Palladium (Pd)
transition-metalSolid
Standard Atomic Weight
106.42 uElectron configuration
[Kr] 4d10Melting point
1554.9 °C (1828.05 K)Boiling point
2962.85 °C (3236 K)Density
1.200000e+4 kg/m³Oxidation states
+1, +2, +3, +4, +5Electronegativity (Pauling)
2.2Ionization energy (1st)
Discovery year
1803Atomic radius
140 pmDetails
Palladium is a silvery platinum-group metal with high catalytic activity and an unusual ability to absorb large amounts of hydrogen into its lattice. It is chemically noble in air at ordinary temperatures, but it forms many complexes and is readily used in surface chemistry. Its technological importance is dominated by vehicle emission control, fine-chemical catalysis, electronics, and hydrogen-related applications.
The element is a silvery-white metal, it does not tarnish in air, and it is the least dense and lowest melting of the platinum group of metals. When annealed, it is soft and ductile; cold-working greatly increases its strength and hardness. Palladium is attacked by nitric and sulfuric acid.
At room temperatures, the metal has the unusual property of absorbing up to 900 times its own volume of hydrogen, possibly forming Pd2H. It is not yet clear if this is a true compound. Hydrogen readily diffuses through heated palladium, providing a means of purifying the gas.
The name derives from the second largest asteroid of the solar system Pallas (named after the goddess of wisdom and arts—Pallas Athene). The element was discovered by the English chemist and physicist William Hyde Wollaston in 1803, one year after the discovery of Pallas by the German astronomer Wilhelm Olbers in 1802. The discovery was originally published anonymously by Wollaston to obtain priority, while not disclosing any details about his preparation.
Palladium was discovered by William Hyde Wollaston, an English chemist, in 1803 while analyzing samples of platinum ore that were obtained from South America. Although it is a rare element, palladium tends to occur along with deposits of platinum, nickel, copper, silver and gold and is recovered as a byproduct of mining these other metals.
Palladium was named after the asteroid Pallas, which was discovered at about the same time. Pallas was the Greek goddess of wisdom.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
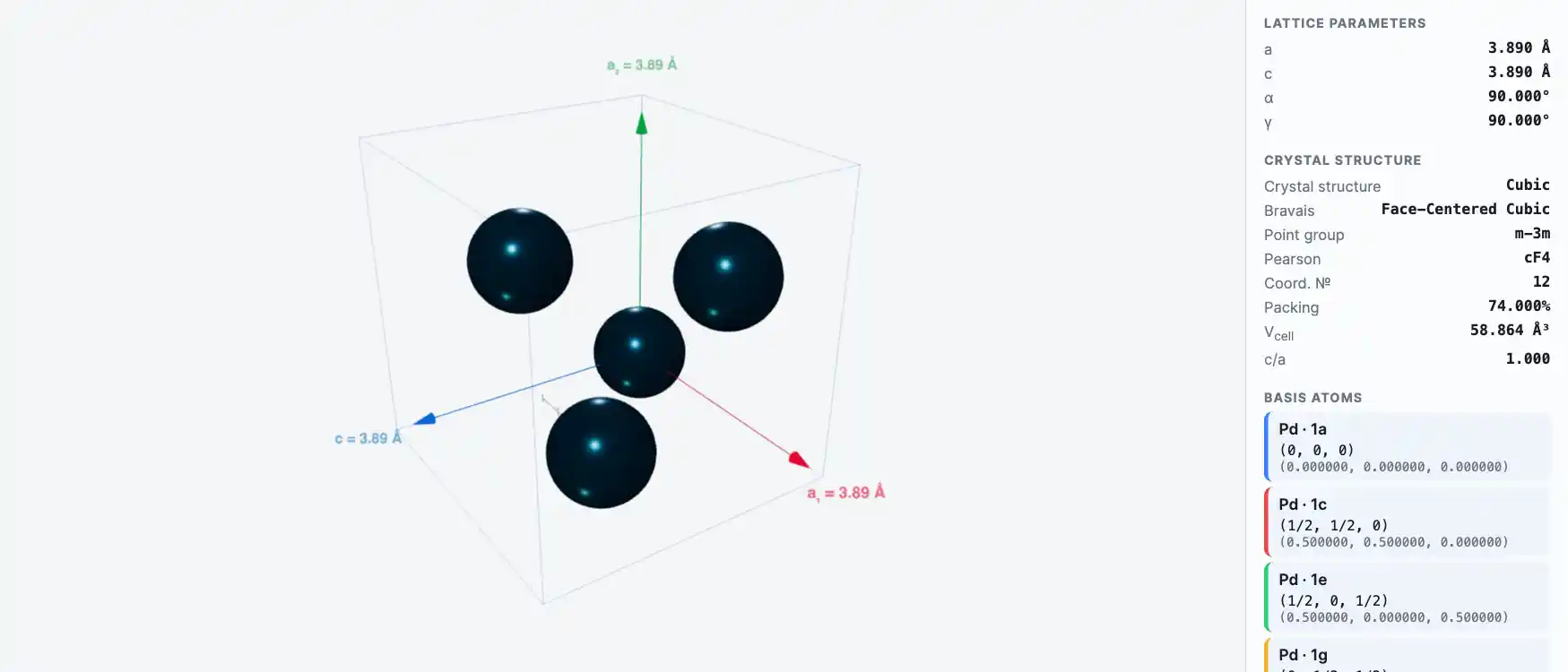
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Pd: 4d¹⁰[Kr] 4d¹⁰1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 104 Stable | 103.9040305 ± 0.0000014 | 11.1400% | Stable |
| 105 Stable | 104.9050796 ± 0.0000012 | 22.3300% | Stable |
| 106 Stable | 105.9034804 ± 0.0000012 | 27.3300% | Stable |
| 108 Stable | 107.9038916 ± 0.0000012 | 26.4600% | Stable |
Phase / State
Reason: 1529.9 °C below melting point (1554.9 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 46 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Pd I | 0 | 76 | 8 | 75 |
| Pd II | +1 | 62 | 10 | 62 |
| Pd III | +2 | 75 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Pd I | 0 | 145 |
| Pd II | +1 | 186 |
| Pd III | +2 | 177 |
| Pd IV | +3 | 2 |
| Pd V | +4 | 2 |
| Pd VI | +5 | 2 |
| Pd VII | +6 | 2 |
| Pd VIII | +7 | 2 |
| Pd IX | +8 | 2 |
| Pd X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 2 | N/A | 59 pm |
| +2 | 4 | N/A | 64 pm |
| +2 | 6 | N/A | 86 pm |
| +3 | 6 | N/A | 76 pm |
| +4 | 6 | N/A | 61.5 pm |
Compounds
Isotopes (4)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 104 Stable | 103.9040305 ± 0.0000014 | 11.1400% ± 0.0800% | Stable | stable | |
| 105 Stable | 104.9050796 ± 0.0000012 | 22.3300% ± 0.0800% | Stable | stable | |
| 106 Stable | 105.9034804 ± 0.0000012 | 27.3300% ± 0.0300% | Stable | stable | |
| 108 Stable | 107.9038916 ± 0.0000012 | 26.4600% ± 0.0900% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 383.2286 nm | 1500 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[3/2]* | Measured | NIST | |
| 389.41982 nm | 2200 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[5/2]* | Measured | NIST | |
| 395.86229 nm | 1500 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[5/2]* | Measured | NIST | |
| 408.73428 nm | 290 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[3/2]* | Measured | NIST | |
| 416.98387 nm | 90 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[3/2]* | Measured | NIST | |
| 421.29533 nm | 2500 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[7/2]* | Measured | NIST | |
| 447.35846 nm | 180 | Pd I | emission | 4d9.(2D<3/2>).5s 2[3/2] → 4d9.(2D<5/2>).5p 2[3/2]* | Measured | NIST | |
| 478.81874 nm | N/A | Pd I | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 481.75067 nm | N/A | Pd I | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d9.(2D<5/2>).5d 2[3/2] | Measured | NIST | |
| 487.54251 nm | 35 | Pd I | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d9.(2D<5/2>).5d 2[1/2] | Measured | NIST | |
| 511.08092 nm | 55 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).5d 2[7/2] | Measured | NIST | |
| 511.70072 nm | 75 | Pd I | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d9.(2D<3/2>).5d 2[7/2] | Measured | NIST | |
| 516.38405 nm | 160 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).5d 2[9/2] | Measured | NIST | |
| 523.48612 nm | 55 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).5d 2[7/2] | Measured | NIST | |
| 529.56266 nm | 120 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).5d 2[9/2] | Measured | NIST | |
| 531.25867 nm | 18 | Pd I | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 534.51048 nm | 15 | Pd I | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d9.(2D<3/2>).5d 2[5/2] | Measured | NIST | |
| 539.52204 nm | 35 | Pd I | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d9.(2D<3/2>).5d 2[7/2] | Measured | NIST | |
| 554.28067 nm | 55 | Pd I | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d9.(2D<5/2>).5d 2[7/2] | Measured | NIST | |
| 554.70204 nm | 35 | Pd I | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 561.94631 nm | 27 | Pd I | emission | 4d9.(2D<3/2>).5p 2[3/2]* → 4d9.(2D<3/2>).5d 2[5/2] | Measured | NIST | |
| 564.27039 nm | 15 | Pd I | emission | 4d9.(2D<3/2>).5p 2[1/2]* → 4d9.(2D<3/2>).5d 2[3/2] | Measured | NIST | |
| 565.54366 nm | 14 | Pd I | emission | 4d9.(2D<3/2>).5p 2[3/2]* → 4d9.(2D<3/2>).5d 2[5/2] | Measured | NIST | |
| 567.00702 nm | 75 | Pd I | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d9.(2D<5/2>).5d 2[7/2] | Measured | NIST | |
| 569.0128 nm | 11 | Pd I | emission | 4d9.(2D<3/2>).5p 2[1/2]* → 4d9.(2D<3/2>).5d 2[3/2] | Measured | NIST | |
| 569.50921 nm | N/A | Pd I | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 573.66175 nm | 18 | Pd I | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d9.(2D<5/2>).5d 2[3/2] | Measured | NIST | |
| 677.45174 nm | 23 | Pd I | emission | 4d8.5s2 3F → 4d9.(2D<3/2>).5p 2[5/2]* | Measured | NIST | |
| 678.44893 nm | 65 | Pd I | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d9.(2D<5/2>).6s 2[5/2] | Measured | NIST | |
| 683.3446 nm | N/A | Pd I | emission | 4d9.(2D<3/2>).5p 2[3/2]* → 4d9.(2D<5/2>).5d 2[5/2] | Measured | NIST | |
| 701.6446 nm | 11 | Pd I | emission | 4d9.(2D<3/2>).5p 2[1/2]* → 4d9.(2D<3/2>).6s 2[3/2] | Measured | NIST | |
| 731.0053 nm | N/A | Pd I | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d9.(2D<3/2>).6s 2[3/2] | Measured | NIST | |
| 736.8096 nm | 75 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).6s 2[5/2] | Measured | NIST | |
| 739.19 nm | 27 | Pd I | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d9.(2D<3/2>).6s 2[3/2] | Measured | NIST | |
| 748.6909 nm | 16 | Pd I | emission | 4d9.(2D<5/2>).5p 2[7/2]* → 4d9.(2D<5/2>).6s 2[5/2] | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1827.95 K |
| Boiling point | 3236.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (9)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.9411 |
| 2 | p | 4.07 |
| 2 | s | 12.1172 |
| 3 | d | 14.5489 |
| 3 | p | 16.9804 |
| 3 | s | 16.7788 |
| 4 | d | 32.3824 |
| 4 | p | 28.2768 |
| 4 | s | 27.014 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | II | 73 | ||
| 2 | IVSQ | 78 | ||
| 2 | VI | 100 | ||
| 3 | VI | 90 | ||
| 4 | VI | 75.5 | from r^3 vs V plots, |
Isotope Decay Modes (62)
| Isotope | Mode | Intensity |
|---|---|---|
| 90 | B+ | — |
| 90 | B+p | — |
| 90 | 2p | — |
| 91 | B+ | 100% |
| 91 | B+p | 3.1% |
| 92 | B+ | 100% |
| 92 | B+p | 1.6% |
| 93 | B+ | 100% |
| 93 | B+p | 7.4% |
| 94 | B+ | 100% |
X‑ray Scattering Factors (507)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.15058 |
| 10.1617 | — | 1.2095 |
| 10.3261 | — | 1.27144 |
| 10.4931 | — | 1.33655 |
| 10.6628 | — | 1.40499 |
| 10.8353 | — | 1.47694 |
| 11.0106 | — | 1.54695 |
| 11.1886 | — | 1.61473 |
| 11.3696 | — | 1.68548 |
| 11.5535 | — | 1.75934 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.5×10-2 milligrams per kilogram
References (1)
- [5] Palladium https://education.jlab.org/itselemental/ele046.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Palladium https://education.jlab.org/itselemental/ele046.html
Sources
Sources of this element.
Discovered in 1803 by Wollaston, Palladium is found with platinum and other metals of the platinum group in placer deposits of Russia, South America, North America, Ethiopia, and Australia. It is also found associated with the nickel-copper deposits of South Africa and Ontario. Palladium's separation from the platinum metals depends upon the type of ore in which it is found.
References (1)
- [6] Palladium https://periodic.lanl.gov/46.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Palladium.
The element property data was retrieved from publications.