Iron (Fe)
transition-metalSolid
Standard Atomic Weight
55.845 uElectron configuration
[Ar] 4s2 3d6Melting point
1537.85 °C (1811 K)Boiling point
2860.85 °C (3134 K)Density
7874 kg/m³Oxidation states
−4, −2, −1, 0, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
1.83Ionization energy (1st)
Discovery year
N/AAtomic radius
140 pmDetails
Iron is a first-row transition metal and one of the central elements of technology and biology. It forms strong, workable alloys, especially with carbon, and has accessible Fe²⁺ and Fe³⁺ redox chemistry. In Earth materials it is a major constituent of the core, mantle minerals, and many ores. Its magnetic behavior and ability to bind small molecules in metalloproteins give it importance beyond structural metal use.
The pure metal is very reactive chemically and rapidly corrodes, especially in moist air or at elevated temperatures. It has four allotropic forms or ferrites, known as alpha, beta, gamma, and omega, with transition points at 700, 928, and 1530C. The alpha form is magnetic, but when transformed into the beta form, the magnetism disappears although the lattice remains unchanged. The relations of these forms are peculiar. Pig iron is an alloy containing about 3 percent carbon with varying amounts of sulfur, silicon, manganese, and phosphorus.
Iron is hard, brittle, fairly fusible, and is used to produce other alloys, including steel. Wrought iron contains only a few tenths of a percent of carbon, is tough, malleable, less fusible, and usually has a "fibrous" structure.
Carbon steel is an alloy of iron with small amounts of Mn, S, P, and Si. Alloy steels are carbon steels with other additives such as nickel, chromium, vanadium, etc. Iron is a cheap, abundant, useful, and important metal.
The name derives from the Anglo-Saxon iron of unknown origin. The element has been known from prehistoric times. The symbol Fe is derived from the Latin ferrum for "firmness". It is of interest to note that 56Fe requires more energy to be formed than any other nuclide. It is, therefore, the ultimate endproduct of stellar nuclear fusion.
Archaeological evidence suggests that people have been using iron for at least 5000 years. Iron is the cheapest and one of the most abundant of all metals, comprising nearly 5.6% of the earth's crust and nearly all of the earth's core. Iron is primarily obtained from the minerals hematite (Fe2O3) and magnetite (Fe3O4). The minerals taconite, limonite (FeO(OH)·nH2O) and siderite (FeCO3) are other important sources.
Latin ferrum. Iron was used prehistorically:
▸ Iron is mentioned numerous times in the Old Testament of the Bible.
▸ A remarkable iron pillar, dating to about A.D. 400, remains standing today in Delhi, India. This solid shaft of wrought iron is about 7 1/4 m high by 40 cm in diameter. Corrosion to the pillar has been minimal although it has been exposed to the weather since its creation.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
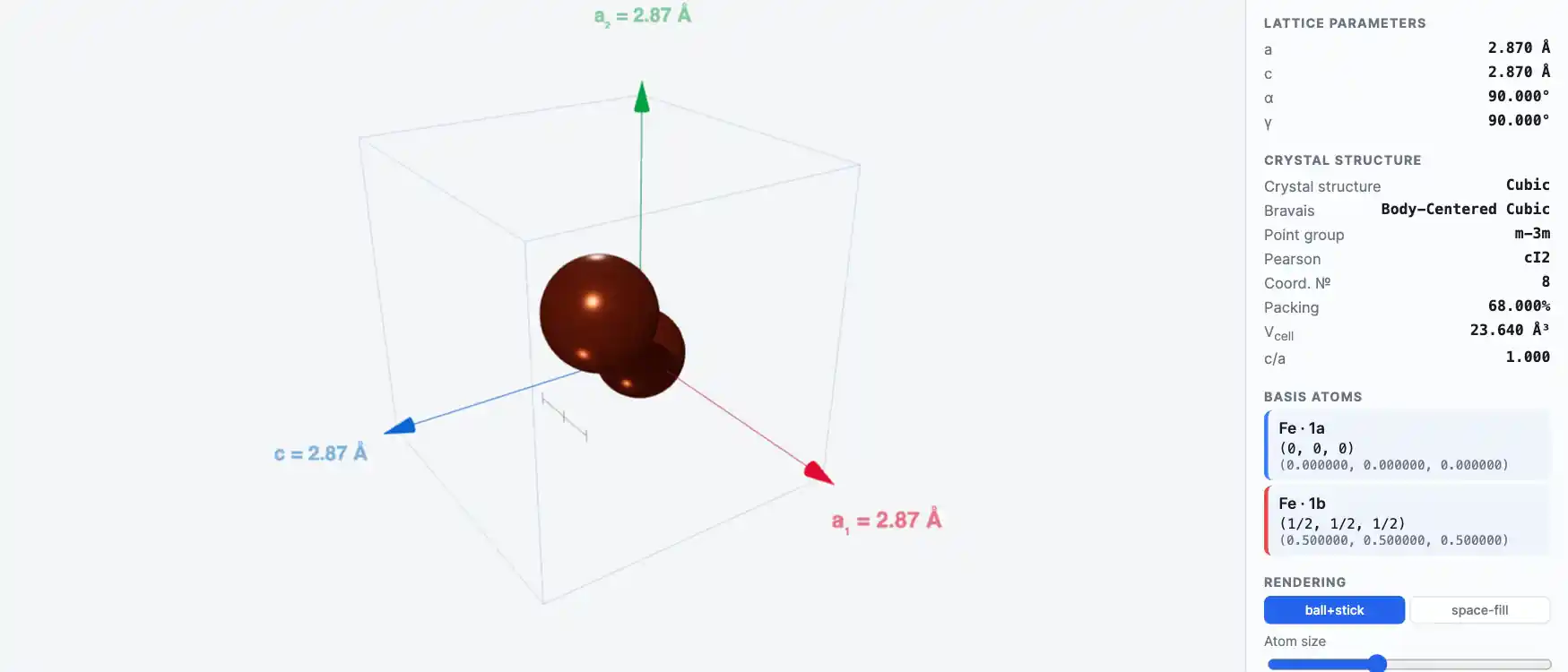
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Fe: 3d⁶ 4s²[Ar] 3d⁶ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁶ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 54 Stable | 53.93960899 ± 0.00000053 | 5.8450% | Stable |
| 56 Stable | 55.93493633 ± 0.00000049 | 91.7540% | Stable |
| 57 Stable | 56.93539284 ± 0.00000049 | 2.1190% | Stable |
| 58 Stable | 57.93327443 ± 0.00000053 | 0.2820% | Stable |
Phase / State
Reason: 1512.8 °C below melting point (1537.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 26 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Fe I | 0 | 10031 | 2542 | 9906 |
| Fe II | +1 | 14471 | 7293 | 14471 |
| Fe III | +2 | 4702 | 2361 | 4687 |
| Fe IV | +3 | 258 | 102 | 102 |
| Fe V | +4 | 2310 | 2018 | 2310 |
| Fe VI | +5 | 159 | 159 | 159 |
| Fe VII | +6 | 651 | 651 | 651 |
| Fe VIII | +7 | 92 | 63 | 92 |
| Fe IX | +8 | 56 | 14 | 56 |
| Fe X | +9 | 98 | 64 | 98 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Fe I | 0 | 847 |
| Fe II | +1 | 1028 |
| Fe III | +2 | 596 |
| Fe IV | +3 | 277 |
| Fe V | +4 | 332 |
| Fe VI | +5 | 94 |
| Fe VII | +6 | 210 |
| Fe VIII | +7 | 42 |
| Fe IX | +8 | 35 |
| Fe X | +9 | 65 |
Ionic Radii
Showing 10 of 12 Ionic Radii.
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | high | 63 pm |
| +2 | 4 | high | 64 pm |
| +2 | 6 | low | 61 pm |
| +2 | 6 | high | 78 pm |
| +2 | 8 | high | 92 pm |
| +3 | 4 | high | 49 pm |
| +3 | 5 | N/A | 57.99999999999999 pm |
| +3 | 6 | low | 55.00000000000001 pm |
| +3 | 6 | high | 64.5 pm |
| +3 | 8 | high | 78 pm |
Compounds
Isotopes (4)
Common iron is a mixture of four isotopes. Ten other isotopes are known to exist.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 54 Stable | 53.93960899 ± 0.00000053 | 5.8450% ± 0.0350% | Stable | stable | |
| 56 Stable | 55.93493633 ± 0.00000049 | 91.7540% ± 0.0360% | Stable | stable | |
| 57 Stable | 56.93539284 ± 0.00000049 | 2.1190% ± 0.0100% | Stable | stable | |
| 58 Stable | 57.93327443 ± 0.00000053 | 0.2820% ± 0.0040% | Stable | stable |
Spectral Lines
Showing 50 of 5034 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 387.857282 nm | 1290000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 393.02964 nm | 1150000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 385.637115 nm | 1100000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 389.970707 nm | 1070000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 526.95366 nm | 1020000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 382.444329 nm | 1000000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 392.291129 nm | 1000000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 404.581193 nm | 1000000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 649.49801 nm | 870000 | Fe I | emission | 3d6.4s2 a 3H → 3d7.(4F).4p z 5G* | Measured | NIST | |
| 406.359365 nm | 830000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 432.57616 nm | 830000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 3G* | Measured | NIST | |
| 440.47498 nm | 810000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 5G* | Measured | NIST | |
| 381.58397 nm | 760000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3D* | Measured | NIST | |
| 382.588058 nm | 760000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 516.74879 nm | 760000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d6.(5D).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 389.565597 nm | 740000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 532.80381 nm | 740000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 407.173752 nm | 710000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 392.025748 nm | 650000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 427.17599 nm | 630000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 3G* | Measured | NIST | |
| 430.79017 nm | 630000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 3G* | Measured | NIST | |
| 382.78222 nm | 590000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3D* | Measured | NIST | |
| 383.422219 nm | 590000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 388.628183 nm | 550000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 640 nm | 490000 | Fe I | emission | 3d6.(5D).4s.4p.(3P*) z 5P* → 3d6.(5D).4s (6D).5s e 5D | Measured | NIST | |
| 384.10475 nm | 457000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3D* | Measured | NIST | |
| 522.71889 nm | 437000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d6.(5D).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 396.925691 nm | 427000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 381.296424 nm | 398000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d6.(5D).4s.4p.(3P*) z 3P* | Measured | NIST | |
| 537.14891 nm | 389000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 384.04372 nm | 380000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 414.386752 nm | 363000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 390.294512 nm | 302000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3D* | Measured | NIST | |
| 639.36001 nm | 302000 | Fe I | emission | 3d6.4s2 a 3H → 3d7.(4F).4p z 5G* | Measured | NIST | |
| 495.75961 nm | 295000 | Fe I | emission | 3d6.(5D).4s.4p.(3P*) z 7F* → 3d6.(5D).4s (6D).5s e 7D | Measured | NIST | |
| 441.51221 nm | 288000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 5G* | Measured | NIST | |
| 527.03561 nm | 288000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d6.(5D).4s.4p.(3P*) z 3D* | Measured | NIST | |
| 387.801779 nm | 275000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 642.13496 nm | 257000 | Fe I | emission | 3d6.4s2 a 3P2 → 3d6.(5D).4s.4p.(3P*) z 3P* | Measured | NIST | |
| 388.704779 nm | 251000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 667.79848 nm | 240000 | Fe I | emission | 3d7.(2G).4s a 3G → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 387.250102 nm | 234000 | Fe I | emission | 3d7.(4F).4s a 5F → 3d7.(4F).4p y 5D* | Measured | NIST | |
| 390.647918 nm | 234000 | Fe I | emission | 3d6.4s2 a 5D → 3d6.(5D).4s.4p.(3P*) z 5D* | Measured | NIST | |
| 413.205785 nm | 224000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 426.04736 nm | 224000 | Fe I | emission | 3d6.(5D).4s.4p.(3P*) z 7D* → 3d6.(5D).4s (6D).5s e 7D | Measured | NIST | |
| 654.62373 nm | 224000 | Fe I | emission | 3d7.(2G).4s a 3G → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 641.16477 nm | 219000 | Fe I | emission | 3d6.(5D).4s.4p.(3P*) z 5P* → 3d6.(5D).4s (6D).5s e 5D | Measured | NIST | |
| 425.07864 nm | 214000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p z 3G* | Measured | NIST | |
| 400.524148 nm | 209000 | Fe I | emission | 3d7.(4F).4s a 3F → 3d7.(4F).4p y 3F* | Measured | NIST | |
| 643.08447 nm | 209000 | Fe I | emission | 3d7.(4P).4s a 5P → 3d7.(4F).4p y 5D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1811.15 K |
| Boiling point | 3134.15 K |
| Critical point (temperature) | 9340.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.619 |
| 2 | p | 3.9112 |
| 2 | s | 7.401 |
| 3 | d | 14.8202 |
| 3 | p | 13.2221 |
| 3 | s | 12.3239 |
| 4 | s | 20.566 |
Crystal Radii Detail (12)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | HS | 77 | |
| 2 | IVSQ | HS | 78 | |
| 2 | VI | LS | 75 | estimated, |
| 2 | VI | HS | 92 | from r^3 vs V plots, |
| 2 | VIII | HS | 106 | calculated, |
| 3 | IV | HS | 63 | |
| 3 | V | 72 | ||
| 3 | VI | LS | 69 | from r^3 vs V plots, |
| 3 | VI | HS | 78.5 | from r^3 vs V plots, |
| 3 | VIII | HS | 92 |
Isotope Decay Modes (55)
| Isotope | Mode | Intensity |
|---|---|---|
| 45 | 2p | 70% |
| 45 | B+ | 30% |
| 45 | B+p | 18.9% |
| 45 | 2p | 7.8% |
| 46 | B+ | 100% |
| 46 | B+p | 78.7% |
| 46 | 2p | — |
| 47 | B+ | 100% |
| 47 | B+p | 88.4% |
| 48 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.37852 |
| 10.1617 | — | 1.42961 |
| 10.3261 | — | 1.48259 |
| 10.4931 | — | 1.53754 |
| 10.6628 | — | 1.59453 |
| 10.8353 | — | 1.65362 |
| 11.0106 | — | 1.71491 |
| 11.1886 | — | 1.77847 |
| 11.3696 | — | 1.84438 |
| 11.5535 | — | 1.91274 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5.63×104 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-3 milligrams per liter
References (1)
Sources
Sources of this element.
Iron is a relatively abundant element in the universe. It is found in the sun and many types of stars in considerable quantity. Its nuclei are very stable. Iron is a principal component of a meteorite class known as siderites and is a minor constituent of the other two meteorite classes. The core of the earth 2150 miles in radius is thought to be largely composed of iron with about 10 percent occluded hydrogen. The metal is the fourth most abundant element, by weight that makes up the crust of the earth.
The most common ore is hematite, which is frequently seen as black sands along beaches and banks of streams.
References (1)
- [6] Iron https://periodic.lanl.gov/26.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Iron.
The element property data was retrieved from publications.