Krypton (Kr)
noble-gasGas
Standard Atomic Weight
83.798 uElectron configuration
[Ar] 4s2 3d10 4p6Melting point
-157.36 °C (115.79 K)Boiling point
-153.42 °C (119.73 K)Density
3.733 kg/m³Oxidation states
0, +1, +2Electronegativity (Pauling)
3Ionization energy (1st)
Discovery year
1898Atomic radius
N/ADetails
Krypton is a heavy noble gas in group 18. It is chemically very inert under ordinary conditions, monatomic, colorless, and present in air only as a minor trace constituent. Its closed electron shell makes compound formation difficult, but not impossible under strongly oxidizing or low-temperature laboratory conditions. Technologically, krypton is valued mainly for specialized lighting, gas lasers, insulating gas mixtures, and isotope applications rather than bulk chemical reactivity.
Krypton is a "noble" gas. It is characterized by its brilliant green and orange spectral lines.
The name derives from the Greek kryptos for "concealed" or "hidden". It was discovered in liquefied atmospheric air by the Scottish chemist William Ramsay and the English chemist Morris William Travers in 1898. A wavelength in the atomic spectrum of 86Kr is a fundamental standard of length.
Krypton was discovered on May 30, 1898 by Sir William Ramsay, a Scottish chemist, and Morris M. Travers, an English chemist, while studying liquefied air. Small amounts of liquid krypton remained behind after the more volatile components of liquid air had boiled away. The earth's atmosphere is about 0.0001% krypton.
From the Greek word kryptos, hidden. Discovered in 1898 by Ramsay and Travers in the residue left after liquid air had nearly boiled away. In 1960 it was internationally agreed that the fundamental unit of length, the meter, should be defined in terms of the orange-red spectral line of 86Kr. This replaced the standard meter of Paris, which was defined in terms of a bar made of a platinum-iridium alloy. In October 1983, the meter, which originally was defined as being one ten millionth of a quadrant of the earth's polar circumference, was again redefined by the International Bureau of Weights and Measures as being the length of a path traveled by light in a vacuum during a time interval of 1/299,792,458 of a second.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
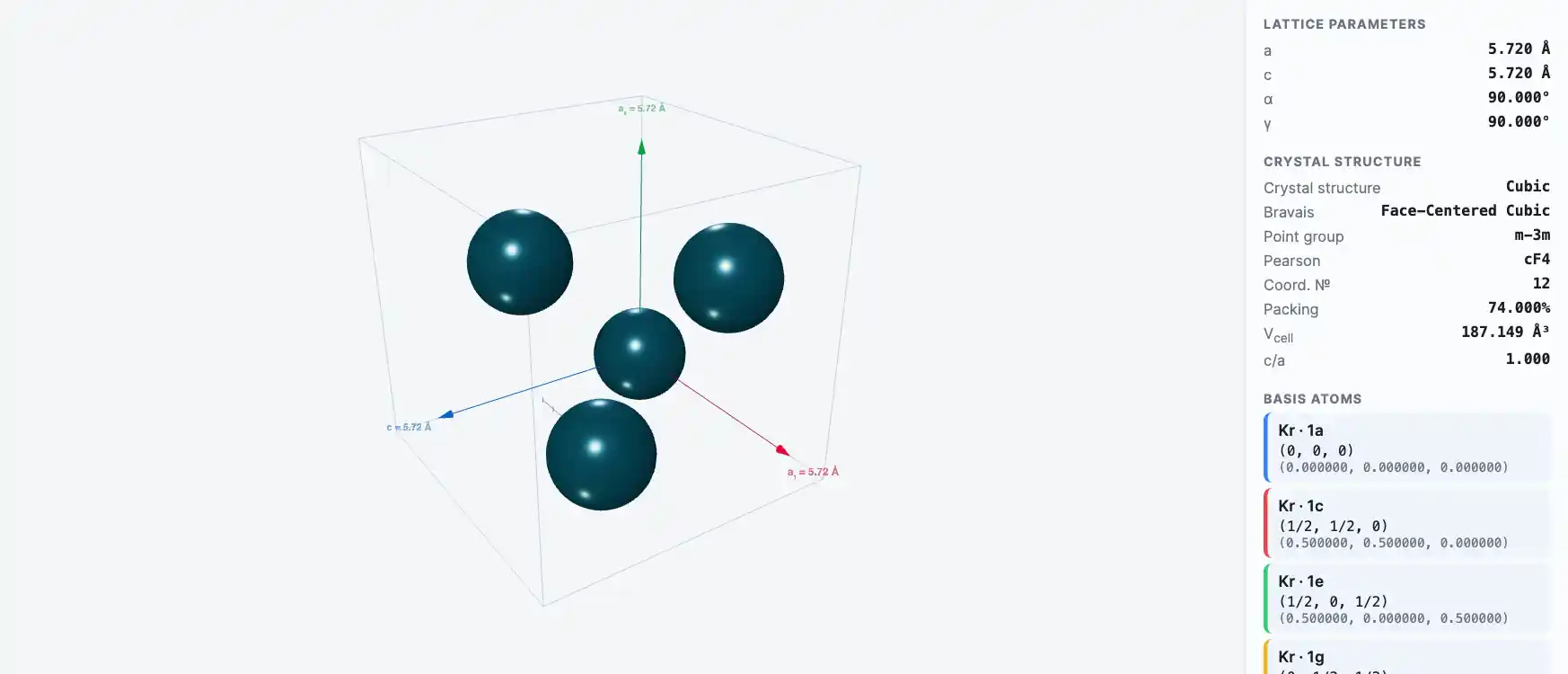
Crystal Structure
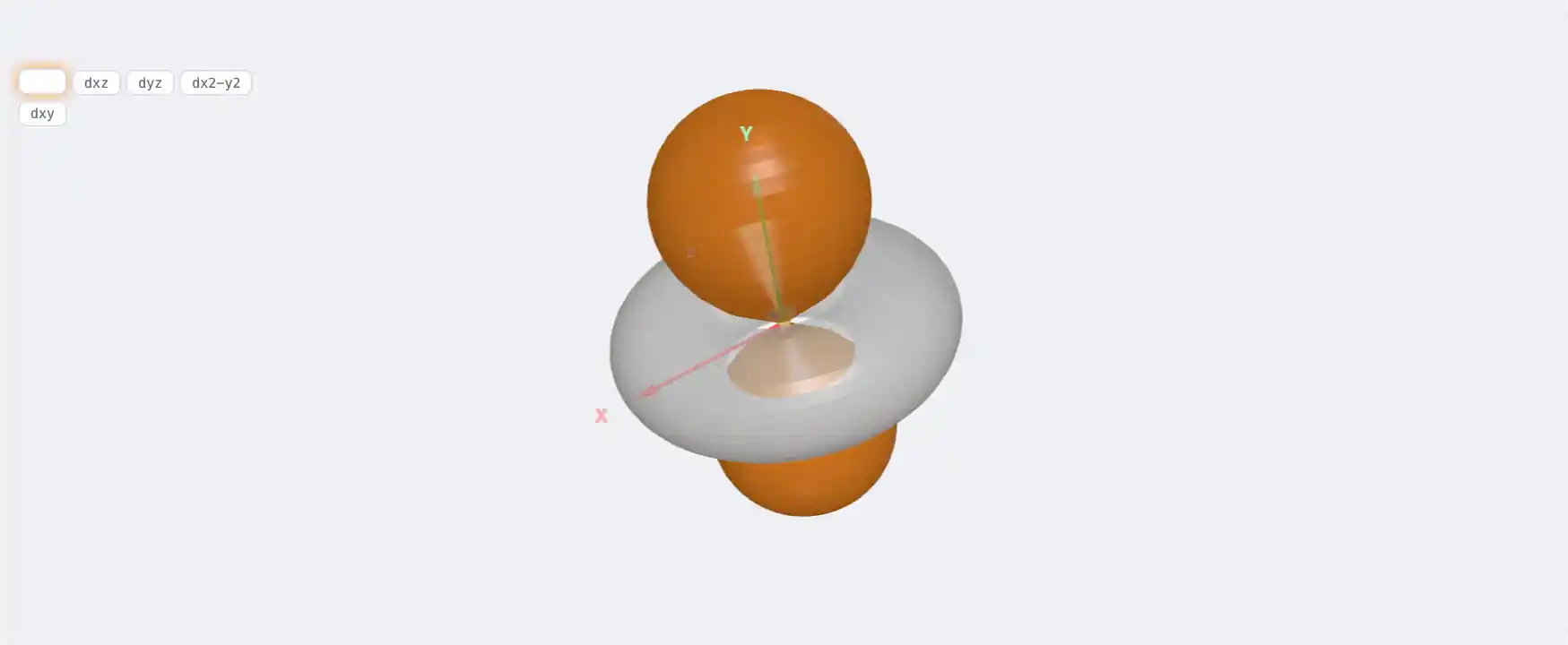
Electronic Structure
Identifiers
Electron Configuration Measured
Kr: 3d¹⁰ 4s² 4p⁶[Ar] 3d¹⁰ 4s² 4p⁶1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 80 Stable | 79.91637808 ± 0.00000075 | 2.2860% | Stable |
| 82 Stable | 81.91348273 ± 0.00000094 | 11.5930% | Stable |
| 83 Stable | 82.91412716 ± 0.00000032 | 11.5000% | Stable |
| 84 Stable | 83.9114977282 ± 0.0000000044 | 56.9870% | Stable |
| 86 Stable | 85.9106106269 ± 0.0000000041 | 17.2790% | Stable |
Phase / State
Reason: 178.4 °C above boiling point (-153.42 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Showing 10 of 36 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Kr I | 0 | 862 | 184 | 862 |
| Kr II | +1 | 1178 | 20 | 1178 |
| Kr III | +2 | 877 | 0 | 877 |
| Kr IV | +3 | 485 | 0 | 485 |
| Kr V | +4 | 174 | 0 | 174 |
| Kr VI | +5 | 142 | 0 | 142 |
| Kr VII | +6 | 73 | 0 | 73 |
| Kr VIII | +7 | 177 | 0 | 177 |
| Kr IX | +8 | 125 | 0 | 125 |
| Kr X | +9 | 46 | 0 | 46 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Kr I | 0 | 528 |
| Kr II | +1 | 163 |
| Kr III | +2 | 123 |
| Kr IV | +3 | 79 |
| Kr V | +4 | 43 |
| Kr VI | +5 | 45 |
| Kr VII | +6 | 28 |
| Kr VIII | +7 | 110 |
| Kr IX | +8 | 58 |
| Kr X | +9 | 36 |
Compounds
Isotopes (5)
Naturally occurring krypton contains six stable isotopes. Seventeen other unstable isotopes are recognized. The spectral lines of krypton are easily produced and some are very sharp. While krypton is generally thought of as a rare gas that normally does not combine with other elements to form compounds, it now appears that the existence of some krypton compounds can exist. Krypton difluoride has been prepared in gram quantities and can be made by several methods. A higher fluoride of krypton and a salt of an oxyacid of krypton also have been reported. Molecule-ions of ArKr+ and KrH+ have been identified and investigated, and evidence is provided for the formation of KrXe or KrXe+.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 80 Stable | 79.91637808 ± 0.00000075 | 2.2860% ± 0.0100% | Stable | stable | |
| 82 Stable | 81.91348273 ± 0.00000094 | 11.5930% ± 0.0310% | Stable | stable | |
| 83 Stable | 82.91412716 ± 0.00000032 | 11.5000% ± 0.0190% | Stable | stable | |
| 84 Stable | 83.9114977282 ± 0.0000000044 | 56.9870% ± 0.0150% | Stable | stable | |
| 86 Stable | 85.9106106269 ± 0.0000000041 | 17.2790% ± 0.0410% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 384.09 nm | N/A | ID 649 | emission | 3s2.3p2 3P → 3s2.3p2 3P | Measured | NIST | |
| 392.9 nm | N/A | Kr VIII | emission | 3d10.8f 2F* → 3d10.9g 2G | Measured | NIST | |
| 392.9 nm | N/A | Kr VIII | emission | 3d10.8f 2F* → 3d10.9g 2G | Measured | NIST | |
| 392.9 nm | N/A | Kr VIII | emission | 3d10.8f 2F* → 3d10.9g 2G | Measured | NIST | |
| 429.9 nm | N/A | Kr VIII | emission | 3d10.8g 2G → 3d10.9h 2H* | Measured | NIST | |
| 429.9 nm | N/A | Kr VIII | emission | 3d10.8g 2G → 3d10.9h 2H* | Measured | NIST | |
| 430 nm | N/A | Kr VIII | emission | 3d10.8g 2G → 3d10.9h 2H* | Measured | NIST | |
| 433.2 nm | N/A | Kr VIII | emission | 3d10.8h 2H* → 3d10.9i 2I | Measured | NIST | |
| 433.2 nm | N/A | Kr VIII | emission | 3d10.8h 2H* → 3d10.9i 2I | Measured | NIST | |
| 433.2 nm | N/A | Kr VIII | emission | 3d10.8h 2H* → 3d10.9i 2I | Measured | NIST | |
| 433.77 nm | N/A | Kr VIII | emission | 3d10.8i 2I → 3d10.9k 2K* | Measured | NIST | |
| 433.77 nm | N/A | Kr VIII | emission | 3d10.8i 2I → 3d10.9k 2K* | Measured | NIST | |
| 433.77 nm | N/A | Kr VIII | emission | 3d10.8i 2I → 3d10.9k 2K* | Measured | NIST | |
| 433.81 nm | N/A | Kr VIII | emission | 3d10.8k 2K* → 3d10.9l 2L | Measured | NIST | |
| 433.81 nm | N/A | Kr VIII | emission | 3d10.8k 2K* → 3d10.9l 2L | Measured | NIST | |
| 433.81 nm | N/A | Kr VIII | emission | 3d10.8k 2K* → 3d10.9l 2L | Measured | NIST | |
| 464 nm | N/A | ID 672 | emission | 1s.5s 3S → 1s.5p 3P* | Measured | NIST | |
| 466.79 nm | N/A | Kr VIII | emission | 3d10.10m 2M* → 3d10.12n 2N | Measured | NIST | |
| 466.79 nm | N/A | Kr VIII | emission | 3d10.10m 2M* → 3d10.12n 2N | Measured | NIST | |
| 466.79 nm | N/A | Kr VIII | emission | 3d10.10m 2M* → 3d10.12n 2N | Measured | NIST | |
| 510 nm | N/A | ID 672 | emission | 1s.4p 3P* → 1s.4d 3D | Measured | NIST | |
| 563 nm | N/A | Kr VIII | emission | 3d10.8p 2P* → 3d10.8d 2D | Measured | NIST | |
| 565.6 nm | N/A | Kr VIII | emission | 3d10.8d 2D → 3d10.9p 2P* | Measured | NIST | |
| 568.6 nm | N/A | Kr VIII | emission | 3d10.8d 2D → 3d10.8f 2F* | Measured | NIST | |
| 572.7 nm | N/A | Kr VIII | emission | 3d10.8d 2D → 3d10.8f 2F* | Measured | NIST | |
| 576.1 nm | N/A | Kr VIII | emission | 3d10.8d 2D → 3d10.9p 2P* | Measured | NIST | |
| 578.5 nm | N/A | Kr VIII | emission | 3d10.7f 2F* → 3d10.8d 2D | Measured | NIST | |
| 580.7 nm | N/A | Kr VIII | emission | 3d10.8p 2P* → 3d10.8d 2D | Measured | NIST | |
| 583.2 nm | N/A | Kr VIII | emission | 3d10.7f 2F* → 3d10.8d 2D | Measured | NIST | |
| 584.9 nm | N/A | Kr VIII | emission | 3d10.8p 2P* → 3d10.8d 2D | Measured | NIST | |
| 605.6 nm | N/A | Kr VIII | emission | 3d10.9k 2K* → 3d10.10l 2L | Measured | NIST | |
| 605.6 nm | N/A | Kr VIII | emission | 3d10.9k 2K* → 3d10.10l 2L | Measured | NIST | |
| 605.6 nm | N/A | Kr VIII | emission | 3d10.9k 2K* → 3d10.10l 2L | Measured | NIST | |
| 606.6 nm | N/A | Kr VIII | emission | 3d10.9l 2L → 3d10.10m 2M* | Measured | NIST | |
| 606.6 nm | N/A | Kr VIII | emission | 3d10.9l 2L → 3d10.10m 2M* | Measured | NIST | |
| 606.6 nm | N/A | Kr VIII | emission | 3d10.9l 2L → 3d10.10m 2M* | Measured | NIST | |
| 637 nm | N/A | ID 647 | emission | 3p6.3d 2D → 3p6.3d 2D | Measured | NIST | |
| 719.57 nm | N/A | ID 674 | emission | 2p 2P* → 2s 2S | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Noble Gas Properties
| HALOGENS | KrF2 |
Phase Transitions & Allotropes
| Melting point | 115.78 K |
| Boiling point | 119.73 K |
| Critical point (temperature) | 209.48 K |
| Critical point (pressure) | 5.53 MPa |
| Triple point (temperature) | 115.77 K |
| Triple point (pressure) | 73.53 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7684 |
| 2 | p | 3.953 |
| 2 | s | 9.602 |
| 3 | d | 15.3741 |
| 3 | p | 15.5658 |
| 3 | s | 14.9673 |
| 4 | p | 26.2308 |
| 4 | s | 24.6844 |
Isotope Decay Modes (55)
| Isotope | Mode | Intensity |
|---|---|---|
| 67 | 2p | 37% |
| 67 | B+ | — |
| 68 | B+ | — |
| 68 | B+p | 90% |
| 68 | p | — |
| 69 | B+ | 100% |
| 69 | B+p | 94% |
| 70 | B+ | 100% |
| 70 | B+p | 1.3% |
| 71 | B+ | 100% |
X‑ray Scattering Factors (509)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0 |
| 10.1617 | — | 0 |
| 10.3261 | — | 0 |
| 10.4931 | — | 0 |
| 10.6628 | — | 0 |
| 10.8353 | — | 0 |
| 11.0106 | — | 0 |
| 11.1886 | — | 0 |
| 11.3696 | — | 0 |
| 11.5535 | — | 0 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1×10-4 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2.1×10-4 milligrams per liter
References (1)
Sources
Sources of this element.
Krypton is present in the air to the extent of about 1 ppm. The atmosphere of Mars has been found to contain 0.3 ppm of krypton. Solid krypton is a white crystalline substance with a face-centered cubic structure which is common to all the "rare gases."
References (1)
- [6] Krypton https://periodic.lanl.gov/36.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
85Kr (with a half-life of 10.7 years) has been used in atmospheric monitoring programs to track the effect of atomic facilities on the surrounding environment. 85Kr is co-generated with plutonium in the fuel elements of nuclear fission reactors and can be monitored at short distances (i.e. 1 to 5 km) from an area of clandestine plutonium separation from spent fuel from the nuclear reactor. The differences in 85Kr levels in the atmosphere have been used to estimate the amount of plutonium separated at weekly intervals. The production of plutonium for nuclear weapons and the output from commercial reprocessing plants have released large amounts of 85Kr into the atmosphere [283] [283] M. B. Kalinowski, H. Sartorius, S. Uhl, W. Weiss. J. Environ. Radioact.73, 203 (2004).[283] M. B. Kalinowski, H. Sartorius, S. Uhl, W. Weiss. J. Environ. Radioact.73, 203 (2004)..
References (2)
- [283] M. B. Kalinowski, H. Sartorius, S. Uhl, W. Weiss. J. Environ. Radioact.73, 203 (2004).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Krypton.
The element property data was retrieved from publications.