Germanium (Ge)
metalloidSolid
Standard Atomic Weight
72.63 uElectron configuration
[Ar] 4s2 3d10 4p2Melting point
938.25 °C (1211.4 K)Boiling point
2832.85 °C (3106 K)Density
5323.4 kg/m³Oxidation states
−4, −3, −2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
2.01Ionization energy (1st)
Discovery year
1886Atomic radius
125 pmDetails
Germanium is a hard, brittle metalloid in group 14, chemically intermediate between silicon and tin. It is a covalent semiconductor with a narrow band gap and forms stable compounds mainly in the +4 and +2 oxidation states. The element is not mined as a principal ore in most operations; it is commonly recovered as a by-product from zinc processing and from some coal-derived materials. Its technological importance rests on infrared optics, fiber-optic materials, semiconductor devices, and specialty catalysts.
The element is a gray-white metalloid. In pure state, the element is crystalline and brittle, retaining its luster in air at room temperature. It is a very important semiconductor. Zone-refining techniques have led to production of crystalline germanium for semiconductor use with an impurity of only one part in 1010.
The name derives from the Latin germania for Germany. It was discovered and isolated by the German chemist Clemens-Alexander Winkler in 1886 in the mineral argyrodite (GeS2×4Ag2S).
First proposed to exist by Dmitri Mendeleyev in 1871 based on gaps in his newly created Periodic Table of Elements, germanium was discovered by the German chemist Clemens Winkler in the mineral argyrodite (Ag8GeS6) in 1886. Today, germanium is primarily obtained from the smelting of zinc ores and from the byproducts of burning certain types of coal.
From the Latin word Germania, Germany. Mendeleev predicted the existence of Germanium in 1871 as ekasilicon, and the element was discovered by Winkler in 1886.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
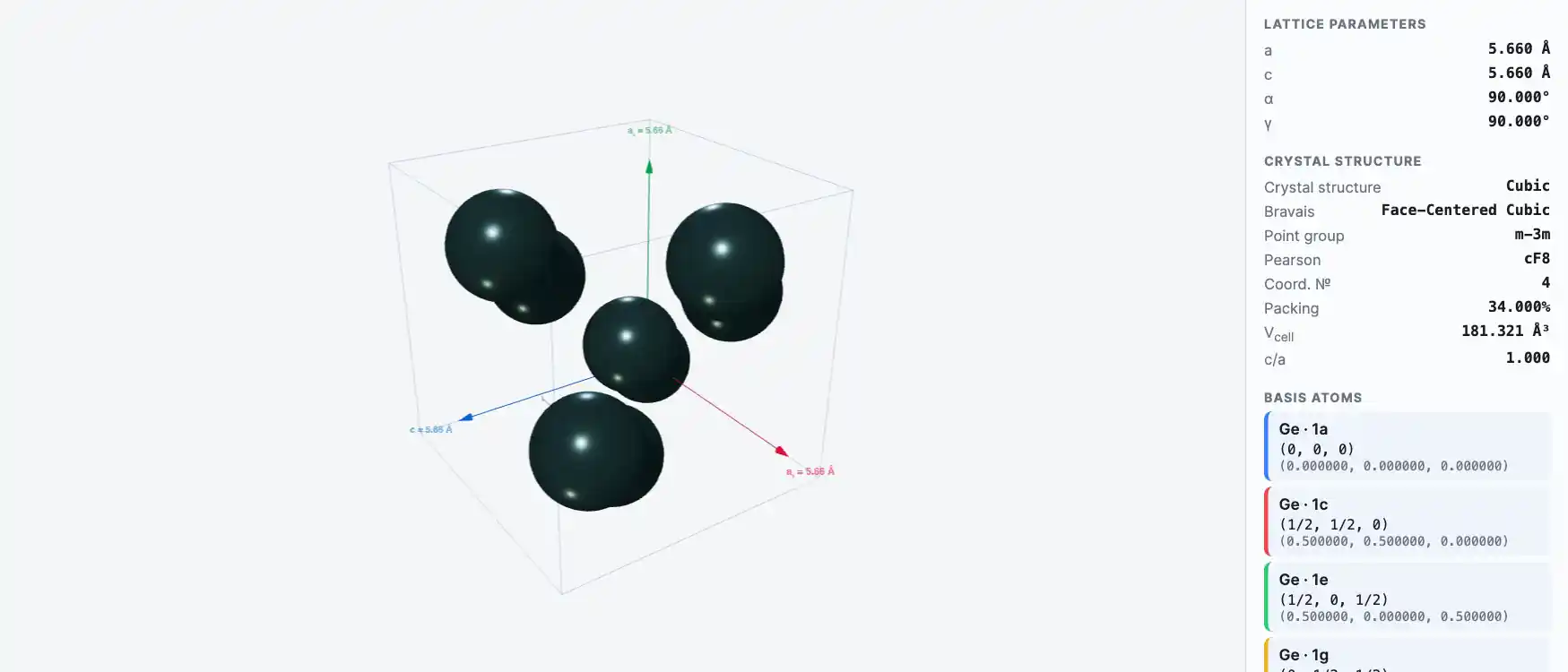
Crystal Structure
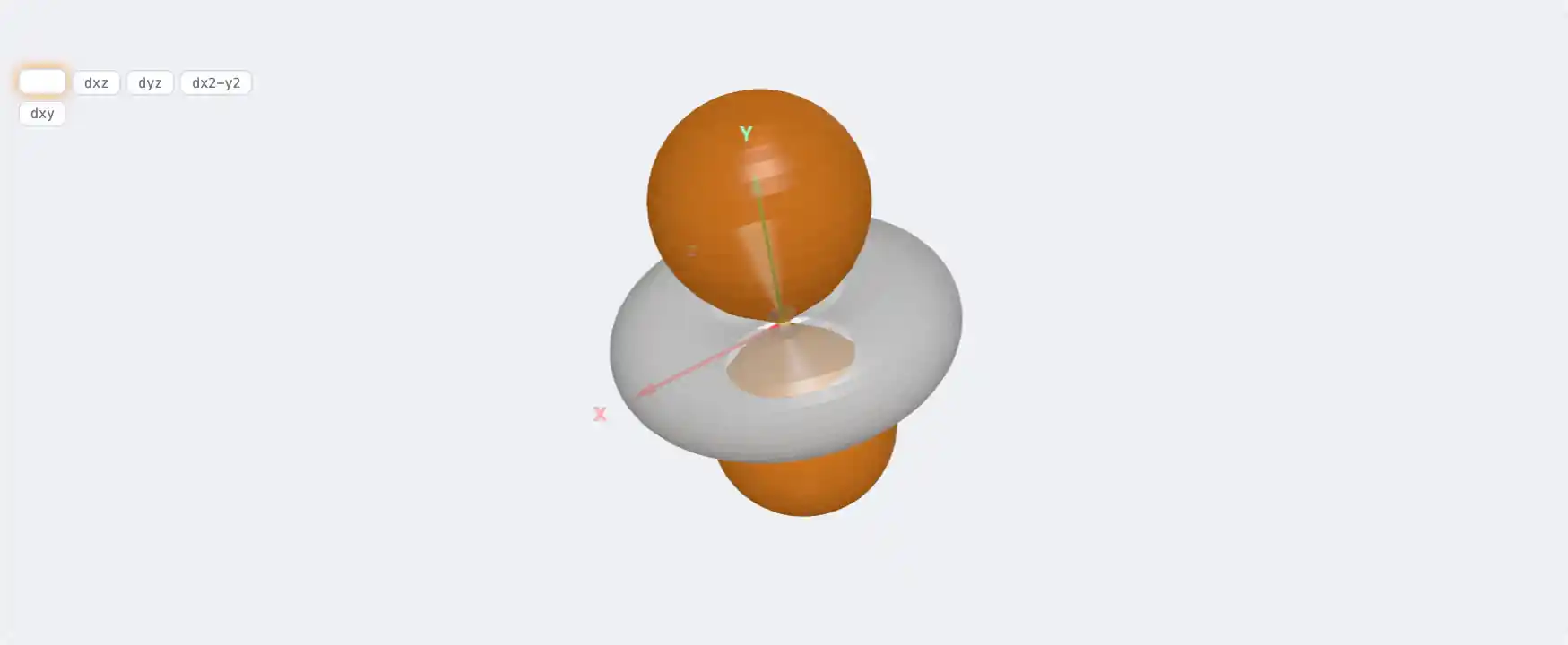
Electronic Structure
Identifiers
Electron Configuration Measured
Ge: 3d¹⁰ 4s² 4p²[Ar] 3d¹⁰ 4s² 4p²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 70 Stable | 69.92424875 ± 0.0000009 | 20.5700% | Stable |
| 72 Stable | 71.922075826 ± 0.000000081 | 27.4500% | Stable |
| 73 Stable | 72.923458956 ± 0.000000061 | 7.7500% | Stable |
| 74 Stable | 73.921177761 ± 0.000000013 | 36.5000% | Stable |
Phase / State
Reason: 913.3 °C below melting point (938.25 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 32 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ge I | 0 | 224 | 26 | 223 |
| Ge II | +1 | 149 | 20 | 149 |
| Ge III | +2 | 55 | 0 | 0 |
| Ge IV | +3 | 27 | 0 | 0 |
| Ge V | +4 | 37 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ge I | 0 | 621 |
| Ge II | +1 | 129 |
| Ge III | +2 | 48 |
| Ge IV | +3 | 55 |
| Ge V | +4 | 102 |
| Ge VI | +5 | 105 |
| Ge VII | +6 | 168 |
| Ge VIII | +7 | 2 |
| Ge IX | +8 | 2 |
| Ge X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 73 pm |
| +4 | 4 | N/A | 39 pm |
| +4 | 6 | N/A | 53 pm |
Compounds
Isotopes (4)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 70 Stable | 69.92424875 ± 0.0000009 | 20.5700% ± 0.2700% | Stable | stable | |
| 72 Stable | 71.922075826 ± 0.000000081 | 27.4500% ± 0.3200% | Stable | stable | |
| 73 Stable | 72.923458956 ± 0.000000061 | 7.7500% ± 0.1200% | Stable | stable | |
| 74 Stable | 73.921177761 ± 0.000000013 | 36.5000% ± 0.2000% | Stable | stable |
Spectral Lines
Showing 50 of 57 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 474.18054 nm | 1000 | Ge II | emission | 4s2.5p 2P* → 4s2.5d 2D | Measured | NIST | |
| 481.46084 nm | 1000 | Ge II | emission | 4s2.5p 2P* → 4s2.5d 2D | Measured | NIST | |
| 589.33885 nm | 1000 | Ge II | emission | 4s2.5s 2S → 4s2.5p 2P* | Measured | NIST | |
| 602.10412 nm | 500 | Ge II | emission | 4s2.5s 2S → 4s2.5p 2P* | Measured | NIST | |
| 517.86474 nm | 200 | Ge II | emission | 4s2.4d 2D → 4s2.4f 2F* | Measured | NIST | |
| 607.834 nm | 150 | Ge II | emission | 4s.4p.(3P*).5s 4P* → 4s.4p.(3P*).5p 4D | Measured | NIST | |
| 626.8068 nm | 150 | Ge II | emission | 4s2.4f 2F* → 4s2.6g 2G | Measured | NIST | |
| 513.17516 nm | 100 | Ge II | emission | 4s2.4d 2D → 4s2.4f 2F* | Measured | NIST | |
| 626.8341 nm | 100 | Ge II | emission | 4s2.4f 2F* → 4s2.6g 2G | Measured | NIST | |
| 633.63765 nm | 100 | Ge II | emission | 4s2.5p 2P* → 4s2.6s 2S | Measured | NIST | |
| 648.41813 nm | 100 | Ge II | emission | 4s2.5p 2P* → 4s2.6s 2S | Measured | NIST | |
| 628.34518 nm | 75 | Ge II | emission | 4s2.5d 2D → 4s2.6f 2F* | Measured | NIST | |
| 422.656259 nm | 70 | Ge I | emission | 4s2.4p2 1S → 4s2.4p.5s 1P* | Measured | NIST | |
| 482.40972 nm | 50 | Ge II | emission | 4s2.5p 2P* → 4s2.5d 2D | Measured | NIST | |
| 626.7136 nm | 50 | Ge II | emission | 4s2.5d 2D → 4s2.6f 2F* | Measured | NIST | |
| 678.0486 nm | 50 | Ge II | emission | 4s2.6p 2P* → 4s2.7d 2D | Measured | NIST | |
| 704.93692 nm | 50 | Ge II | emission | 4s.4p2 2D → 4s2.5p 2P* | Measured | NIST | |
| 384.50994 nm | 30 | Ge II | emission | 4s.4p2 4P → 4s2.5p 2P* | Measured | NIST | |
| 714.53898 nm | 30 | Ge II | emission | 4s.4p2 2D → 4s2.5p 2P* | Measured | NIST | |
| 494.12769 nm | 20 | Ge II | emission | 4s2.4d 2D → 4s2.6p 2P* | Measured | NIST | |
| 520.56488 nm | 20 | Ge II | emission | 4s2.4f 2F* → 4s2.7g 2G | Measured | NIST | |
| 696.63205 nm | 20 | Ge II | emission | 4s.4p2 2D → 4s2.5p 2P* | Measured | NIST | |
| 439.1656 nm | 15 | Ge II | emission | 4s2.4f 2F* → 4s2.9g 2G | Measured | NIST | |
| 520.58372 nm | 15 | Ge II | emission | 4s2.4f 2F* → 4s2.7g 2G | Measured | NIST | |
| 552.2987 nm | 15 | Ge II | emission | 4s2.6p 2P* → 4s2.8d 2D | Measured | NIST | |
| 439.179 nm | 10 | Ge II | emission | 4s2.4f 2F* → 4s2.9g 2G | Measured | NIST | |
| 466.2311 nm | 10 | Ge II | emission | 4s2.5d 2D → 4s2.8f 2F* | Measured | NIST | |
| 468.582849 nm | 10 | Ge I | emission | 4s2.4p2 1S → 4s2.4p.5s 3P* | Measured | NIST | |
| 493.40754 nm | 10 | Ge II | emission | 4s2.4d 2D → 4s2.6p 2P* | Measured | NIST | |
| 517.84615 nm | 10 | Ge II | emission | 4s2.4d 2D → 4s2.4f 2F* | Measured | NIST | |
| 569.19543 nm | 9 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3D | Measured | NIST | |
| 580.2093 nm | 9 | Ge I | emission | 4s2.4p.5s 1P* → 4s2.4p.6p 1D | Measured | NIST | |
| 556.47408 nm | 8 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3S | Measured | NIST | |
| 560.70101 nm | 8 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3P | Measured | NIST | |
| 565.596 nm | 8 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3D | Measured | NIST | |
| 562.14256 nm | 7 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 1P | Measured | NIST | |
| 733.0383 nm | 7 | Ge I | emission | 4s2.4p.5p 1P → 4s2.4p.7d (1/2,3/2)* | Measured | NIST | |
| 738.4208 nm | 7 | Ge I | emission | 4s2.4p.5p 3D → 4s2.4p.8s (1/2,1/2)* | Measured | NIST | |
| 526.58915 nm | 6 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3P | Measured | NIST | |
| 551.32634 nm | 6 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 1D | Measured | NIST | |
| 561.61353 nm | 6 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3P | Measured | NIST | |
| 566.4226 nm | 6 | Ge I | emission | 4s2.4p.5s 1P* → 4s2.4p.6p 1S | Measured | NIST | |
| 570.17765 nm | 6 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 1P | Measured | NIST | |
| 580.1029 nm | 6 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3D | Measured | NIST | |
| 655.74883 nm | 6 | Ge I | emission | 4s2.4p.5s 1P* → 4s2.4p.6p 3D | Measured | NIST | |
| 713.0126 nm | 6 | Ge I | emission | 4s2.4p.5p 3D → 4s2.4p.7d (3/2,5/2)* | Measured | NIST | |
| 740.2648 nm | 6 | Ge I | emission | 4s2.4p.5p 1P → 4s2.4p.6d (3/2,3/2)* | Measured | NIST | |
| 518.4103 nm | 5 | Ge II | emission | 4s2.5d 2D → 4s2.7f 2F* | Measured | NIST | |
| 566.4842 nm | 5 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 571.78769 nm | 5 | Ge I | emission | 4s2.4p.5s 3P* → 4s2.4p.6p 3D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1211.4 K |
| Boiling point | 3106.15 K |
| Critical point (temperature) | 9802.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7063 |
| 2 | p | 3.9178 |
| 2 | s | 8.6352 |
| 3 | d | 15.7487 |
| 3 | p | 14.9864 |
| 3 | s | 14.2103 |
| 4 | p | 25.2196 |
| 4 | s | 23.9564 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 87 | Ahrens (1952) ionic radius, | |
| 4 | IV | 53 | ||
| 4 | VI | 67 | from r^3 vs V plots, |
Isotope Decay Modes (50)
| Isotope | Mode | Intensity |
|---|---|---|
| 58 | 2p | — |
| 59 | B+ | 100% |
| 59 | B+p | 93% |
| 59 | 2p | 0.2% |
| 60 | B+ | 100% |
| 60 | B+p | 100% |
| 60 | 2p | 14% |
| 61 | B+ | 100% |
| 61 | B+p | 87% |
| 62 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.01183 |
| 10.1617 | — | 3.05548 |
| 10.3261 | — | 3.09976 |
| 10.4931 | — | 3.14468 |
| 10.6628 | — | 3.19025 |
| 10.8353 | — | 3.21825 |
| 11.0106 | — | 3.20755 |
| 11.1886 | — | 3.19689 |
| 11.3696 | — | 3.18626 |
| 11.5535 | — | 3.17568 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.5 milligrams per kilogram
References (1)
- [5] Germanium https://education.jlab.org/itselemental/ele032.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
5×10-5 milligrams per liter
References (1)
- [5] Germanium https://education.jlab.org/itselemental/ele032.html
Sources
Sources of this element.
The metal is found in
▸ argyrodite, a sulfide of germanium and silver;
▸ germanite, which contains 8 percent of the element;
▸ zinc ores;
▸ coal; and
▸ other minerals
The element is commercially obtained from the dust from smelters that process zinc ores. It is also recovered from combustion by-products of certain coals.
Germanium can be separated from other metals by fractional distillation of its volatile tetrachloride. These techniques permit the production of germanium of ultra-high purity.
References (1)
- [6] Germanium https://periodic.lanl.gov/32.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Germanium.
The element property data was retrieved from publications.