Nickel (Ni)
transition-metalSolid
Standard Atomic Weight
58.6934 uElectron configuration
[Ar] 4s2 3d8Melting point
1454.85 °C (1728 K)Boiling point
2912.85 °C (3186 K)Density
8912 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
1.91Ionization energy (1st)
Discovery year
1751Atomic radius
135 pmDetails
Nickel is a silvery transition metal of group 10, valued for corrosion resistance, strength at high temperature, and its ability to form useful alloys. It is ferromagnetic near room temperature and commonly occurs in the +2 oxidation state, although several other states are known in coordination chemistry. Natural nickel is mostly found in sulfide and laterite ores, and it is a key metal for stainless steels, superalloys, plating, catalysts, and rechargeable batteries.
Nickel is silvery white and takes on a high polish. It is hard, malleable, ductile, somewhat ferromagnetic, and a fair conductor of heat and electricity. It belongs to the iron-cobalt group of metals and is chiefly valuable for the alloys it forms.
The name derives from the German Nickel for "deceptive little spirit" because miners called mineral niccolite (NiAs) by the name Kupfernickel (false copper) because it resembled copper ores in appearance, but no copper was found in the ore. It was discovered by the Swedish metallurgist Axel-Frederik Cronstedt in 1751.
Nickel was discovered by the Swedish chemist Axel Fredrik Cronstedt in the mineral niccolite (NiAs) in 1751. Today, most nickel is obtained from the mineral pentlandite (NiS·2FeS). Most of the world's supply of nickel is mined in the Sudbury region of Ontario, Canada. It is believed that this large deposit of nickel ore is a result of an ancient meteor impact.
From the German word Nickel (Satan), and from kupfernickel, Old Nick's copper. Cronstedt discovered nickel in 1751 in kupfernickel (niccolite).
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
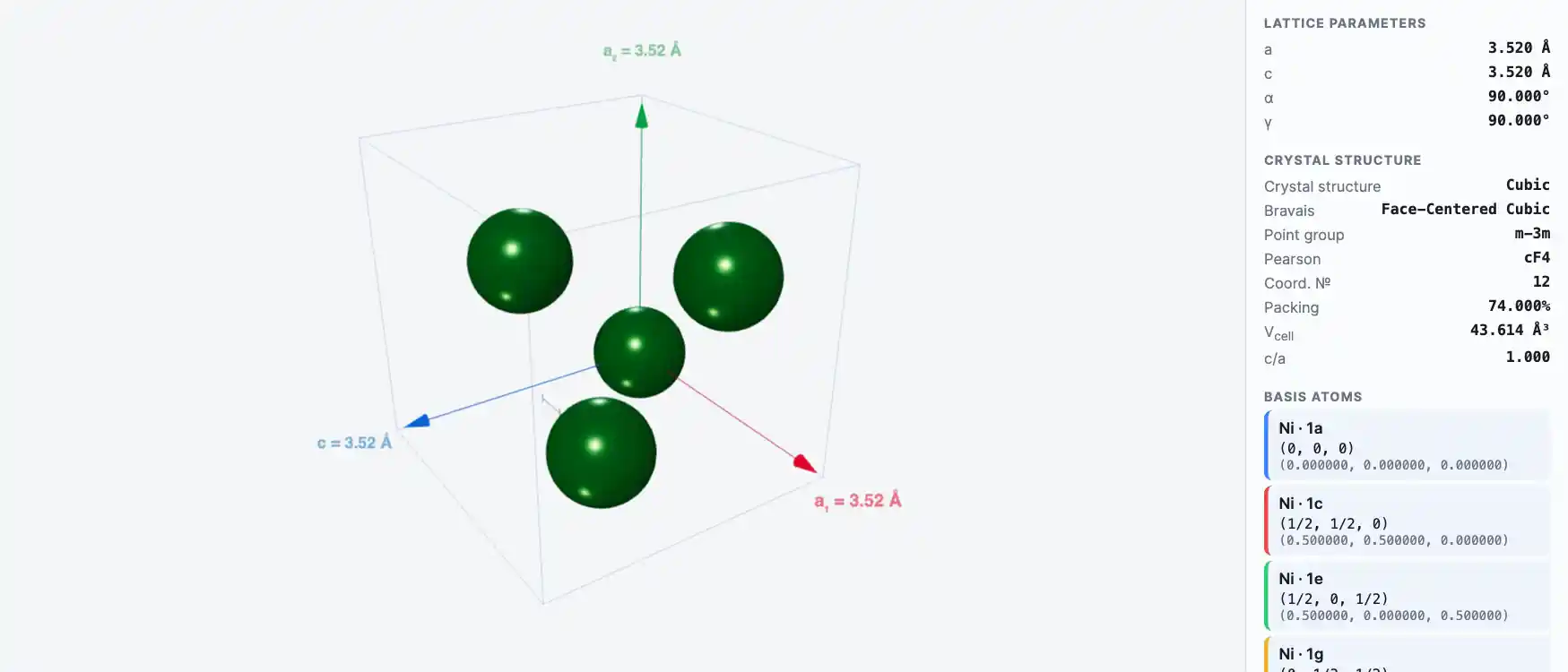
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ni: 3d⁸ 4s²[Ar] 3d⁸ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁸ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 60 Stable | 59.93078588 ± 0.00000052 | 26.2230% | Stable |
| 61 Stable | 60.93105557 ± 0.00000052 | 1.1399% | Stable |
| 62 Stable | 61.92834537 ± 0.00000055 | 3.6346% | Stable |
| 64 Stable | 63.92796682 ± 0.00000058 | 0.9255% | Stable |
Phase / State
Reason: 1429.8 °C below melting point (1454.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 28 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ni I | 0 | 576 | 522 | 522 |
| Ni II | +1 | 249 | 208 | 208 |
| Ni III | +2 | 128 | 50 | 50 |
| Ni IV | +3 | 216 | 169 | 169 |
| Ni V | +4 | 1582 | 1566 | 1582 |
| Ni VII | +6 | 24 | 24 | 24 |
| Ni IX | +8 | 38 | 20 | 38 |
| Ni X | +9 | 41 | 0 | 41 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ni I | 0 | 288 |
| Ni II | +1 | 719 |
| Ni III | +2 | 345 |
| Ni IV | +3 | 236 |
| Ni V | +4 | 324 |
| Ni VI | +5 | 273 |
| Ni VII | +6 | 45 |
| Ni VIII | +7 | 44 |
| Ni IX | +8 | 31 |
| Ni X | +9 | 34 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 55.00000000000001 pm |
| +2 | 4 | N/A | 49 pm |
| +2 | 5 | N/A | 63 pm |
| +2 | 6 | N/A | 69 pm |
| +3 | 6 | low | 56.00000000000001 pm |
| +3 | 6 | high | 60 pm |
| +4 | 6 | low | 48 pm |
Compounds
Isotopes (4)
The sulfate and the oxides are important compounds. Natural nickel is a mixture of five stable isotopes; nine other unstable isotopes are known.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 60 Stable | 59.93078588 ± 0.00000052 | 26.2230% ± 0.0150% | Stable | stable | |
| 61 Stable | 60.93105557 ± 0.00000052 | 1.1399% ± 0.0013% | Stable | stable | |
| 62 Stable | 61.92834537 ± 0.00000055 | 3.6346% ± 0.0040% | Stable | stable | |
| 64 Stable | 63.92796682 ± 0.00000058 | 0.9255% ± 0.0019% | Stable | stable |
Spectral Lines
Showing 50 of 433 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 385.82968 nm | 1200 | Ni I | emission | 3d9.(2D).4s 1D → 3d9.(2D).4p 3F* | Measured | NIST | |
| 380.71402 nm | 700 | Ni I | emission | 3d9.(2D).4s 1D → 3d9.(2D).4p * | Measured | NIST | |
| 547.6904 nm | 180 | Ni I | emission | 3d10 1S → 3d9.(2D).4p 1P* | Measured | NIST | |
| 383.16908 nm | 110 | Ni I | emission | 3d9.(2D).4s 1D → 3d9.(2D).4p 3P* | Measured | NIST | |
| 397.35547 nm | 110 | Ni I | emission | 3d9.(2D).4s 1D → 3d9.(2D).4p 3P* | Measured | NIST | |
| 440.1541 nm | 110 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5D* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 471.4417 nm | 110 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 503.5362 nm | 100 | Ni I | emission | 3d9.(2D).4p 3F* → 3d9.(2D<5/2>).4d 2[9/2] | Measured | NIST | |
| 508.0533 nm | 100 | Ni I | emission | 3d9.(2D).4p 3F* → 3d9.(2D<5/2>).4d 2[9/2] | Measured | NIST | |
| 464.8652 nm | 75 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 460.4987 nm | 65 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 508.111 nm | 65 | Ni I | emission | 3d9.(2D).4p 1F* → 3d9.(2D<3/2>).4d 2[7/2] | Measured | NIST | |
| 447.0477 nm | 55 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5D* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 501.7576 nm | 50 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5F* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 478.6535 nm | 45 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 485.5411 nm | 45 | Ni I | emission | 3d9.(2D).4p 3P* → 3d9.(2D<5/2>).4d 2[3/2] | Measured | NIST | |
| 498.0173 nm | 45 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5F* → 3d9.(2D<5/2>).4d 2[9/2] | Measured | NIST | |
| 490.4412 nm | 40 | Ni I | emission | 3d9.(2D).4p 3P* → 3d9.(2D<5/2>).4d 2[1/2] | Measured | NIST | |
| 475.6515 nm | 30 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 712.2197 nm | 26 | Ni I | emission | 3d9.(2D).4p 3P* → 3d9.(2D<5/2>).5s 2[5/2] | Measured | NIST | |
| 468.6213 nm | 23 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 513.7074 nm | 23 | Ni I | emission | 3d8.(1D).4s2 1D → 3d9.(2D).4p 1P* | Measured | NIST | |
| 570.9545 nm | 23 | Ni I | emission | 3d8.(1D).4s2 1D → 3d9.(2D).4p 1F* | Measured | NIST | |
| 742.2275 nm | 23 | Ni I | emission | 3d9.(2D).4p 3F* → 3d9.(2D<5/2>).5s 2[5/2] | Measured | NIST | |
| 471.5762 nm | 22 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 480.6993 nm | 22 | Ni I | emission | 3d9.(2D).4p * → 3d8.4s.(4F).5s 3F | Measured | NIST | |
| 491.8364 nm | 22 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 3G* → 3d8.4s.(4F).5s 3F | Measured | NIST | |
| 676.7772 nm | 22 | Ni I | emission | 3d10 1S → 3d9.(2D).4p 3P* | Measured | NIST | |
| 511.5392 nm | 21 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 3G* → 3d8.4s.(4F).5s 3F | Measured | NIST | |
| 483.1176 nm | 19 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5F* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 446.2455 nm | 18 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5D* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 460.0359 nm | 18 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 460.6221 nm | 18 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5G* → 3d9.(2D<3/2>).4d 2[3/2] | Measured | NIST | |
| 501.2443 nm | 18 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 5F* → 3d8.4s.(4F).5s 5F | Measured | NIST | |
| 493.5831 nm | 16 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 3G* → 3d8.4s.(4F).5s 3F | Measured | NIST | |
| 504.8847 nm | 16 | Ni I | emission | 3d9.(2D).4p 1F* → 3d9.(2D<3/2>).4d 2[5/2] | Measured | NIST | |
| 575.4656 nm | 16 | Ni I | emission | 3d8.(3P).4s2 3P → 3d9.(2D).4p 1P* | Measured | NIST | |
| 664.363 nm | 16 | Ni I | emission | 3d8.(1D).4s2 1D → 3d9.(2D).4p 3P* | Measured | NIST | |
| 739.3676 nm | 16 | Ni I | emission | 3d8.(3F).4s2 3F → 3d8.(1D).4s2 1D | Measured | NIST | |
| 517.656 nm | 13 | Ni I | emission | 3d9.(2D).4p 1D* → 3d9.(2D<3/2>).4d 2[3/2] | Measured | NIST | |
| 559.2262 nm | 13 | Ni I | emission | 3d8.(3P).4s2 3P → 3d8.(3F).4s.4p.(3P*) 3D* | Measured | NIST | |
| 625.6355 nm | 13 | Ni I | emission | 3d8.(1D).4s2 1D → 3d9.(2D).4p 3P* | Measured | NIST | |
| 568.2199 nm | 12 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 3F* → 3d9.(2D<3/2>).4d 2[7/2] | Measured | NIST | |
| 571.1888 nm | 10 | Ni I | emission | 3d8.(3P).4s2 3P → 3d8.(3F).4s.4p.(3P*) 3F* | Measured | NIST | |
| 589.2872 nm | 10 | Ni I | emission | 3d8.(3P).4s2 3P → 3d9.(2D).4p 1P* | Measured | NIST | |
| 610.8116 nm | 10 | Ni I | emission | 3d8.(1D).4s2 1D → 3d9.(2D).4p 3D* | Measured | NIST | |
| 617.6811 nm | 10 | Ni I | emission | 3d8.(3F).4s.4p.(3P*) 3F* → 3d9.(2D<5/2>).4d 2[9/2] | Measured | NIST | |
| 631.4659 nm | 10 | Ni I | emission | 3d8.(3P).4s2 3P → 3d9.(2D).4p 1D* | Measured | NIST | |
| 691.4559 nm | 10 | Ni I | emission | 3d8.(3P).4s2 3P → 3d9.(2D).4p 3P* | Measured | NIST | |
| 558.7858 nm | 9 | Ni I | emission | 3d8.(3P).4s2 3P → 3d8.(3F).4s.4p.(3P*) 3D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1728.15 K |
| Boiling point | 3186.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6474 |
| 2 | p | 3.9048 |
| 2 | s | 7.7874 |
| 3 | d | 15.4705 |
| 3 | p | 13.915 |
| 3 | s | 13.039 |
| 4 | s | 22.2892 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | 69 | ||
| 2 | IVSQ | 63 | ||
| 2 | V | 77 | estimated, | |
| 2 | VI | 83 | from r^3 vs V plots, | |
| 3 | VI | LS | 70 | from r^3 vs V plots, |
| 3 | VI | HS | 74 | estimated, |
| 4 | VI | LS | 62 | from r^3 vs V plots, |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 48 | 2p | 70% |
| 48 | B+ | 30% |
| 48 | B+p | — |
| 49 | B+ | 100% |
| 49 | B+p | 83.4% |
| 50 | B+ | 100% |
| 50 | B+p | 73% |
| 50 | 2p | 14% |
| 51 | B+ | 100% |
| 51 | B+p | 87.2% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.37727 |
| 10.1617 | — | 1.38064 |
| 10.3261 | — | 1.38401 |
| 10.4931 | — | 1.3874 |
| 10.6628 | — | 1.39079 |
| 10.8353 | — | 1.39419 |
| 11.0106 | — | 1.39982 |
| 11.1886 | — | 1.44104 |
| 11.3696 | — | 1.48347 |
| 11.5535 | — | 1.52716 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8.4×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
5.6×10-4 milligrams per liter
References (1)
Sources
Sources of this element.
Nickel is found as a constituent in most meteorites and often serves as one of the criteria for distinguishing a meteorite from other minerals. Iron meteorites, or siderites, may contain iron alloyed with from 5 percent to nearly 20 percent nickel. Nickel is obtained commercially from pentlandite and pyrrhotite of the Sudbury region of Ontario, a district that produces about 30 percent of the world's supply of nickel.
Other deposits are found in New Caledonia, Australia, Cuba, Indonesia, and elsewhere.
References (1)
- [6] Nickel https://periodic.lanl.gov/28.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Nickel.
The element property data was retrieved from publications.