Bromine (Br)
halogenLiquid
Standard Atomic Weight
79.904 u [79.901, 79.907]Electron configuration
[Ar] 4s2 3d10 4p5Melting point
-7.2 °C (265.95 K)Boiling point
58.8 °C (331.95 K)Density
3102.8 kg/m³Oxidation states
−1, +1, +2, +3, +4, +5, +7Electronegativity (Pauling)
2.96Ionization energy (1st)
Discovery year
1825Atomic radius
115 pmDetails
Bromine is a halogen, group 17 element, and the only nonmetal that is liquid near room temperature. Elemental bromine occurs as diatomic Br₂ and is a dense, volatile, strongly oxidizing substance. In nature bromine is found mainly as bromide ions in seawater, salt lakes, and subsurface brines. Its chemistry is intermediate between chlorine and iodine, with important roles in flame retardants, drilling fluids, pharmaceuticals, and photographic chemistry.
Bromine is the only nonmetallic liquid element. It is a heavy, mobile, reddish-brown liquid, volatilizing readily at room temperature to a red vapor with a strong disagreeable odor, resembling chlorine, and having a very irritating effect on the eyes and throat; it is readily soluble in water or carbon disulfide, forming a red solution, is less active than chlorine but more so than iodine; it unites readily with many elements and has a bleaching action; when spilled on the skin it produces painful sores. It presents a serious health hazard, and maximum safety precautions should be taken when handling it.
The name derives from the Greek bromos for "bad stench" or "bad odour". It was first prepared by the German chemist Carl Löwig in 1825, but it was first publicly announced in 1826 by the French chemist and pharmacist Antoine-Jérôme Balard, and so the discovery is, therefore, credited to him.
The only nonmetallic element that is a liquid at normal room temperatures, bromine was produced by Carl Löwig, a young chemistry student, the summer before starting his freshman year at Heidelberg. When he showed his professor, Leopold Gmelin, the red, smelly liquid he had produced, Gmelin realized that this was an unknown substance and encouraged Löwig to produce more of it so they could study it in detail. Unfortunately, winter exams and the holidays delayed Löwig's work long enough for another chemist, Antoine-Jérôme Balard, to publish a paper in 1826 describing the new element. Balard was credited with the discovery and named it after the greek word for stench, bromos. Today, bromine is primarily obtained by treating brines from wells in Michigan and Arkansas with chlorine.
From the Greek word bromos, stench. Discovered by Balard in 1826, but not prepared in quantity until 1860.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
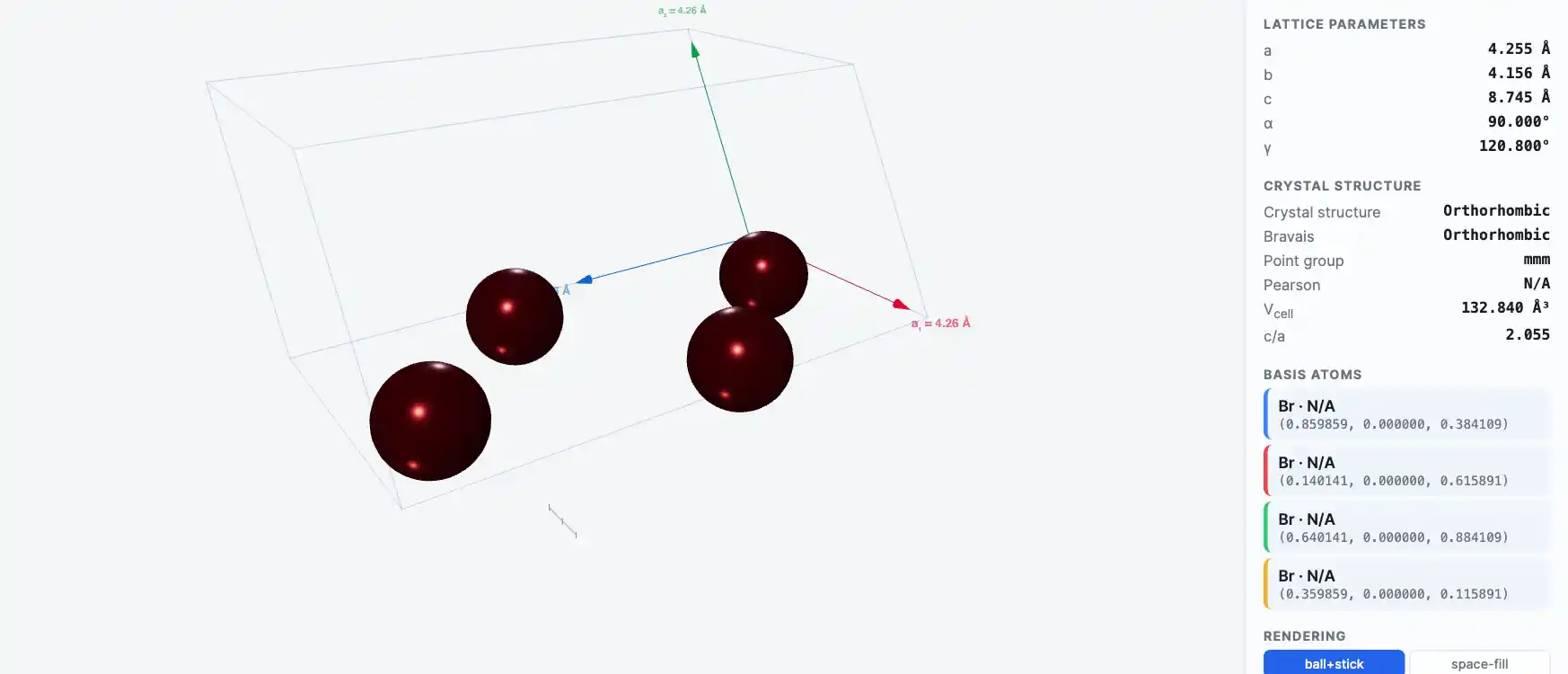
Crystal Structure
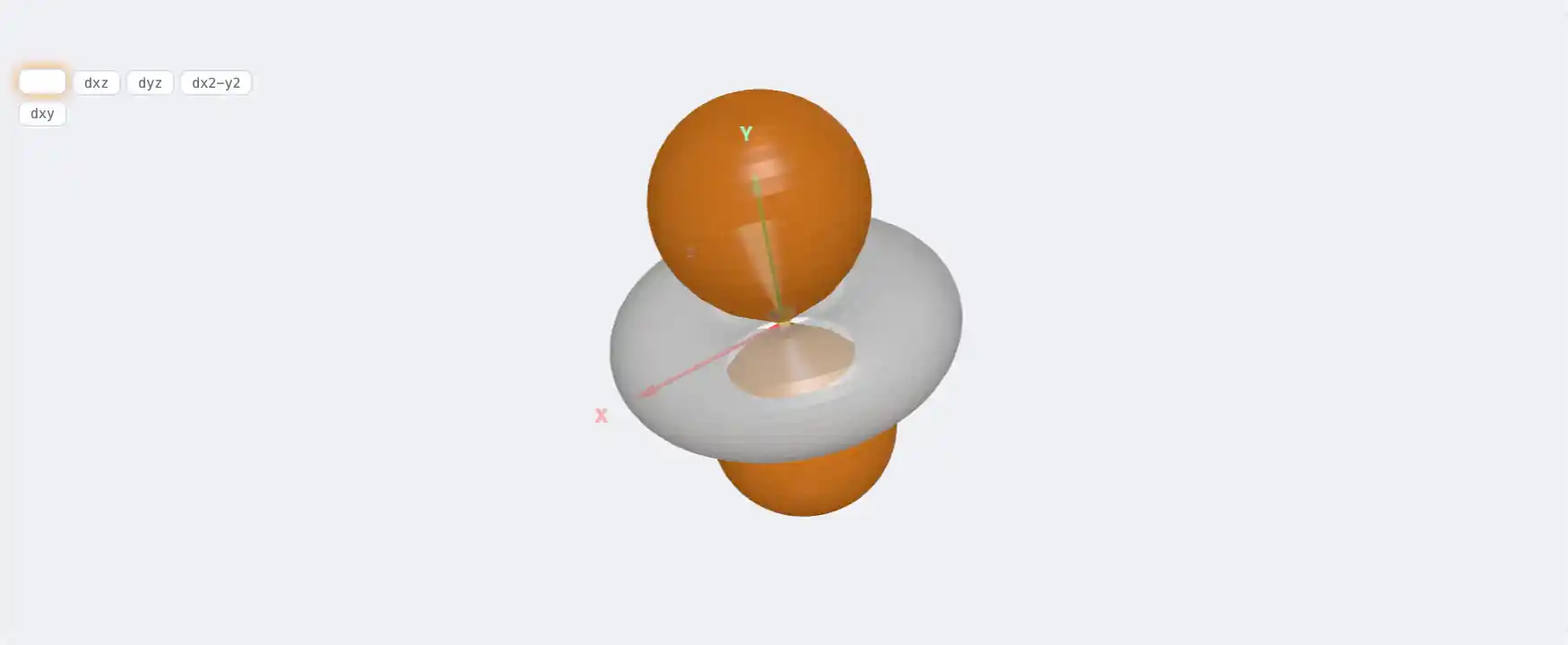
Electronic Structure
Identifiers
Electron Configuration Measured
Br: 3d¹⁰ 4s² 4p⁵[Ar] 3d¹⁰ 4s² 4p⁵1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁵Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 79 Stable | 78.9183376 ± 0.0000014 | 50.6900% | Stable |
| 81 Stable | 80.9162897 ± 0.0000014 | 49.3100% | Stable |
Phase / State
Reason: between melting point (-7.2 °C) and boiling point (58.8 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Not available for liquid phase
Advanced
Atomic Spectra
Showing 10 of 35 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Br I | 0 | 170 | 54 | 170 |
| Br II | +1 | 118 | 3 | 0 |
| Br III | +2 | 75 | 0 | 0 |
| Br IV | +3 | 136 | 0 | 136 |
| Br V | +4 | 22 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Br I | 0 | 265 |
| Br II | +1 | 132 |
| Br III | +2 | 53 |
| Br IV | +3 | 43 |
| Br V | +4 | 10 |
| Br VI | +5 | 12 |
| Br VII | +6 | 6 |
| Br VIII | +7 | 12 |
| Br IX | +8 | 5 |
| Br X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -1 | 6 | N/A | 196 pm |
| +3 | 4 | N/A | 59 pm |
| +5 | 3 | N/A | 31 pm |
| +7 | 4 | N/A | 25 pm |
| +7 | 6 | N/A | 39 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 79 Stable | 78.9183376 ± 0.0000014 | 50.6900% ± 0.0700% | Stable | stable | |
| 81 Stable | 80.9162897 ± 0.0000014 | 49.3100% ± 0.0700% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 265.95 K |
| Boiling point | 331.95 K |
| Critical point (temperature) | 588.15 K |
| Critical point (pressure) | 10.34 MPa |
| Triple point (temperature) | 265.9 K |
| Triple point (pressure) | 5.88 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7529 |
| 2 | p | 3.9436 |
| 2 | s | 9.3566 |
| 3 | d | 15.4409 |
| 3 | p | 15.4292 |
| 3 | s | 14.7815 |
| 4 | p | 25.972 |
| 4 | s | 24.4472 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -1 | VI | 182 | Pauling's (1960) crystal radius, | |
| 3 | IVSQ | 73 | ||
| 5 | IIIPY | 45 | ||
| 7 | IV | 39 | ||
| 7 | VI | 53 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 65 | p | — |
| 66 | p | — |
| 67 | p | — |
| 68 | p | — |
| 69 | p | 100% |
| 70 | B+ | 100% |
| 70 | B+p | — |
| 71 | B+ | 100% |
| 72 | B+ | 100% |
| 73 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 5.16199 |
| 10.1617 | — | 5.31855 |
| 10.3261 | — | 5.47986 |
| 10.4931 | — | 5.64606 |
| 10.6628 | — | 5.8173 |
| 10.8353 | — | 5.99373 |
| 11.0106 | — | 6.17552 |
| 11.1886 | — | 6.36281 |
| 11.3696 | — | 6.5558 |
| 11.5535 | — | 6.75463 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.4 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
6.73×101 milligrams per liter
References (1)
Sources
Sources of this element.
A member of the halogen group, bromine is obtained from natural brines from wells in Michigan and Arkansas. Some bromine is extracted today from seawater, which contains only about 85 ppm.
References (1)
- [6] Bromine https://periodic.lanl.gov/35.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Much of the bromine output in the U.S. was used in the production of ethylene dibromide, a lead scavenger used in making gasoline anti-knock compounds. Lead in gasoline, however, has been drastically reduced due to environmental considerations. This will greatly affect future production of bromine.
References (1)
- [6] Bromine https://periodic.lanl.gov/35.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Bromine.
The element property data was retrieved from publications.