Gallium (Ga)
post-transition-metalSolid
Standard Atomic Weight
69.723 uElectron configuration
[Ar] 4s2 3d10 4p1Melting point
29.76 °C (302.91 K)Boiling point
2203.85 °C (2477 K)Density
5910 kg/m³Oxidation states
−5, −4, −3, −2, −1, 0, +1, +2, +3Electronegativity (Pauling)
1.81Ionization energy (1st)
Discovery year
1875Atomic radius
130 pmDetails
Gallium is a soft post-transition metal in group 13, chemically related to aluminium and indium. It is notable for melting just above room temperature, forming low-melting alloys, and supplying semiconductors through compounds such as gallium arsenide and gallium nitride. In nature it is dispersed rather than concentrated in its own ores, so it is recovered mainly as a by-product of aluminium and zinc processing.
It is one of four metals mercury, cesium, and rubidium which can be liquid near room temperature and, thus, can be used in high-temperature thermometers. It has one of the longest liquid ranges of any metal and has a low vapor pressure even at high temperatures.
There is a strong tendency for gallium to supercool below its freezing point. Therefore, seeding may be necessary to initiate solidification.
Ultra-pure gallium has a beautiful, silvery appearance, and the solid metal exhibits a conchoidal fracture similar to glass. The metal expands 3.1 percent on solidifying; therefore, it should not be stored in glass or metal containers, because they may break as the metal solidifies.
High-purity gallium is attacked only slowly by mineral acids.
The name derives from the Latin gallia for France. It was discovered in zinc blende by the French chemist Paul-Emile Lecoq de Boisbaudran in 1875. It was first isolated in 1878 by Lecoq de Boisbaudran and the French chemist Émile-Clément Jungflesch.
First proposed to exist by Dmitri Mendeleyev in 1871 based on gaps in his newly created Periodic Table of Elements, gallium was discovered spectroscopically by the French chemist Paul-Émile Lecoq de Boisbaudran in 1875. Later that same year, Lecoq was able to obtain pure gallium through the electrolysis of a solution of gallium hydroxide (Ga(OH)3) in potassium hydroxide (KOH). Trace amounts of gallium are found in diaspore, sphalerite, germanite and bauxite as well as in the byproducts of burning coal.
From the Latin word Gallia, France; also from Latin, gallus, a translation of "Lecoq," a cock. Predicted and described by Mendeleev as ekaaluminum, and discovered spectroscopically by Lecoq de Boisbaudran in 1875, who in the same year obtained the free metal by electrolysis of a solution of the hydroxide in KOH.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
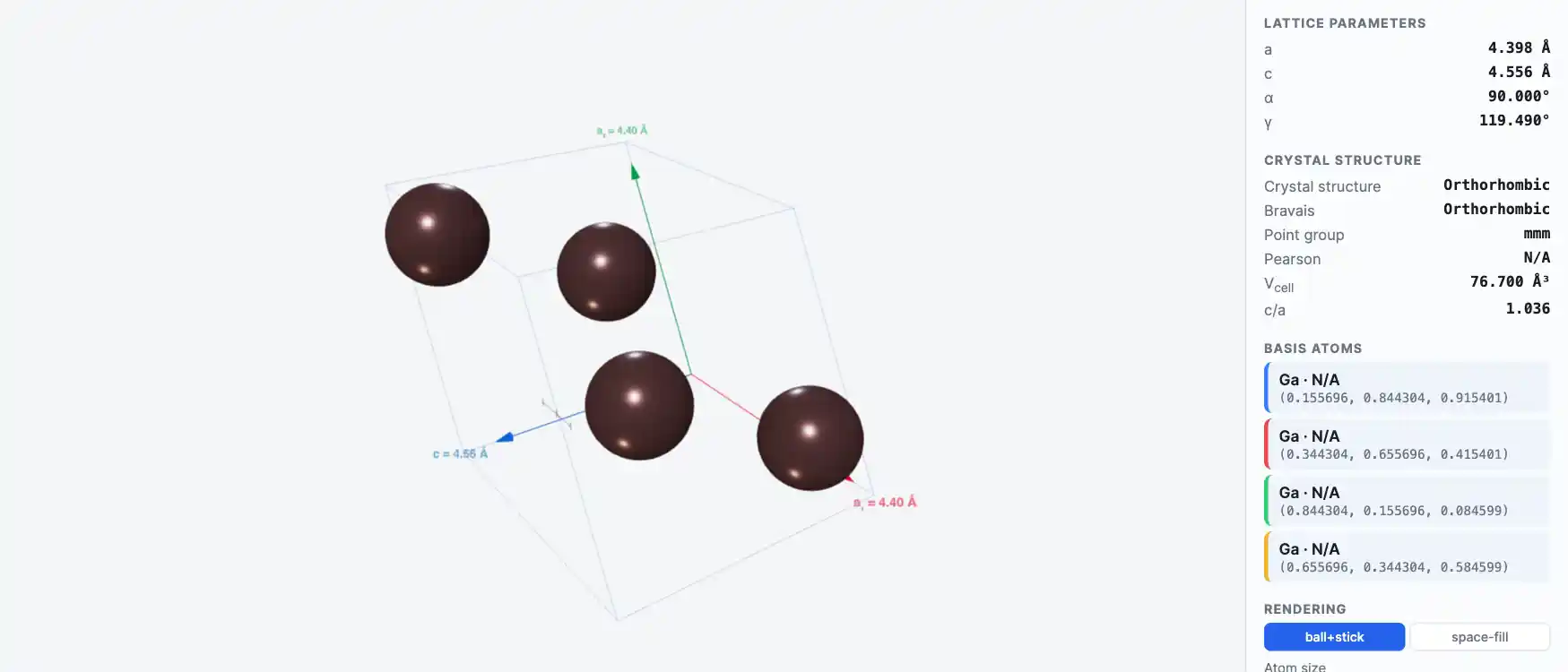
Crystal Structure
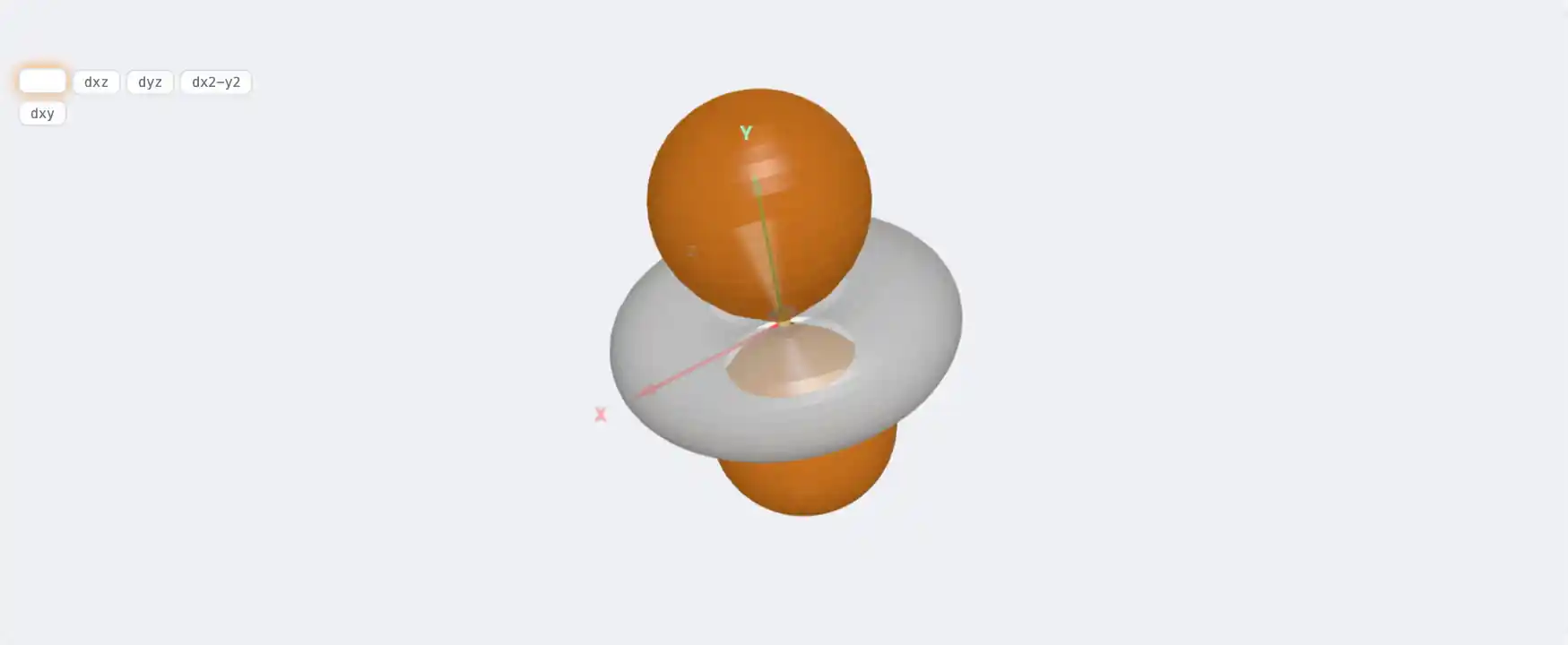
Electronic Structure
Identifiers
Electron Configuration Measured
Ga: 3d¹⁰ 4s² 4p¹[Ar] 3d¹⁰ 4s² 4p¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 69 Stable | 68.9255735 ± 0.0000013 | 60.1080% | Stable |
| 71 Stable | 70.92470258 ± 0.00000087 | 39.8920% | Stable |
Phase / State
Reason: 4.8 °C below melting point (29.76 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 31 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ga I | 0 | 342 | 23 | 342 |
| Ga II | +1 | 176 | 10 | 176 |
| Ga III | +2 | 113 | 2 | 113 |
| Ga IV | +3 | 594 | 0 | 594 |
| Ga V | +4 | 185 | 0 | 185 |
| Ga VI | +5 | 501 | 0 | 501 |
| Ga VII | +6 | 451 | 0 | 451 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ga I | 0 | 262 |
| Ga II | +1 | 96 |
| Ga III | +2 | 61 |
| Ga IV | +3 | 192 |
| Ga V | +4 | 92 |
| Ga VI | +5 | 158 |
| Ga VII | +6 | 181 |
| Ga VIII | +7 | 2 |
| Ga IX | +8 | 2 |
| Ga X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 4 | N/A | 47 pm |
| +3 | 5 | N/A | 55.00000000000001 pm |
| +3 | 6 | N/A | 62 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 69 Stable | 68.9255735 ± 0.0000013 | 60.1080% ± 0.0090% | Stable | stable | |
| 71 Stable | 70.92470258 ± 0.00000087 | 39.8920% ± 0.0090% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 417.33 nm | N/A | ID 486 | emission | 3s2.3p3 2P* → 3s2.3p3 2P* | Measured | NIST | |
| 424.0525 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.70p 2P* | Measured | NIST | |
| 424.0525 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.70p 2P* | Measured | NIST | |
| 424.0651 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.69p 2P* | Measured | NIST | |
| 424.0651 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.69p 2P* | Measured | NIST | |
| 424.08 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.68p 2P* | Measured | NIST | |
| 424.08 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.68p 2P* | Measured | NIST | |
| 424.0924 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.67p 2P* | Measured | NIST | |
| 424.0924 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.67p 2P* | Measured | NIST | |
| 424.1098 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.66p 2P* | Measured | NIST | |
| 424.1098 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.66p 2P* | Measured | NIST | |
| 424.1257 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.65p 2P* | Measured | NIST | |
| 424.1257 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.65p 2P* | Measured | NIST | |
| 424.1406 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.64p 2P* | Measured | NIST | |
| 424.1406 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.64p 2P* | Measured | NIST | |
| 424.1588 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.63p 2P* | Measured | NIST | |
| 424.1588 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.63p 2P* | Measured | NIST | |
| 424.1761 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.62p 2P* | Measured | NIST | |
| 424.1761 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.62p 2P* | Measured | NIST | |
| 424.1948 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.61p 2P* | Measured | NIST | |
| 424.1948 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.61p 2P* | Measured | NIST | |
| 424.2157 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.60p 2P* | Measured | NIST | |
| 424.2157 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.60p 2P* | Measured | NIST | |
| 424.2367 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.59p 2P* | Measured | NIST | |
| 424.2367 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.59p 2P* | Measured | NIST | |
| 424.2582 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.58p 2P* | Measured | NIST | |
| 424.2582 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.58p 2P* | Measured | NIST | |
| 424.2826 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.57p 2P* | Measured | NIST | |
| 424.2826 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.57p 2P* | Measured | NIST | |
| 424.3887 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.53p 2P* | Measured | NIST | |
| 424.3887 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.53p 2P* | Measured | NIST | |
| 424.4204 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.52p 2P* | Measured | NIST | |
| 424.4204 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.52p 2P* | Measured | NIST | |
| 424.4531 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.51p 2P* | Measured | NIST | |
| 424.4531 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.51p 2P* | Measured | NIST | |
| 424.4886 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.50p 2P* | Measured | NIST | |
| 424.4886 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.50p 2P* | Measured | NIST | |
| 424.5261 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.49p 2P* | Measured | NIST | |
| 424.5261 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.49p 2P* | Measured | NIST | |
| 424.5675 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.48p 2P* | Measured | NIST | |
| 424.5675 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.48p 2P* | Measured | NIST | |
| 424.6112 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.47p 2P* | Measured | NIST | |
| 424.6112 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.47p 2P* | Measured | NIST | |
| 424.6563 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.46p 2P* | Measured | NIST | |
| 424.6563 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.46p 2P* | Measured | NIST | |
| 424.7046 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.45p 2P* | Measured | NIST | |
| 424.7046 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.45p 2P* | Measured | NIST | |
| 424.7569 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.44p 2P* | Measured | NIST | |
| 424.7569 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.44p 2P* | Measured | NIST | |
| 424.8143 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.43p 2P* | Measured | NIST | |
| 424.8143 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.43p 2P* | Measured | NIST | |
| 424.8743 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.42p 2P* | Measured | NIST | |
| 424.8743 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.42p 2P* | Measured | NIST | |
| 424.94 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.41p 2P* | Measured | NIST | |
| 424.94 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.41p 2P* | Measured | NIST | |
| 425.4789 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.34d 2D | Measured | NIST | |
| 425.4799 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.34d 2D | Measured | NIST | |
| 426.035 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.30d 2D | Measured | NIST | |
| 426.0365 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.30d 2D | Measured | NIST | |
| 426.6348 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.27d 2D | Measured | NIST | |
| 426.6367 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.27d 2D | Measured | NIST | |
| 427.1688 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.25d 2D | Measured | NIST | |
| 427.1712 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.25d 2D | Measured | NIST | |
| 427.8589 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.23d 2D | Measured | NIST | |
| 427.8621 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.23d 2D | Measured | NIST | |
| 428.7731 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.21d 2D | Measured | NIST | |
| 428.7774 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.21d 2D | Measured | NIST | |
| 429.3459 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.20d 2D | Measured | NIST | |
| 429.3507 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.20d 2D | Measured | NIST | |
| 430.0203 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.19d 2D | Measured | NIST | |
| 430.026 nm | N/A | Ga I | emission | 4s2.5s 2S → 4s2.19d 2D | Measured | NIST | |
| 448.84 nm | N/A | ID 505 | emission | 5p 2P* → 5d 2D | Measured | NIST | |
| 459.16 nm | N/A | ID 505 | emission | 5s 2S → 5p 2P* | Measured | NIST | |
| 557 nm | N/A | ID 482 | emission | 1s.5s 3S → 1s.5p 3P* | Measured | NIST | |
| 567.7 nm | N/A | ID 498 | emission | 3s2.3p2 3P → 3s2.3p2 3P | Measured | NIST | |
| 587 nm | N/A | ID 482 | emission | 1s.4p 3P* → 1s.4d 3D | Measured | NIST | |
| 675 nm | N/A | ID 486 | emission | 3s2.3p3 2D* → 3s2.3p3 2D* | Measured | NIST | |
| 706.7 nm | N/A | ID 505 | emission | 4p 2P* → 4d 2D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 302.91 K |
| Boiling point | 2502.15 K |
| Triple point (temperature) | 302.92 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6906 |
| 2 | p | 3.9092 |
| 2 | s | 8.401 |
| 3 | d | 15.9067 |
| 3 | p | 14.7964 |
| 3 | s | 14.0038 |
| 4 | p | 24.7784 |
| 4 | s | 23.9332 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | IV | 61 | ||
| 3 | V | 69 | ||
| 3 | VI | 76 | from r^3 vs V plots, |
Isotope Decay Modes (51)
| Isotope | Mode | Intensity |
|---|---|---|
| 56 | p | — |
| 57 | p | — |
| 58 | p | — |
| 59 | p | — |
| 60 | B+ | 100% |
| 60 | B+p | 1.6% |
| 60 | B+A | 0% |
| 61 | B+ | 100% |
| 61 | B+p | 0.3% |
| 62 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.98527 |
| 10.1617 | — | 2.98141 |
| 10.3261 | — | 2.97756 |
| 10.4931 | — | 2.9737 |
| 10.6628 | — | 2.96986 |
| 10.8353 | — | 2.96602 |
| 11.0106 | — | 2.95695 |
| 11.1886 | — | 2.90859 |
| 11.3696 | — | 2.86103 |
| 11.5535 | — | 2.81425 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.9×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
Gallium is often found as a trace element in diaspore, sphalerite, germanite, bauxite, and coal. Some flue dusts from burning coal have been shown to contain as much 1.5 percent gallium.
References (1)
- [6] Gallium https://periodic.lanl.gov/31.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Gallium.
The element property data was retrieved from publications.