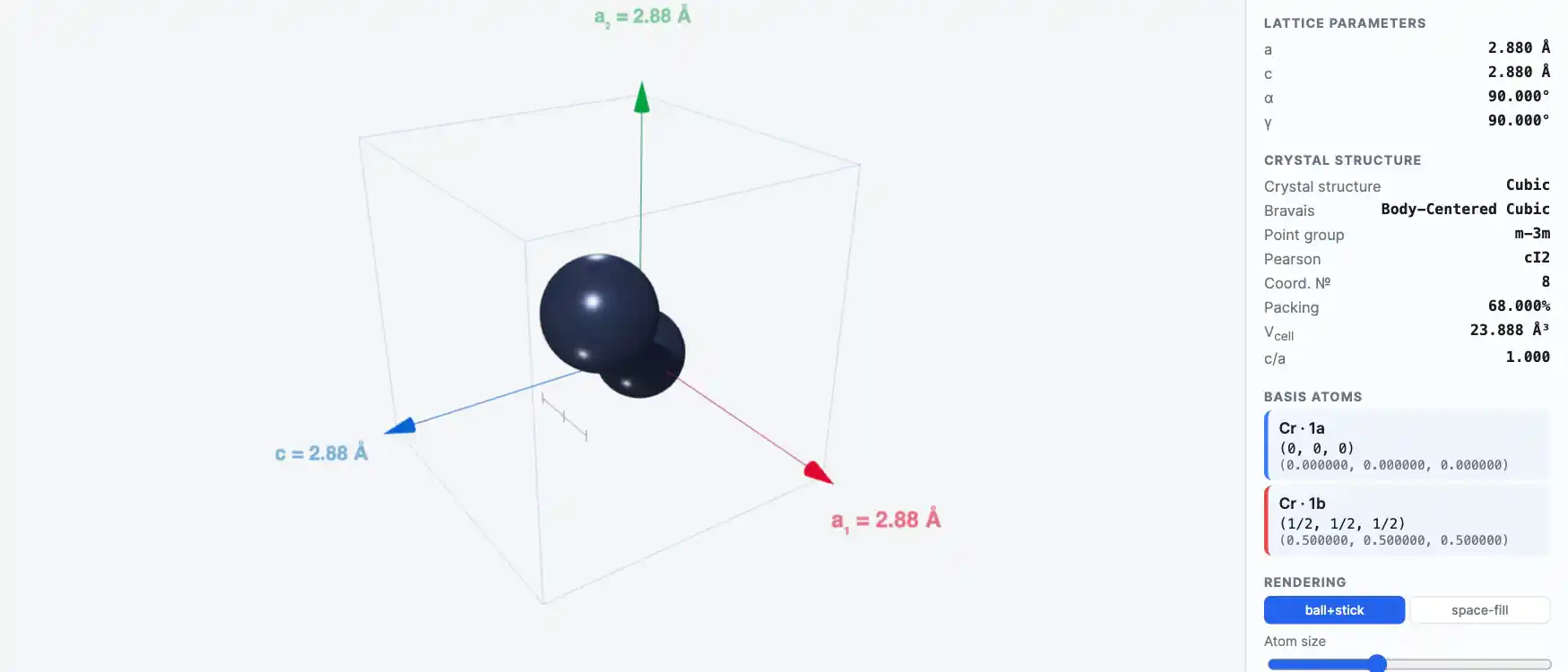
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Cr: 3d⁵ 4s¹[Ar] 3d⁵ 4s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁵ 4s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 52 Stable | 51.94050623 ± 0.00000063 | 83.7890% | Stable |
| 53 Stable | 52.94064815 ± 0.00000062 | 9.5010% | Stable |
| 54 Stable | 53.93887916 ± 0.00000061 | 2.3650% | Stable |
Phase / State
Reason: 1881.8 °C below melting point (1906.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 24 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Cr I | 0 | 4369 | 527 | 4369 |
| Cr II | +1 | 5370 | 92 | 5370 |
| Cr III | +2 | 136 | 0 | 0 |
| Cr IV | +3 | 188 | 102 | 102 |
| Cr V | +4 | 193 | 104 | 193 |
| Cr VI | +5 | 102 | 26 | 102 |
| Cr VII | +6 | 143 | 4 | 143 |
| Cr VIII | +7 | 36 | 9 | 36 |
| Cr IX | +8 | 58 | 18 | 58 |
| Cr X | +9 | 75 | 46 | 75 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Cr I | 0 | 659 |
| Cr II | +1 | 914 |
| Cr III | +2 | 215 |
| Cr IV | +3 | 155 |
| Cr V | +4 | 47 |
| Cr VI | +5 | 63 |
| Cr VII | +6 | 66 |
| Cr VIII | +7 | 34 |
| Cr IX | +8 | 49 |
| Cr X | +9 | 39 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | low | 73 pm |
| +2 | 6 | high | 80 pm |
| +3 | 6 | N/A | 61.5 pm |
| +4 | 4 | N/A | 41 pm |
| +4 | 6 | N/A | 55.00000000000001 pm |
| +5 | 4 | N/A | 34.5 pm |
| +5 | 6 | N/A | 49 pm |
| +5 | 8 | N/A | 56.99999999999999 pm |
| +6 | 4 | N/A | 26 pm |
| +6 | 6 | N/A | 44 pm |
Compounds
Isotopes (3)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 52 Stable | 51.94050623 ± 0.00000063 | 83.7890% ± 0.0180% | Stable | stable | |
| 53 Stable | 52.94064815 ± 0.00000062 | 9.5010% ± 0.0170% | Stable | stable | |
| 54 Stable | 53.93887916 ± 0.00000061 | 2.3650% ± 0.0070% | Stable | stable |
Spectral Lines
Showing 50 of 2531 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 540.978408 nm | 8500 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 534.57965 nm | 5100 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 529.827202 nm | 3540 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 435.17622 nm | 3500 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 534.83147 nm | 3200 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 434.450128 nm | 3100 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 433.944609 nm | 2600 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 526.415341 nm | 2600 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 464.616212 nm | 2400 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 529.669109 nm | 2100 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 391.915844 nm | 2030 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 433.755701 nm | 1900 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 740.01798 nm | 1900 | Cr I | emission | 3d5.(6S).4p z 7P* → 3d5.(6S).5s e 7S | Measured | NIST | |
| 465.215743 nm | 1750 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 435.962444 nm | 1420 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 437.127465 nm | 1400 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 735.58903 nm | 1400 | Cr I | emission | 3d5.(6S).4p z 7P* → 3d5.(6S).5s e 7S | Measured | NIST | |
| 461.612404 nm | 1360 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 390.875593 nm | 1310 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 449.685195 nm | 1300 | Cr I | emission | 3d5.(6S).4s a 5S → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 524.756509 nm | 1250 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 460.074835 nm | 1190 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 433.971051 nm | 1120 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 462.617342 nm | 1100 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 435.104951 nm | 1080 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 438.4975 nm | 1060 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5F* | Measured | NIST | |
| 526.571497 nm | 1050 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 452.64538 nm | 960 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p z 5G* | Measured | NIST | |
| 454.595302 nm | 930 | Cr I | emission | 3d5.(6S).4s a 5S → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 532.832346 nm | 930 | Cr I | emission | 3d5.(6S).4p z 7P* → 3d5.(6S).4d e 7D | Measured | NIST | |
| 388.32867 nm | 910 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 392.86372 nm | 880 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 388.521354 nm | 810 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 746.23079 nm | 800 | Cr I | emission | 3d5.(6S).4p z 7P* → 3d5.(6S).5s e 7S | Measured | NIST | |
| 453.073802 nm | 770 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p z 5G* | Measured | NIST | |
| 388.679508 nm | 740 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 392.102067 nm | 740 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 394.148757 nm | 720 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 697.83983 nm | 640 | Cr I | emission | 3d4.(5D).4s.4p.(3P*) y 7P* → 3d5.(6S).4d e 7D | Measured | NIST | |
| 396.3684 nm | 620 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p y 5H* | Measured | NIST | |
| 453.569676 nm | 600 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p z 5G* | Measured | NIST | |
| 390.290908 nm | 590 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 455.864413 nm | 590 | Cr II | emission | 3d5 b 4F → 3d4.(5D).4p z 4D* | Measured | NIST | |
| 461.335727 nm | 590 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 396.974253 nm | 570 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p y 5H* | Measured | NIST | |
| 458.004789 nm | 560 | Cr I | emission | 3d5.(6S).4s a 5S → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST | |
| 530.074563 nm | 530 | Cr I | emission | 3d4.4s2 a 5D → 3d5.(6S).4p z 5P* | Measured | NIST | |
| 397.665859 nm | 520 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p y 5H* | Measured | NIST | |
| 454.04987 nm | 500 | Cr I | emission | 3d5.(4G).4s a 5G → 3d5.(4G).4p z 5G* | Measured | NIST | |
| 459.139098 nm | 490 | Cr I | emission | 3d4.4s2 a 5D → 3d4.(5D).4s.4p.(3P*) y 5P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2180.15 K |
| Boiling point | 2944.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.5862 |
| 2 | p | 3.9248 |
| 2 | s | 7.0162 |
| 3 | d | 14.2434 |
| 3 | p | 12.534 |
| 3 | s | 11.6322 |
| 4 | s | 18.8668 |
Crystal Radii Detail (10)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | LS | 87 | estimated, |
| 2 | VI | HS | 94 | from r^3 vs V plots, |
| 3 | VI | 75.5 | from r^3 vs V plots, | |
| 4 | IV | 55 | ||
| 4 | VI | 69 | from r^3 vs V plots, | |
| 5 | IV | 48.5 | from r^3 vs V plots, | |
| 5 | VI | 63 | estimated, from r^3 vs V plots, | |
| 5 | VIII | 71 | ||
| 6 | IV | 40 | ||
| 6 | VI | 58 | calculated, |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 41 | p | — |
| 42 | B+ | 100% |
| 42 | B+p | 94.4% |
| 42 | 2p | — |
| 43 | B+ | 100% |
| 43 | B+p | 79.3% |
| 43 | 2p | 11.6% |
| 43 | 3p | 0.1% |
| 43 | B+A | — |
| 44 | B+ | 100% |
X‑ray Scattering Factors (751)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 0.42 | -0.0076 | 0.02756 |
| 0.46 | -0.01 | 0.03022 |
| 0.5 | -0.0123 | 0.03301 |
| 0.54 | -0.0148 | 0.03586 |
| 0.58 | -0.0176 | 0.03897 |
| 0.62 | -0.0202 | 0.04225 |
| 0.66 | -0.0225 | 0.04515 |
| 0.7 | -0.0257 | 0.04771 |
| 0.74 | -0.0292 | 0.05056 |
| 0.78 | -0.0332 | 0.05331 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.02×102 milligrams per kilogram
References (1)
- [5] Chromium https://education.jlab.org/itselemental/ele024.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3×10-4 milligrams per liter
References (1)
- [5] Chromium https://education.jlab.org/itselemental/ele024.html
Sources
Sources of this element.
The principal ore is chromite, which is found in Zimbabwe, Russia, New Zealand, Turkey, Iran, Albania, Finland, Democratic Republic of Madagascar, and the Phillippines. The metal is usually produced by reducing the oxide with aluminum.
References (1)
- [6] Chromium https://periodic.lanl.gov/24.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Chromium.
The element property data was retrieved from publications.