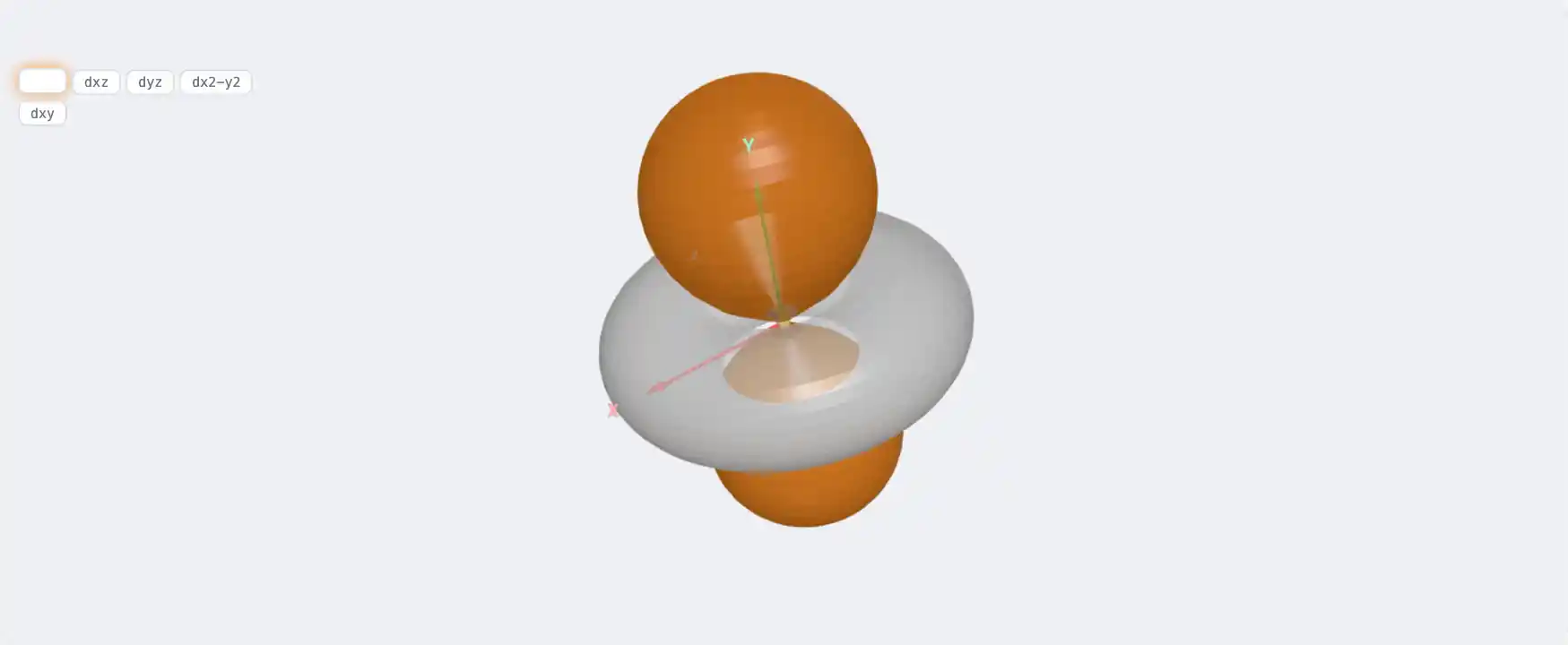
Molybdenum (Mo)
transition-metalSolid
Standard Atomic Weight
95.95 uElectron configuration
[Kr] 5s1 4d5Melting point
2622.85 °C (2896 K)Boiling point
4638.85 °C (4912 K)Density
1.020000e+4 kg/m³Oxidation states
−4, −2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
2.16Ionization energy (1st)
Discovery year
1778Atomic radius
145 pmDetails
Molybdenum is a hard refractory transition metal of group 6. It is notable for its high melting point, useful alloying behavior, and rich redox chemistry. In nature it occurs mainly as molybdenite, and industrially it is important in steels, superalloys, catalysts, and lubricating sulfide materials. In biology, molybdenum is an essential trace element because several enzymes use molybdenum cofactors for oxygen-atom transfer and related redox reactions.
The metal is silvery white, very hard, but is softer and more ductile than tungsten. It has a high elastic modulus, and only tungsten and tantalum, of the more readily available metals, have higher melting points. It is a valuable alloying agent, as it contributes to the hardenability and toughness of quenched and tempered steels. It also improves the strength of steel at high temperatures.
The name derives from the Greek molybdos for "lead". The ancients used the term "lead" for any black mineral that leaves a mark on paper. Molybdenum was discovered by the Swedish pharmacist and chemist Carl Wilhelm Scheele in 1778. It was first isolated by the Swedish chemist Peter-Jacob Hjelm in 1781.
Molybdenum was discovered by Carl Welhelm Scheele, a Swedish chemist, in 1778 in a mineral known as molybdenite (MoS2) which had been confused as a lead compound. Molybdenum was isolated by Peter Jacob Hjelm in 1781. Today, most molybdenum is obtained from molybdenite, wulfenite (PbMoO4) and powellite (CaMoO4). These ores typically occur in conjunction with ores of tin and tungsten. Molybdenum is also obtained as a byproduct of mining and processing tungsten and copper.
From the Greek word molybdo, lead. Before Scheele recognized molybdenite as a distinct ore of a new element in 1778, it was confused with graphite and lead ore. The metal was prepared in impure form in 1782 by Hjelm. Molybdenum does not occur natively, but is obtained principally from molybdenite. Wulfenite, and Powellite are also minor commercial ores.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
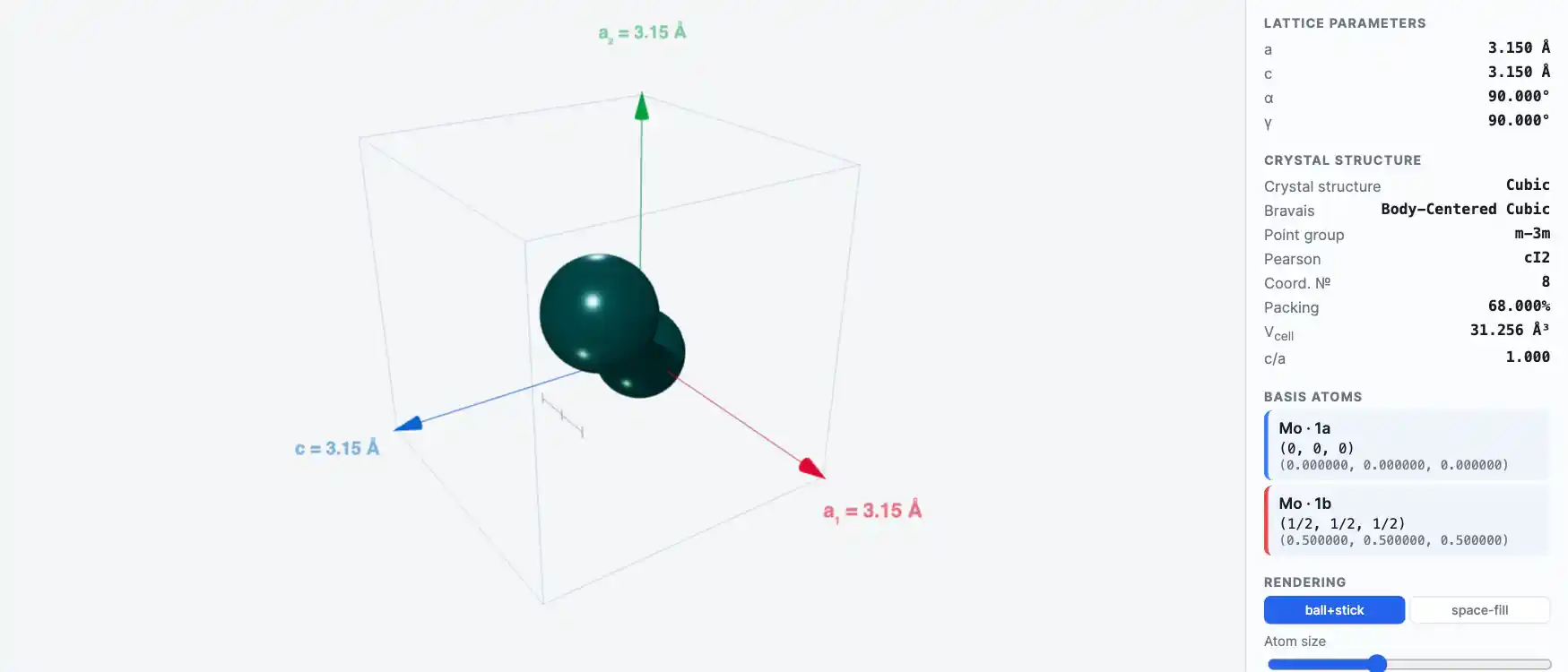
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Mo: 4d⁵ 5s¹[Kr] 4d⁵ 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d⁵ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 94 Stable | 93.9050849 ± 0.00000048 | 9.1500% | Stable |
| 95 Stable | 94.90583877 ± 0.00000047 | 15.8400% | Stable |
| 96 Stable | 95.90467612 ± 0.00000047 | 16.6700% | Stable |
| 97 Stable | 96.90601812 ± 0.00000049 | 9.6000% | Stable |
Phase / State
Reason: 2597.8 °C below melting point (2622.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 42 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Mo I | 0 | 818 | 721 | 808 |
| Mo II | +1 | 209 | 0 | 0 |
| Mo III | +2 | 62 | 0 | 0 |
| Mo IV | +3 | 29 | 0 | 0 |
| Mo V | +4 | 966 | 923 | 929 |
| Mo VI | +5 | 245 | 245 | 245 |
| Mo VII | +6 | 413 | 0 | 413 |
| Mo VIII | +7 | 109 | 0 | 109 |
| Mo IX | +8 | 231 | 0 | 231 |
| Mo X | +9 | 120 | 0 | 120 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Mo I | 0 | 428 |
| Mo II | +1 | 249 |
| Mo III | +2 | 120 |
| Mo IV | +3 | 81 |
| Mo V | +4 | 258 |
| Mo VI | +5 | 113 |
| Mo VII | +6 | 96 |
| Mo VIII | +7 | 77 |
| Mo IX | +8 | 93 |
| Mo X | +9 | 48 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 69 pm |
| +4 | 6 | N/A | 65 pm |
| +5 | 4 | N/A | 46 pm |
| +5 | 6 | N/A | 61 pm |
| +6 | 4 | N/A | 41 pm |
| +6 | 5 | N/A | 50 pm |
| +6 | 6 | N/A | 59 pm |
| +6 | 7 | N/A | 73 pm |
Compounds
Isotopes (4)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 94 Stable | 93.9050849 ± 0.00000048 | 9.1500% ± 0.0900% | Stable | stable | |
| 95 Stable | 94.90583877 ± 0.00000047 | 15.8400% ± 0.1100% | Stable | stable | |
| 96 Stable | 95.90467612 ± 0.00000047 | 16.6700% ± 0.1500% | Stable | stable | |
| 97 Stable | 96.90601812 ± 0.00000049 | 9.6000% ± 0.1400% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 382.2548 nm | 290 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3P* | Measured | NIST | |
| 383.9084 nm | 360 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3P* | Measured | NIST | |
| 386 nm | N/A | ID 915 | emission | 1s.5s 3S → 1s.5p 3P* | Measured | NIST | |
| 393.8911 nm | 1400 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3P* | Measured | NIST | |
| 394.8336 nm | 50 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 1F* | Measured | NIST | |
| 400.9437 nm | 35 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 1D* | Measured | NIST | |
| 403.6485 nm | 40 | Mo VI | emission | 4p6.7f 2F* → 4p6.8g 2G | Measured | NIST | |
| 405.4556 nm | 50 | Mo VI | emission | 4p6.7f 2F* → 4p6.8g 2G | Measured | NIST | |
| 406.1547 nm | 210 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3P* | Measured | NIST | |
| 406.2019 nm | 15000 | Mo VI | emission | 4p6.7p 2P* → 4p6.7d 2D | Measured | NIST | |
| 406.4706 nm | 14 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 1D* | Measured | NIST | |
| 406.527 nm | 3500 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3F* | Measured | NIST | |
| 407.1568 nm | 2800 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 1F* | Measured | NIST | |
| 407.4773 nm | 3100 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3F* | Measured | NIST | |
| 416.4901 nm | 75 | Mo VI | emission | 4p6.6g 2G → 4p6.7f 2F* | Measured | NIST | |
| 418.4284 nm | 60 | Mo VI | emission | 4p6.6g 2G → 4p6.7f 2F* | Measured | NIST | |
| 418.6616 nm | 2700 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3D* | Measured | NIST | |
| 422.59 nm | N/A | ID 896 | emission | 2p 2P* → 2s 2S | Measured | NIST | |
| 423.2026 nm | 40000 | Mo VI | emission | 4p6.7p 2P* → 4p6.7d 2D | Measured | NIST | |
| 427.2928 nm | 100 | Mo VI | emission | 4p6.7p 2P* → 4p6.7d 2D | Measured | NIST | |
| 433.4926 nm | 840 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3D* | Measured | NIST | |
| 436 nm | N/A | ID 915 | emission | 1s.4p 3P* → 1s.4d 3D | Measured | NIST | |
| 438.442 nm | 2900 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3D* | Measured | NIST | |
| 439.9605 nm | 28 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3D* | Measured | NIST | |
| 446.6307 nm | 79 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 1D* | Measured | NIST | |
| 447.4143 nm | 63 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3F* | Measured | NIST | |
| 454.3076 nm | 570 | Mo V | emission | 4p6.4d.(2D<3/2>).6s 2[3/2] → 4p6.4d.6p 3F* | Measured | NIST | |
| 462.464 nm | 840 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 1D* | Measured | NIST | |
| 463.7675 nm | 41 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3F* | Measured | NIST | |
| 466.0971 nm | 100 | Mo VI | emission | 4p6.5f 2F* → 4p6.6d 2D | Measured | NIST | |
| 468.7277 nm | 22 | Mo V | emission | 4p6.4d.(2D<5/2>).6s 2[5/2] → 4p6.4d.6p 3D* | Measured | NIST | |
| 474.6519 nm | 8000 | Mo VI | emission | 4p6.5f 2F* → 4p6.6d 2D | Measured | NIST | |
| 504.622 nm | N/A | Mo VI | emission | 4p6.7g 2G → 4p6.8h 2H* | Measured | NIST | |
| 504.622 nm | N/A | Mo VI | emission | 4p6.7g 2G → 4p6.8h 2H* | Measured | NIST | |
| 524.749 nm | N/A | Mo VI | emission | 4p6.7h 2H* → 4p6.8i 2I | Measured | NIST | |
| 524.749 nm | N/A | Mo VI | emission | 4p6.7h 2H* → 4p6.8i 2I | Measured | NIST | |
| 527.675 nm | N/A | Mo VI | emission | 4p6.7i 2I → 4p6.8k 2K* | Measured | NIST | |
| 527.675 nm | N/A | Mo VI | emission | 4p6.7i 2I → 4p6.8k 2K* | Measured | NIST | |
| 558.5 nm | 200 | Mo VI | emission | 4p6.8d 2D → 4p6.8f 2F* | Measured | NIST | |
| 562 nm | 350 | Mo VI | emission | 4p6.8d 2D → 4p6.8f 2F* | Measured | NIST | |
| 587.138 nm | 300 | Mo VI | emission | 4p6.7d 2D → 4p6.8p 2P* | Measured | NIST | |
| 603.562 nm | 10 | Mo VI | emission | 4p6.4f 2F* → 4p6.5d 2D | Measured | NIST | |
| 618.867 nm | 1400 | Mo VI | emission | 4p6.4f 2F* → 4p6.5d 2D | Measured | NIST | |
| 633.604 nm | 1000 | Mo VI | emission | 4p6.4f 2F* → 4p6.5d 2D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2895.15 K |
| Boiling point | 4912.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.8744 |
| 2 | p | 4.0282 |
| 2 | s | 11.1232 |
| 3 | d | 14.7717 |
| 3 | p | 16.5264 |
| 3 | s | 16.0185 |
| 4 | d | 30.6076 |
| 4 | p | 27.0232 |
| 4 | s | 25.9036 |
| 5 | s | 35.894 |
Crystal Radii Detail (8)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 83 | estimated, | |
| 4 | VI | 79 | from r^3 vs V plots, from metallic oxides, | |
| 5 | IV | 60 | from r^3 vs V plots, | |
| 5 | VI | 75 | from r^3 vs V plots, | |
| 6 | IV | 55 | from r^3 vs V plots, | |
| 6 | V | 64 | ||
| 6 | VI | 73 | from r^3 vs V plots, | |
| 6 | VII | 87 |
Isotope Decay Modes (58)
| Isotope | Mode | Intensity |
|---|---|---|
| 81 | B+ | — |
| 81 | B+p | — |
| 82 | B+ | — |
| 82 | B+p | — |
| 83 | B+ | 100% |
| 83 | B+p | — |
| 84 | B+ | 100% |
| 84 | B+p | — |
| 85 | B+ | 100% |
| 85 | B+p | 0.1% |
X‑ray Scattering Factors (909)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.2382 |
| 10.1447 | — | 2.20464 |
| 10.3088 | — | 2.17288 |
| 10.4756 | — | 2.14408 |
| 10.645 | — | 2.11566 |
| 10.8172 | — | 2.09307 |
| 10.9921 | — | 2.12057 |
| 11.1699 | — | 2.20711 |
| 11.3506 | — | 2.32651 |
| 11.5342 | — | 2.50051 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.2 milligrams per kilogram
References (1)
- [5] Molybdenum https://education.jlab.org/itselemental/ele042.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1×10-2 milligrams per liter
References (1)
- [5] Molybdenum https://education.jlab.org/itselemental/ele042.html
Sources
Sources of this element.
Molybdenum is also recovered as a by-product of copper and tungsten mining operations. The metal is prepared from the powder made by the hydrogen reduction of purified molybdic trioxide or ammonium molybdate.
References (1)
- [6] Molybdenum https://periodic.lanl.gov/42.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Molybdenum.
The element property data was retrieved from publications.