Copper (Cu)
transition-metalSolid
Standard Atomic Weight
63.546 uElectron configuration
[Ar] 4s1 3d10Melting point
1084.62 °C (1357.77 K)Boiling point
2561.85 °C (2835 K)Density
8933 kg/m³Oxidation states
−2, 0, +1, +2, +3, +4Electronegativity (Pauling)
1.9Ionization energy (1st)
Discovery year
N/AAtomic radius
135 pmDetails
Copper is a transition metal with high electrical and thermal conductivity, good ductility, and a chemistry dominated by the +1 and +2 oxidation states. It is one of the few metals found naturally in native form and has been worked since prehistory. Modern importance rests on electrical conductors, plumbing, heat exchangers, alloys, and catalytic or biological redox chemistry. Its surfaces oxidize slowly in air, often developing protective films rather than deep rusting.
Copper is reddish and takes on a bright metallic luster. It is malleable, ductile, and a good conductor of heat and electricity (second only to silver in electrical conductivity).
The name derives from the Latin cuprum for Cyprus, the island where the Romans first obtained copper. The symbol Cu also comes from the Latin cuprum. The element has been known since prehistoric times.
Archaeological evidence suggests that people have been using copper for at least 11,000 years. Relatively easy to mine and refine, people discovered methods for extracting copper from its ores at least 7,000 years ago. The Roman Empire obtained most of its copper from the island of Cyprus, which is where copper's name originated. Today, copper is primarily obtained from the ores cuprite (CuO2), tenorite (CuO), malachite (CuO3·Cu(OH)2), chalcocite (Cu2S), covellite (CuS) and bornite (Cu6FeS4). Large deposits of copper ore are located in the United States, Chile, Zambia, Zaire, Peru and Canada.
From the Latin word cuprum, from the island of Cyprus. It is believed that copper has been mined for 5,000 years.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
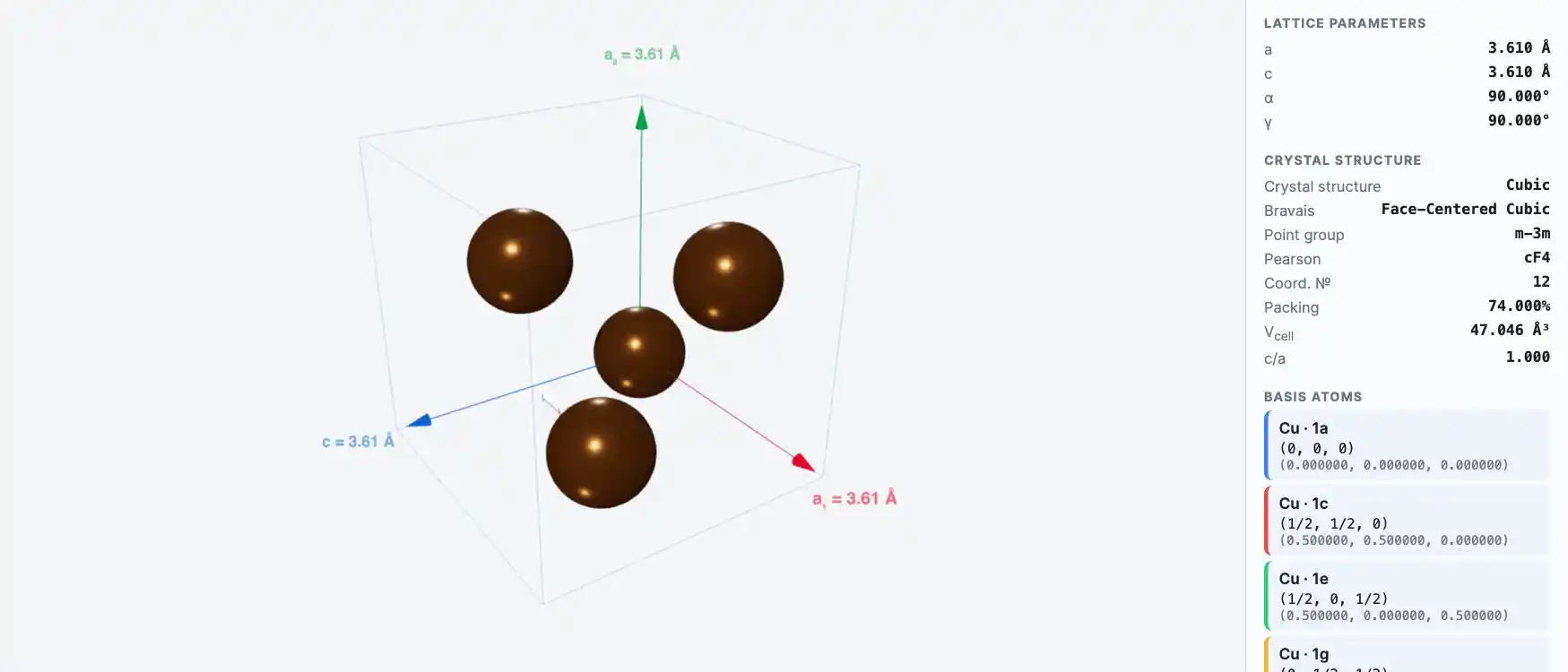
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Cu: 3d¹⁰ 4s¹[Ar] 3d¹⁰ 4s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 63 Stable | 62.92959772 ± 0.00000056 | 69.1500% | Stable |
| 65 Stable | 64.9277897 ± 0.00000071 | 30.8500% | Stable |
Phase / State
Reason: 1059.6 °C below melting point (1084.62 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 29 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Cu I | 0 | 1003 | 37 | 1003 |
| Cu II | +1 | 2557 | 554 | 2557 |
| Cu III | +2 | 100 | 0 | 0 |
| Cu IV | +3 | 60 | 0 | 0 |
| Cu V | +4 | 50 | 0 | 0 |
| Cu X | +9 | 28 | 0 | 28 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Cu I | 0 | 365 |
| Cu II | +1 | 468 |
| Cu III | +2 | 390 |
| Cu IV | +3 | 298 |
| Cu V | +4 | 249 |
| Cu VI | +5 | 255 |
| Cu VII | +6 | 5 |
| Cu VIII | +7 | 2 |
| Cu IX | +8 | 2 |
| Cu X | +9 | 31 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 2 | N/A | 46 pm |
| +1 | 4 | N/A | 60 pm |
| +1 | 6 | N/A | 77 pm |
| +2 | 4 | N/A | 56.99999999999999 pm |
| +2 | 4 | N/A | 56.99999999999999 pm |
| +2 | 5 | N/A | 65 pm |
| +2 | 6 | N/A | 73 pm |
| +3 | 6 | low | 54 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 63 Stable | 62.92959772 ± 0.00000056 | 69.1500% ± 0.1500% | Stable | stable | |
| 65 Stable | 64.9277897 ± 0.00000071 | 30.8500% ± 0.1500% | Stable | stable |
Spectral Lines
Showing 50 of 1058 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 490.973351 nm | 160000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[9/2] → 3d9.(2D<5/2>).4f 2[11/2]* | Measured | NIST | |
| 493.16981 nm | 140000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[9/2] → 3d9.(2D<5/2>).4f 2[11/2]* | Measured | NIST | |
| 505.179209 nm | 120000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 495.37246 nm | 82000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[7/2] → 3d9.(2D<3/2>).4f 2[9/2]* | Measured | NIST | |
| 498.550498 nm | 70000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 506.545861 nm | 70000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[5/2] → 3d9.(2D<3/2>).4f 2[7/2]* | Measured | NIST | |
| 508.827603 nm | 57000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 740.43532 nm | 55000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[3/2]* → 3d9.(2D<5/2>).6s 2[5/2] | Measured | NIST | |
| 491.83778 nm | 54000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[7/2] → 3d9.(2D<3/2>).4f 2[9/2]* | Measured | NIST | |
| 505.890923 nm | 48000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 627.334763 nm | 47000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[7/2]* → 3d9.(2D<5/2>).5d 2[9/2] | Measured | NIST | |
| 500.679978 nm | 46000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[3/2] → 3d9.(2D<3/2>).4f 2[5/2]* | Measured | NIST | |
| 506.709423 nm | 46000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[5/2] → 3d9.(2D<3/2>).4f 2[7/2]* | Measured | NIST | |
| 509.381536 nm | 41000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 621.69385 nm | 39000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[7/2]* → 3d9.(2D<5/2>).5d 2[9/2] | Measured | NIST | |
| 600.01168 nm | 38000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[3/2]* → 3d9.(2D<5/2>).5d 2[3/2] | Measured | NIST | |
| 501.26199 nm | 37000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 468.19935 nm | 36000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[1/2] → 3d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 481.29474 nm | 36000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[1/2] → 3d9.(2D<3/2>).4f 2[3/2]* | Measured | NIST | |
| 500.985058 nm | 35000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 485.498743 nm | 34000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[9/2] → 3d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 502.127849 nm | 32000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 507.230253 nm | 32000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 594.11951 nm | 31000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[3/2]* → 3d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 512.44753 nm | 30000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d8.(3F).4s.4p.(1P*) 3G* | Measured | NIST | |
| 467.170176 nm | 29000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[1/2] → 3d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 491.291987 nm | 29000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 520.7134 nm | 29000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[7/2] → 3d8.(1G).4s.4p.(3P*) 3H* | Measured | NIST | |
| 493.155505 nm | 28000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 404.34858 nm | 27000 | Cu II | emission | 3d9.4p 1F* → 3d8.4s2 1G | Measured | NIST | |
| 504.73477 nm | 27000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 630.10137 nm | 27000 | Cu II | emission | 3d9.(2D<3/2>).5p 2[5/2]* → 3d9.(2D<3/2>).5d 2[7/2] | Measured | NIST | |
| 490.142634 nm | 26000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 492.64232 nm | 26000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 493.722031 nm | 26000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[3/2] → 3d9.(2D<3/2>).4f 2[5/2]* | Measured | NIST | |
| 508.84896 nm | 25000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[5/2] → 3d9.(2D<3/2>).4f 2[5/2]* | Measured | NIST | |
| 615.42211 nm | 25000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[3/2]* → 3d9.(2D<5/2>).5d 2[1/2] | Measured | NIST | |
| 621.98488 nm | 24000 | Cu II | emission | 3d9.(2D<3/2>).5p 2[5/2]* → 3d9.(2D<3/2>).5d 2[7/2] | Measured | NIST | |
| 526.99904 nm | 23000 | Cu II | emission | 3d9.4p 3P* → 3d8.4s2 1D | Measured | NIST | |
| 589.79758 nm | 23000 | Cu II | emission | 3d8.(3F).4s.4p.(3P*) 3G* → 3d9.(2D<5/2>).6s 2[5/2] | Measured | NIST | |
| 490.656612 nm | 21000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 508.397879 nm | 21000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[7/2] → 3d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 467.35774 nm | 20000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[1/2] → 3d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 494.3025 nm | 20000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[3/2] → 3d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 512.075319 nm | 20000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[5/2] → 3d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 644.85593 nm | 20000 | Cu II | emission | 3d9.4p 3D* → 3d8.4s2 3P | Measured | NIST | |
| 508.89421 nm | 19000 | Cu II | emission | 3d9.(2D<3/2>).4d 2[5/2] → 3d9.(2D<3/2>).4f 2[5/2]* | Measured | NIST | |
| 518.33664 nm | 19000 | Cu II | emission | 3d9.(2D<5/2>).4d 2[1/2] → 3d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 524.53423 nm | 19000 | Cu II | emission | 3d8.(3F).4s.4p.(3P*) 3F* → 3d9.(2D<5/2>).5d 2[9/2] | Measured | NIST | |
| 626.18464 nm | 19000 | Cu II | emission | 3d9.(2D<5/2>).5p 2[5/2]* → 3d9.(2D<5/2>).5d 2[7/2] | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1357.77 K |
| Boiling point | 2833.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6614 |
| 2 | p | 3.903 |
| 2 | s | 7.9802 |
| 3 | d | 15.7994 |
| 3 | p | 14.2694 |
| 3 | s | 13.4057 |
| 4 | s | 23.1576 |
Crystal Radii Detail (8)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | II | 60 | ||
| 1 | IV | 74 | estimated, | |
| 1 | VI | 91 | estimated, | |
| 2 | IV | 71 | ||
| 2 | IVSQ | 71 | ||
| 2 | V | 79 | ||
| 2 | VI | 87 | ||
| 3 | VI | LS | 60 |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 52 | p | — |
| 53 | p | — |
| 54 | p | — |
| 55 | B+ | 100% |
| 55 | B+p | — |
| 56 | B+ | 100% |
| 56 | B+p | 0.4% |
| 57 | B+ | 100% |
| 58 | B+ | 100% |
| 59 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.30088 |
| 10.1617 | — | 1.33374 |
| 10.3261 | — | 1.36743 |
| 10.4931 | — | 1.40197 |
| 10.6628 | — | 1.43738 |
| 10.8353 | — | 1.47369 |
| 11.0106 | — | 1.51091 |
| 11.1886 | — | 1.54908 |
| 11.3696 | — | 1.58821 |
| 11.5535 | — | 1.62833 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
6.0×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2.5×10-4 milligrams per liter
References (1)
Sources
Sources of this element.
Copper occasionally occurs natively, and is found in many minerals such as cuprite, malachite, azurite, chalcopyrite, and bornite.
Large copper ore deposits are found in the U.S., Chile, Zambia, Zaire, Peru, and Canada. The most important copper ores are the sulfides, the oxides, and carbonates. From these, copper is obtained by smelting, leaching, and by electrolysis.
References (1)
- [6] Copper https://periodic.lanl.gov/29.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
The copper isotope-amount ratio n(65Cu)/n(63Cu) along with the silver isotope-amount ratio n(109Ag)/n(107Ag) and lead isotope-amount ratios n(206Pb)/n(204Pb), n(207Pb)/n(204Pb), and n(208Pb)/n(204Pb) have been used to determine the origin of European coins and the flow of goods in the historical world market. Metals from Peru and Mexico and those from European mining sites have distinct isotopic signatures that enable the origin of the metal to be determined based on the isotopic compositions of silver, copper, and lead in the coins. Silver from mines in Mexico and Peru in the 16 th century was used to mint coins but did not influence the European coin market until the 18 th century [237] [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).[237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011)..
References (2)
- [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Copper.
The element property data was retrieved from publications.