Silver (Ag)
transition-metalSolid
Standard Atomic Weight
107.8682 uElectron configuration
[Kr] 5s1 4d10Melting point
961.78 °C (1234.93 K)Boiling point
2161.85 °C (2435 K)Density
1.050100e+4 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3Electronegativity (Pauling)
1.93Ionization energy (1st)
Discovery year
N/AAtomic radius
160 pmDetails
Silver is a soft, dense transition metal in group 11, chemically related to copper and gold but more reactive than gold. It is the best elemental conductor of electricity and heat at ordinary temperatures and has exceptional optical reflectivity when freshly polished. In nature it occurs as native metal and in sulfide, sulfosalt, chloride, and telluride minerals, commonly associated with lead, zinc, copper, and gold ores.
Pure silver has a brilliant white metallic luster. It is a little harder than gold and is very ductile and malleable, being exceeded only by gold and perhaps palladium. Pure silver has the highest electrical and thermal conductivity of all metals, and possesses the lowest contact resistance. It is stable in pure air and water, but tarnishes when exposed to ozone, hydrogen sulfide, or air containing sulfur. The alloys of silver are important.
The name derives from the Anglo-Saxon seofor and siolfur, which is of unknown origin. The symbol Ag derives from the Latin argentum and Sanskrit argunas from "bright". Silver was known in prehistoric times.
Archaeological evidence suggests that people have been using silver for at least 5000 years. Silver can be obtained from pure deposits, from silver ores such as argentite (Ag2S) and horn silver (AgCl), and in conjunction with deposits of ores containing lead, gold or copper.
The Latin word for silver is argentum. Silver has been known since ancient times. It is mentioned in Genesis. Slag dumps in Asia Minor and on islands in the Aegean Sea indicate that man learned to separate silver from lead as early as 3000 B.C.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
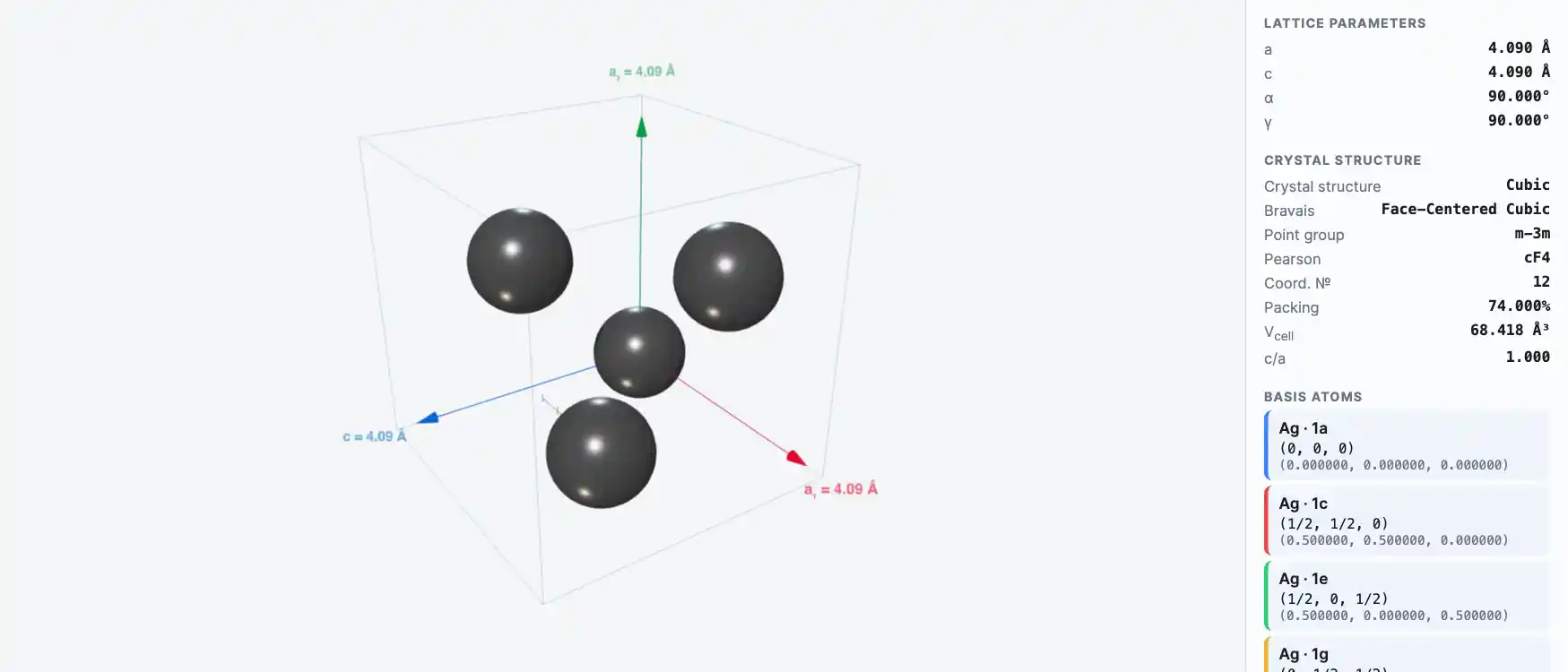
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ag: 4d¹⁰ 5s¹[Kr] 4d¹⁰ 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 107 Stable | 106.9050916 ± 0.0000026 | 51.8390% | Stable |
| 109 Stable | 108.9047553 ± 0.0000014 | 48.1610% | Stable |
Phase / State
Reason: 936.8 °C below melting point (961.78 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 47 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ag I | 0 | 103 | 7 | 97 |
| Ag II | +1 | 455 | 237 | 455 |
| Ag III | +2 | 140 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ag I | 0 | 107 |
| Ag II | +1 | 100 |
| Ag III | +2 | 64 |
| Ag IV | +3 | 2 |
| Ag V | +4 | 2 |
| Ag VI | +5 | 2 |
| Ag VII | +6 | 2 |
| Ag VIII | +7 | 2 |
| Ag IX | +8 | 2 |
| Ag X | +9 | 2 |
Ionic Radii
Showing 10 of 11 Ionic Radii.
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 2 | N/A | 67 pm |
| +1 | 4 | N/A | 100 pm |
| +1 | 4 | N/A | 102 pm |
| +1 | 5 | N/A | 109.00000000000001 pm |
| +1 | 6 | N/A | 114.99999999999999 pm |
| +1 | 7 | N/A | 122 pm |
| +1 | 8 | N/A | 128 pm |
| +2 | 4 | N/A | 79 pm |
| +2 | 6 | N/A | 94 pm |
| +3 | 4 | N/A | 67 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 107 Stable | 106.9050916 ± 0.0000026 | 51.8390% ± 0.0080% | Stable | stable | |
| 109 Stable | 108.9047553 ± 0.0000014 | 48.1610% ± 0.0080% | Stable | stable |
Spectral Lines
Showing 50 of 125 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 562.2482 nm | 21000 | Ag II | emission | 4d9.(2D<5/2>).5d 2[7/2] → 4d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 540.01037 nm | 20000 | Ag II | emission | 4d9.(2D<5/2>).5d 2[9/2] → 4d9.(2D<5/2>).4f 2[11/2]* | Measured | NIST | |
| 540.31323 nm | 15000 | Ag II | emission | 4d9.(2D<5/2>).5d 2[9/2] → 4d9.(2D<5/2>).4f 2[11/2]* | Measured | NIST | |
| 555.19264 nm | 12000 | Ag II | emission | 4d9.(2D<5/2>).5d 2[7/2] → 4d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 548.81562 nm | 8300 | Ag II | emission | 4d9.(2D<5/2>).5d 2[5/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 558.97829 nm | 4200 | Ag II | emission | 4d9.(2D<5/2>).5d 2[5/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 534.00267 nm | 2900 | Ag II | emission | 4d9.(2D<5/2>).5d 2[9/2] → 4d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 554.32121 nm | 2700 | Ag II | emission | 4d9.(2D<5/2>).5d 2[5/2] → 4d8.(3F).5s.5p.(3P*) 1F* | Measured | NIST | |
| 557.96782 nm | 2400 | Ag II | emission | 4d9.(2D<5/2>).5d 2[7/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 549.38302 nm | 2300 | Ag II | emission | 4d9.(2D<5/2>).5d 2[5/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 555.81412 nm | 2300 | Ag II | emission | 4d9.(2D<5/2>).5d 2[5/2] → 4d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 557.38257 nm | 2300 | Ag II | emission | 4d9.(2D<5/2>).5d 2[7/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 542.40509 nm | 2200 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 541.08117 nm | 1800 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 514.28157 nm | 1700 | Ag II | emission | 4d9.(2D<5/2>).5d 2[1/2] → 4d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 558.84183 nm | 1700 | Ag II | emission | 4d9.(2D<5/2>).5d 2[7/2] → 4d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 536.27883 nm | 1600 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 539.24682 nm | 1500 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[5/2]* | Measured | NIST | |
| 547.86589 nm | 1500 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[1/2]* | Measured | NIST | |
| 533.25049 nm | 1300 | Ag II | emission | 4d9.(2D<5/2>).5d 2[9/2] → 4d9.(2D<5/2>).4f 2[7/2]* | Measured | NIST | |
| 531.24574 nm | 1200 | Ag II | emission | 4d9.(2D<5/2>).5d 2[9/2] → 4d9.(2D<5/2>).4f 2[9/2]* | Measured | NIST | |
| 520.9078 nm | 1000 | Ag I | emission | 4d10.5p 2P* → 4d10.5d 2D | Measured | NIST | |
| 546.54853 nm | 1000 | Ag I | emission | 4d10.5p 2P* → 4d10.5d 2D | Measured | NIST | |
| 441.196 nm | 830 | Ag II | emission | 4d9.(2D<5/2>).6p 2[7/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 541.19338 nm | 740 | Ag II | emission | 4d9.(2D<5/2>).5d 2[3/2] → 4d9.(2D<5/2>).4f 2[3/2]* | Measured | NIST | |
| 513.72469 nm | 720 | Ag II | emission | 4d9.(2D<5/2>).5d 2[1/2] → 4d8.(3F).5s.5p.(3P*) 1D* | Measured | NIST | |
| 421.09542 nm | 700 | Ag I | emission | 4d10.5p 2P* → 4d10.6d 2D | Measured | NIST | |
| 431.959 nm | 630 | Ag II | emission | 4d9.(2D<5/2>).6p 2[7/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 443.063 nm | 580 | Ag II | emission | 4d9.(2D<3/2>).6p 2[5/2]* → 4d9.(2D<3/2>).8s 2[3/2] | Measured | NIST | |
| 408.59155 nm | 470 | Ag II | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d8.5s2 1G | Measured | NIST | |
| 449.492 nm | 410 | Ag II | emission | 4d9.(2D<5/2>).6p 2[5/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 453.041 nm | 410 | Ag II | emission | 4d9.(2D<5/2>).6p 2[5/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 405.5475 nm | 400 | Ag I | emission | 4d10.5p 2P* → 4d10.6d 2D | Measured | NIST | |
| 431.354 nm | 290 | Ag II | emission | 4d9.(2D<3/2>).6p 2[5/2]* → 4d9.(2D<3/2>).8s 2[3/2] | Measured | NIST | |
| 436.409 nm | 290 | Ag II | emission | 4d9.(2D<3/2>).6p 2[1/2]* → 4d9.(2D<3/2>).8s 2[3/2] | Measured | NIST | |
| 444.917 nm | 290 | Ag II | emission | 4d9.(2D<3/2>).6p 2[3/2]* → 4d9.(2D<3/2>).8s 2[3/2] | Measured | NIST | |
| 478.83966 nm | 260 | Ag II | emission | 4d9.(2D<3/2>).5p 2[3/2]* → 4d8.5s2 1D | Measured | NIST | |
| 418.547499 nm | 250 | Ag II | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d8.5s2 1D | Measured | NIST | |
| 723.9381 nm | 250 | Ag II | emission | 4d9.(2D<5/2>).6s 2[5/2] → 4d8.(3F).5s.5p.(3P*) 5G* | Measured | NIST | |
| 398.51904 nm | 220 | Ag II | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d8.5s2 3P | Measured | NIST | |
| 433.316 nm | 210 | Ag II | emission | 4d9.(2D<5/2>).6p 2[7/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 447.909 nm | 210 | Ag II | emission | 4d9.(2D<5/2>).6p 2[3/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 451.558 nm | 210 | Ag II | emission | 4d9.(2D<5/2>).6p 2[5/2]* → 4d9.(2D<5/2>).8s 2[5/2] | Measured | NIST | |
| 381.09396 nm | 200 | Ag I | emission | 4d10.5p 2P* → 4d10.7d 2D | Measured | NIST | |
| 699.906 nm | 200 | Ag II | emission | 4d8.(3F).5s.5p.(3P*) 5D* → 4d9.(2D<3/2>).7s 2[3/2] | Measured | NIST | |
| 392.01238 nm | 180 | Ag II | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d8.5s2 3P | Measured | NIST | |
| 462.00355 nm | 170 | Ag II | emission | 4d9.(2D<3/2>).5p 2[5/2]* → 4d8.5s2 1D | Measured | NIST | |
| 394.9435 nm | 160 | Ag II | emission | 4d9.(2D<5/2>).5p 2[3/2]* → 4d8.5s2 3P | Measured | NIST | |
| 502.73432 nm | 160 | Ag II | emission | 4d9.(2D<3/2>).5p 2[3/2]* → 4d8.5s2 1D | Measured | NIST | |
| 390.930327 nm | 140 | Ag II | emission | 4d9.(2D<5/2>).5p 2[5/2]* → 4d8.5s2 1D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1234.93 K |
| Boiling point | 2435.15 K |
| Critical point (temperature) | 6410.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.9577 |
| 2 | p | 4.0806 |
| 2 | s | 12.3658 |
| 3 | d | 14.4602 |
| 3 | p | 17.1914 |
| 3 | s | 16.9688 |
| 4 | d | 32.2372 |
| 4 | p | 28.4376 |
| 4 | s | 27.1352 |
| 5 | s | 40.2445 |
Crystal Radii Detail (11)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | II | 81 | ||
| 1 | IV | 114 | calculated, | |
| 1 | IVSQ | 116 | ||
| 1 | V | 123 | calculated, | |
| 1 | VI | 129 | calculated, | |
| 1 | VII | 136 | ||
| 1 | VIII | 142 | ||
| 2 | IVSQ | 93 | ||
| 2 | VI | 108 | ||
| 3 | IVSQ | 81 |
Isotope Decay Modes (68)
| Isotope | Mode | Intensity |
|---|---|---|
| 92 | B+ | — |
| 92 | p | — |
| 93 | p | — |
| 93 | B+ | — |
| 93 | B+p | — |
| 94 | B+ | 100% |
| 94 | B+p | 0.2% |
| 95 | B+ | 100% |
| 95 | B+p | 2.3% |
| 96 | B+ | 100% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.18566 |
| 10.1617 | — | 1.22941 |
| 10.3261 | — | 1.27478 |
| 10.4931 | — | 1.32182 |
| 10.6628 | — | 1.38215 |
| 10.8353 | — | 1.45541 |
| 11.0106 | — | 1.53256 |
| 11.1886 | — | 1.61379 |
| 11.3696 | — | 1.69933 |
| 11.5535 | — | 1.78755 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
7.5×10-2 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
Silver occurs natively and in ores such as argentite (Ag2S) and horn silver (AgCl); lead, lead-zinc, copper, gold, and copper-nickel ores are principal sources. Mexico, Canada, Peru, and the U.S. are the principal silver producers in the western hemisphere.
References (1)
- [6] Silver https://periodic.lanl.gov/47.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Silver is also recovered during electrolytic refining of copper. Commercial fine silver contains at least 99.9% silver. Purities of 99.999+% are available commercially.
References (1)
- [6] Silver https://periodic.lanl.gov/47.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Silver isotope-amount ratiosn(107Ag)/n(109Ag) along with isotope-amount ratios of copper n(65Cu)/n(63Cu), and isotope-amount ratios of lead (n(206Pb)/n(204Pb), n(207Pb)/n(204Pb) and n(208Pb)/n(204Pb)) have been used to determine origins of European coins and information on the flow of goods in the world market over time (Fig. IUPAC.47.1). Metals from Peru and Mexico and those from European mining have distinct isotopic signatures that enable the origin of the metal to be determined by examining the isotopic compositions of silver, copper, and lead in the coins. Abundant silver sources, mined in Mexico and Peru in the 16 th century, were used to mint coins, but they were not a major influence in the European coin market until the 18 th century (Fig. IUPAC.47.1) [237] [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).[237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011)..
References (2)
- [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Silver.
The element property data was retrieved from publications.