Technetium (Tc)
transition-metalSolid
Standard Atomic Weight
[98]Electron configuration
[Kr] 5s2 4d5Melting point
2156.85 °C (2430 K)Boiling point
4264.85 °C (4538 K)Density
1.100000e+4 kg/m³Oxidation states
−3, −1, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
1.9Ionization energy (1st)
Discovery year
1937Atomic radius
135 pmDetails
Technetium is a radioactive transition metal in group 7, between molybdenum and ruthenium. It was the first element discovered without a stable isotope. Only trace natural technetium occurs, mainly from spontaneous fission of uranium and from neutron capture processes; practical quantities are made artificially. Its chemistry resembles rhenium and manganese in several oxidation states, and the isotope ⁹⁹ᵐTc is central to diagnostic nuclear medicine.
Technetium is a silvery-gray metal that tarnishes slowly in moist air. The common oxidation states of technetium are +7, +5, and +4. Under oxidizing conditions technetium (VII) will exist as the pertechnetate ion, TcO4-. The chemistry of technetium is said to be similar to that of rhenium. Technetium dissolves in nitric acid, aqua regia, and concentrated sulfuric acid, but is not soluble in hydrochloric acid of any strength. The element is a remarkable corrosion inhibitor for steel. The metal is an excellent superconductor at 11K and below.
Technetium was the first artificially produced element. It was isolated by Carlo Perrier and Emilio Segrè in 1937. Technetium was created by bombarding molybdenum atoms with deuterons that had been accelerated by a device called a cyclotron. Today, technetium is produced by bombarding molybdenum-98 with neutrons. Molybdenum-98 becomes molybdenum-99 when it captures a neutron. Molybdenum-99, with a half-life of 65.94 hours, decays into technetium-99 through beta decay. While technetium has never been found to occur naturally on earth, its spectral lines have been observed in S-, M- and N-type stars.
Technetium's most stable isotope, technetium-98, has a half-life of about 4,200,000 years. It decays into ruthenium-98 through beta decay.
From the Greek word technetos, artificial. Element 43 was predicted on the basis of the periodic table, and was erroneously reported as having been discovered in 1925, at which time it was named masurium. The element was actually discovered by Perrier and Segre in Italy in 1937. It was also found in a sample of molybdenum sent by E. Lawrence that was bombarded by deuterons in the Berkeley cyclotron. Technetium was the first element to be produced artificially. Since its discovery, searches for the element in terrestrial material have been made. Finally in 1962, technetium-99 was isolated and identified in African pitchblende (a uranium rich ore) in extremely minute quantities as a spontaneous fission product of uranium-238 by B.T. Kenna and P.K. Kuroda. If it does exist, the concentration must be very small. Technetium has been found in the spectrum of S-, M-, and N-type stars, and its presence in stellar matter is leading to new theories of the production of heavy elements in the stars.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
N/A
Reactivity
N/A
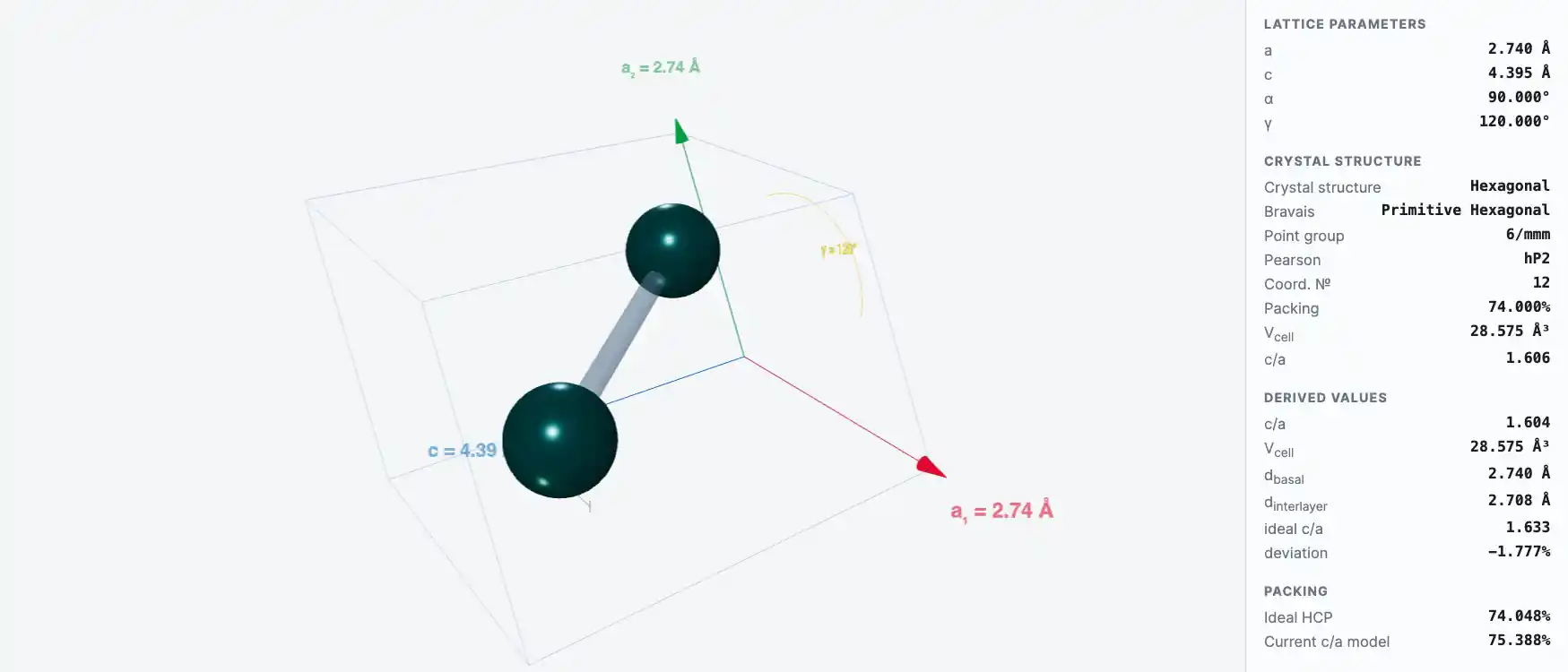
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Tc: 4d⁵ 5s²[Kr] 4d⁵ 5s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d⁵ 5s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 109 Radioactive | 108.920256 ± 0.00001 | N/A | 905 ms |
| 110 Radioactive | 109.923744 ± 0.00001 | N/A | 900 ms |
| 111 Radioactive | 110.925901 ± 0.000011 | N/A | 350 ms |
| 112 Radioactive | 111.9299458 ± 0.000006 | N/A | 323 ms |
| 94 Radioactive | 93.9096536 ± 0.0000044 | N/A | 293 minutes |
Phase / State
Reason: 2131.8 °C below melting point (2156.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 43 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Tc I | 0 | 600 | 13 | 561 |
| Tc II | +1 | 40 | 6 | 23 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Tc I | 0 | 290 |
| Tc II | +1 | 34 |
| Tc III | +2 | 2 |
| Tc IV | +3 | 2 |
| Tc V | +4 | 2 |
| Tc VI | +5 | 2 |
| Tc VII | +6 | 2 |
| Tc VIII | +7 | 2 |
| Tc IX | +8 | 2 |
| Tc X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 6 | N/A | 64.5 pm |
| +5 | 6 | N/A | 60 pm |
| +7 | 4 | N/A | 37 pm |
| +7 | 6 | N/A | 56.00000000000001 pm |
Compounds
Isotopes (5)
Twenty-two isotopes of technetium with masses ranging from 90 to 111 are reported. All the isotopes of technetium are radioactive. It is one of two elements with Z < 83 that have no stable isotopes; the other element is promethium (Z = 61). Technetium has three long lived radioactive isotopes: 97Tc (T1/2 = 2.6 x 106 years), 98Tc (T1/2 = 4.2 x 106 years) and 99Tc (T1/2 = 2.1 x 105 years). 95Tcm ("m" stands for meta state) (T1/2 = 61 days) is used in tracer work. However, the most useful isotope of technetium is 99Tcm (T1/2 = 6.01 hours) is used in many medical radioactive isotope tests because of its half-life being short, the energy of the gamma ray it emits, and the ability of technetium to be chemically bound to many biologically active molecules. Because 99Tc is produced as a fission product from the fission of uranium in nuclear reactors, large quantities have been produced over the years. There are kilogram quantities of technetium currently existing.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 109 Radioactive | 108.920256 ± 0.00001 | N/A | 905 ms | β- =100%β-n =0.08±0.2% | |
| 110 Radioactive | 109.923744 ± 0.00001 | N/A | 900 ms | β- =100%β-n =0.04±0.2% | |
| 111 Radioactive | 110.925901 ± 0.000011 | N/A | 350 ms | β- =100%β-n =0.85±2% | |
| 112 Radioactive | 111.9299458 ± 0.000006 | N/A | 323 ms | β- =100%β-n =1.5±0.2% | |
| 94 Radioactive | 93.9096536 ± 0.0000044 | N/A | 293 minutes | β+ =100% |
Spectral Lines
Showing 50 of 277 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 485.359 nm | 20000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 409.5662 nm | 15000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 408.8702 nm | 10000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 411.5065 nm | 10000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 416.5605 nm | 10000 | Tc I | emission | 4d6.(5D).5s 4D → 4d5.(6S).5s.(5S).5p 4P* | Measured | NIST | |
| 426.2245 nm | 10000 | Tc I | emission | 4d5.5s2 6S → 4d5.(6S).5s.(7S).5p 6P* | Measured | NIST | |
| 429.7034 nm | 10000 | Tc I | emission | 4d5.5s2 6S → 4d5.(6S).5s.(7S).5p 6P* | Measured | NIST | |
| 452.283 nm | 10000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 474.0602 nm | 10000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 482.0744 nm | 10000 | Tc I | emission | 4d5.(6S).5s.(7S).5p 8P* → 4d5.(6S).5s.(7S).6s e 8S | Measured | NIST | |
| 486.6732 nm | 10000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 412.4217 nm | 8000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 489.1909 nm | 8000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 414.4961 nm | 6000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 417.2523 nm | 5000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 497.6341 nm | 5000 | Tc I | emission | 4d5.(6S).5s.(7S).5p 8P* → 4d5.(6S).5s.(7S).6s e 8S | Measured | NIST | |
| 509.6269 nm | 5000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 6F* | Measured | NIST | |
| 417.0266 nm | 4000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 477.1539 nm | 4000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 414.5126 nm | 3000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 448.7049 nm | 3000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 463.7499 nm | 3000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 394.709 nm | 2000 | Tc I | emission | 4d6.(3H).5s 4H → 4d6.(3H).5p 2I* | Measured | NIST | |
| 399.4498 nm | 2000 | Tc I | emission | 4d6.(3H).5s 4H → 4d6.(3H).5p 4G* | Measured | NIST | |
| 402.0759 nm | 2000 | Tc I | emission | 4d6.(3H).5s 2H → 4d6.(3H).5p 2H* | Measured | NIST | |
| 453.9513 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 456.4541 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 464.8328 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 466.9303 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4P* | Measured | NIST | |
| 471.7758 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 490.9509 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4F* | Measured | NIST | |
| 517.4813 nm | 2000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 6F* | Measured | NIST | |
| 383.7565 nm | 1500 | Tc I | emission | 4d6.(3H).5s 4H → 4d6.(3H).5p 4I* | Measured | NIST | |
| 564.2116 nm | 1500 | Tc I | emission | 4d6.(3F2).5s 4F → 4d6.(5D).5p 4D* | Measured | NIST | |
| 386.8248 nm | 1000 | Tc I | emission | 4d6.(3H).5s 4H → 4d6.(3H).5p 4I* | Measured | NIST | |
| 401.1998 nm | 1000 | Tc I | emission | 4d6.(3H).5s 4H → 4d5.(4D).5s.(5D).5p 6F* | Measured | NIST | |
| 403.9232 nm | 1000 | Tc I | emission | 4d6.(3H).5s 4H → 4d6.(3H).5p 4G* | Measured | NIST | |
| 411.0214 nm | 1000 | Tc I | emission | 4d6.(1I).5s 2I → 4d6.(1I).5p 2K* | Measured | NIST | |
| 412.8263 nm | 1000 | Tc I | emission | 4d5.5s2 4G → 4d5.(4G).5s.(5G).5p 4H* | Measured | NIST | |
| 416.966 nm | 1000 | Tc I | emission | 4d6.(3H).5s 4H → 4d5.(4G).5s.(5G).5p 4H* | Measured | NIST | |
| 417.6253 nm | 1000 | Tc I | emission | 4d6.(5D).5s 6D → 4d6.(5D).5p 6D* | Measured | NIST | |
| 426.2682 nm | 1000 | Tc I | emission | 4d6.(5D).5s 4D → 4d5.(6S).5s.(5S).5p 4P* | Measured | NIST | |
| 442.9581 nm | 1000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 448.1534 nm | 1000 | Tc I | emission | 4d6.(3H).5s 2H → 4d6.(3H).5p 4H* | Measured | NIST | |
| 451.5974 nm | 1000 | Tc I | emission | 4d6.(3H).5s 2H → 4d6.(3H).5p 4H* | Measured | NIST | |
| 455.7038 nm | 1000 | Tc I | emission | 4d6.(3F2).5s 4F → 4d6.(3F2).5p 4G* | Measured | NIST | |
| 457.8438 nm | 1000 | Tc I | emission | 4d6.(3G).5s 4G → 4d6.(3H).5p 2I* | Measured | NIST | |
| 459.3334 nm | 1000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST | |
| 461.6842 nm | 1000 | Tc I | emission | 4d5.(6S).5s.(7S).5p 6P* → 4d5.(6S).5s.(7S).5d f 6D | Measured | NIST | |
| 463.0527 nm | 1000 | Tc I | emission | 4d6.(5D).5s 4D → 4d6.(5D).5p 4D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 2430.15 K |
| Boiling point | 4535.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.891 |
| 2 | p | 4.0592 |
| 2 | s | 11.3718 |
| 3 | d | 14.647 |
| 3 | p | 16.6159 |
| 3 | s | 16.2088 |
| 4 | d | 30.118 |
| 4 | p | 27.1888 |
| 4 | s | 25.8016 |
| 5 | s | 35.7735 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | VI | 78.5 | from r^3 vs V plots, from metallic oxides, | |
| 5 | VI | 74 | estimated, from r^3 vs V plots, | |
| 7 | IV | 51 | ||
| 7 | VI | 70 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (70)
| Isotope | Mode | Intensity |
|---|---|---|
| 83 | p | — |
| 83 | B+ | — |
| 83 | B+p | — |
| 84 | p | — |
| 84 | B+ | — |
| 84 | B+p | — |
| 85 | p | — |
| 86 | B+ | 100% |
| 86 | B+p | — |
| 87 | B+ | 100% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.1689 |
| 10.1617 | — | 1.2263 |
| 10.3261 | — | 1.28651 |
| 10.4931 | — | 1.34968 |
| 10.6628 | — | 1.41595 |
| 10.8353 | — | 1.48547 |
| 11.0106 | — | 1.55841 |
| 11.1886 | — | 1.63493 |
| 11.3696 | — | 1.7152 |
| 11.5535 | — | 1.7906 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
Not Applicable
References (1)
- [5] Technetium https://education.jlab.org/itselemental/ele043.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Technetium https://education.jlab.org/itselemental/ele043.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Technetium.
The element property data was retrieved from publications.