Manganese (Mn)
transition-metalSolid
Standard Atomic Weight
54.938044 uElectron configuration
[Ar] 4s2 3d5Melting point
1245.85 °C (1519 K)Boiling point
2060.85 °C (2334 K)Density
7300 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
1.55Ionization energy (1st)
Discovery year
1774Atomic radius
140 pmDetails
Manganese is a hard, brittle first-row transition metal and an essential alloying element in steelmaking. It occurs in nature mainly as oxides, carbonates, and silicates rather than as the free metal. Its chemistry is notable for accessible oxidation states from +2 to +7, with strong colors and redox behavior. Small biological amounts are essential, especially in enzymes, but concentrated manganese compounds and dusts can be hazardous.
It is gray-white, resembling iron, but is harder and very brittle. The metal is reactive chemically and decomposes slowly in cold water. Manganese is used to form many important alloys. Manganese improves rolling and forging qualities in steel, along with adding strength, stiffness, wear resistance, hardness.
With aluminum and antimony, and especially with small amounts of copper, it forms highly ferromagnetic alloys.
Manganese metal is ferromagnetic only after special treatment. The pure metal exists in four allotropic forms. The alpha form is stable at ordinary temperature; gamma manganese, which changes to alpha at ordinary temperatures, is said to be flexible, soft, easily cut, and capable of being bent.
The name derives from the Latin magnes for "magnet" since pyrolusite (MnO2) has magnetic properties. It was discovered by the Swedish pharmacist and chemist Carl-Wilhelm Scheele in 1774. In the same year, the Swedish chemist Johan Gottlieb Gahn first isolated the metal.
Proposed to be an element by Carl Wilhelm Scheele in 1774, manganese was discovered by Johan Gottlieb Gahn, a Swedish chemist, by heating the mineral pyrolusite (MnO2) in the presence of charcoal later that year. Today, most manganese is still obtained from pyrolusite, although it is usually burned in a furnace with powdered aluminum or is treated with sulfuric acid (H2SO4) to form manganese sulfate (MnSO4), which is then electrolyzed.
From the Latin word magnes, magnet, from magnetic properties of pyrolusite. Recognized by Carl Wilhelm Scheele, Torbern Olof Bergman, and others as an element and isolated by Gahn in 1774 by reduction of the dioxide with carbon.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
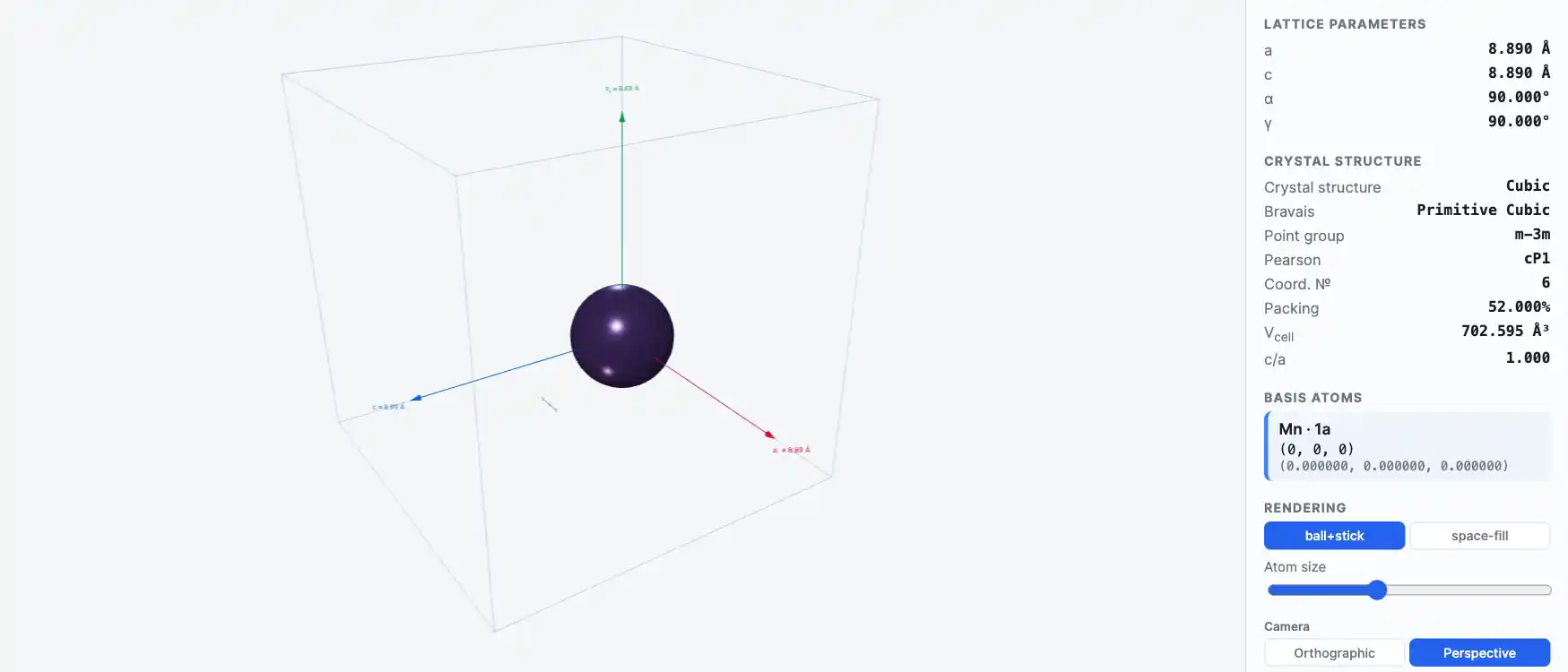
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Mn: 3d⁵ 4s²[Ar] 3d⁵ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁵ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 55 Stable | 54.93804391 ± 0.00000048 | 100.0000% | Stable |
Phase / State
Reason: 1220.8 °C below melting point (1245.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 25 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Mn I | 0 | 631 | 499 | 499 |
| Mn II | +1 | 3975 | 844 | 3781 |
| Mn III | +2 | 86 | 0 | 0 |
| Mn IV | +3 | 50 | 0 | 0 |
| Mn V | +4 | 136 | 112 | 112 |
| Mn VI | +5 | 286 | 97 | 284 |
| Mn VII | +6 | 57 | 26 | 57 |
| Mn VIII | +7 | 49 | 3 | 49 |
| Mn IX | +8 | 43 | 9 | 43 |
| Mn X | +9 | 57 | 18 | 57 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Mn I | 0 | 552 |
| Mn II | +1 | 533 |
| Mn III | +2 | 393 |
| Mn IV | +3 | 104 |
| Mn V | +4 | 85 |
| Mn VI | +5 | 116 |
| Mn VII | +6 | 46 |
| Mn VIII | +7 | 32 |
| Mn IX | +8 | 38 |
| Mn X | +9 | 46 |
Ionic Radii
Showing 10 of 15 Ionic Radii.
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | high | 66 pm |
| +2 | 5 | high | 75 pm |
| +2 | 6 | low | 67 pm |
| +2 | 6 | high | 83 pm |
| +2 | 7 | high | 90 pm |
| +2 | 8 | N/A | 96 pm |
| +3 | 5 | N/A | 57.99999999999999 pm |
| +3 | 6 | low | 57.99999999999999 pm |
| +3 | 6 | high | 64.5 pm |
| +4 | 4 | N/A | 39 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 55 Stable | 54.93804391 ± 0.00000048 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 694 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 403.0753 nm | 27000 | Mn I | emission | 3d5.4s2 a 6S → 3d5.(6S).4s.4p.(3P*) z 6P* | Measured | NIST | |
| 403.3062 nm | 19000 | Mn I | emission | 3d5.4s2 a 6S → 3d5.(6S).4s.4p.(3P*) z 6P* | Measured | NIST | |
| 403.4483 nm | 11000 | Mn I | emission | 3d5.4s2 a 6S → 3d5.(6S).4s.4p.(3P*) z 6P* | Measured | NIST | |
| 404.1355 nm | 5600 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 380.6711 nm | 3200 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 382.3507 nm | 2100 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 405.5544 nm | 1900 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 401.81 nm | 1500 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 383.4362 nm | 1300 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 404.8743 nm | 1100 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 405.893 nm | 1100 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 408.2939 nm | 1100 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 408.3628 nm | 1100 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 475.4042 nm | 1000 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 8P* → 3d5.4s.(7S).5s e 8S | Measured | NIST | |
| 482.3524 nm | 1000 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 8P* → 3d5.4s.(7S).5s e 8S | Measured | NIST | |
| 478.3427 nm | 940 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 8P* → 3d5.4s.(7S).5s e 8S | Measured | NIST | |
| 445.1586 nm | 800 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 476.2367 nm | 750 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4F* | Measured | NIST | |
| 406.173 nm | 730 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 6P* → 3d5.4s.(5S).5s f 6S | Measured | NIST | |
| 406.3528 nm | 730 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 407.9412 nm | 730 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 380.9592 nm | 700 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 384.1071 nm | 670 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 446.2031 nm | 510 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 6P* → 3d5.4s.(7S).4d e 6D | Measured | NIST | |
| 432.6643 nm | 500 | Mn II | emission | 3d5.(4F).4s a 5F → 3d5.(4G).4p z 5F* | Measured | NIST | |
| 434.3983 nm | 500 | Mn II | emission | 3d5.(4F).4s a 5F → 3d5.(4G).4p z 5F* | Measured | NIST | |
| 476.6418 nm | 500 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4F* | Measured | NIST | |
| 383.3861 nm | 480 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 382.3887 nm | 390 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 423.5295 nm | 370 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p y 4P* | Measured | NIST | |
| 383.9819 nm | 350 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 384.3984 nm | 350 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 441.489 nm | 350 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 476.5846 nm | 300 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4F* | Measured | NIST | |
| 407.0278 nm | 290 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6D* | Measured | NIST | |
| 425.7669 nm | 290 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p y 4P* | Measured | NIST | |
| 426.5923 nm | 290 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p y 4P* | Measured | NIST | |
| 446.4682 nm | 290 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 602.182 nm | 290 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 6P* → 3d5.4s.(7S).5s e 6S | Measured | NIST | |
| 428.1097 nm | 270 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p y 4P* | Measured | NIST | |
| 445.8254 nm | 270 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 6P* → 3d5.4s.(7S).4d e 6D | Measured | NIST | |
| 449.8902 nm | 240 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 450.2213 nm | 240 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 443.6357 nm | 210 | Mn I | emission | 3d6.(5D).4s a 4D → 3d6.(5D).4p z 4D* | Measured | NIST | |
| 445.7549 nm | 210 | Mn I | emission | 3d5.(6S).4s.4p.(3P*) z 6P* → 3d5.4s.(7S).4d e 6D | Measured | NIST | |
| 382.9718 nm | 200 | Mn I | emission | 3d6.(5D).4s a 6D → 3d6.(5D).4p z 6F* | Measured | NIST | |
| 384.4166 nm | 200 | Mn II | emission | 3d5.(2F).4s b 3F → 3d5.(4G).4p z 3G* | Measured | NIST | |
| 420.63677 nm | 200 | Mn II | emission | 3d5.(4F).4s a 5F → 3d5.(4P).4p z 5D* | Measured | NIST | |
| 429.22329 nm | 200 | Mn II | emission | 3d5.(2D).4s c 3D → 3d5.(4G).4p z 5F* | Measured | NIST | |
| 434.83962 nm | 200 | Mn II | emission | 3d5.(4F).4s a 5F → 3d5.(4G).4p z 5F* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1519.15 K |
| Boiling point | 2334.15 K |
| Critical point (temperature) | 4325.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6043 |
| 2 | p | 3.916 |
| 2 | s | 7.2062 |
| 3 | d | 14.4718 |
| 3 | p | 12.8908 |
| 3 | s | 11.9821 |
| 4 | s | 19.7168 |
Crystal Radii Detail (15)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | HS | 80 | |
| 2 | V | HS | 89 | calculated, |
| 2 | VI | LS | 81 | estimated, |
| 2 | VI | HS | 97 | from r^3 vs V plots, |
| 2 | VII | HS | 104 | calculated, |
| 2 | VIII | 110 | from r^3 vs V plots, | |
| 3 | V | 72 | ||
| 3 | VI | LS | 72 | from r^3 vs V plots, |
| 3 | VI | HS | 78.5 | from r^3 vs V plots, |
| 4 | IV | 53 | from r^3 vs V plots, |
Isotope Decay Modes (57)
| Isotope | Mode | Intensity |
|---|---|---|
| 43 | p | — |
| 44 | p | — |
| 45 | p | — |
| 46 | B+ | 100% |
| 46 | B+p | 57% |
| 46 | 2p | 18% |
| 46 | B+A | — |
| 47 | B+ | 100% |
| 47 | B+p | 1.7% |
| 48 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.8899 |
| 10.1617 | — | 1.92644 |
| 10.3261 | — | 1.96368 |
| 10.4931 | — | 2.00165 |
| 10.6628 | — | 2.04035 |
| 10.8353 | — | 2.0798 |
| 11.0106 | — | 2.12001 |
| 11.1886 | — | 2.161 |
| 11.3696 | — | 2.20278 |
| 11.5535 | — | 2.24537 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
9.50×102 milligrams per kilogram
References (1)
- [5] Manganese https://education.jlab.org/itselemental/ele025.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-4 milligrams per liter
References (1)
- [5] Manganese https://education.jlab.org/itselemental/ele025.html
Sources
Sources of this element.
Manganese minerals are widely distributed, with oxides, silicates, and carbonates being the most common. Large quantities of manganese nodules are found on the ocean floor and may become a source of manganese. These nodules contain about 24% manganese, together with many other elements in lesser abundance.
Most manganese today is obtained from ores found in Russia, Brazil, Australia, South Africa, Gabon, and India. Pyrolusite and rhodochrosite are among the most common manganese minerals. The metal is obtained by reduction of the oxide with sodium, magnesium, aluminum, or by electrolysis.
References (1)
- [6] Manganese https://periodic.lanl.gov/25.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Manganese.
The element property data was retrieved from publications.