Cadmium (Cd)
transition-metalSolid
Standard Atomic Weight
112.414 uElectron configuration
[Kr] 5s2 4d10Melting point
321.07 °C (594.22 K)Boiling point
766.85 °C (1040 K)Density
8690 kg/m³Oxidation states
−2, +1, +2Electronegativity (Pauling)
1.69Ionization energy (1st)
Discovery year
1817Atomic radius
155 pmDetails
Cadmium is a soft, bluish-white post-transition metal in group 12. It occurs mainly as a minor constituent of zinc ores and is usually recovered as a by-product of zinc refining. Chemically it is dominated by the +2 oxidation state and forms many salts with ionic character. Its technological importance has declined in some applications because cadmium and many cadmium compounds are highly toxic, but it remains useful where particular electrochemical, pigment, or semiconductor properties are required.
Soft bluish metal belonging to group 12 of the periodic table. Extremely toxic even in low concentrations. Chemically similar to zinc, but lends itself to more complex compounds. Discovered in 1817 by F. Stromeyer.
The name derives from Greek kadmeia for "calamine" (zinc carbonate), with which it was found as an impurity in nature. It may have been found in furnace flue dust in Thebes, a city in the Boeottia region of central Greece. The mythological king of Phoenicia, Cadmus, founded Thebes and would be a source for the name of the ore. The element was discovered and first isolated by German physician Friedrich Stromeyer in 1817.
Cadmium was discovered by Friedrich Strohmeyer, a German chemist, in 1817 while studying samples of calamine (ZnCO3). When heated, Strohmeyer noticed that some samples of calamine glowed with a yellow color while other samples did not. After further examination, he determined that the calamine that changed color when heated contained trace amounts of a new element. There is only one mineral that contains significant amounts of cadmium, greenockite (CdS), but it is not common enough to mine profitably. Fortunately, small amounts of cadmium are found in zinc ores and most of the cadmium produced today is obtained as a byproduct of mining and refining zinc.
From the Latin word cadmia, Greek kadmeia - the ancient name for calamine, zinc carbonate. Discovered by Stromeyer in 1817 from an impurity in zinc carbonate. Cadmium most often occurs in small quantities associated with zinc ores, such as sphalerite (ZnS). Greenockite (CdS) is the only mineral of any consequence bearing cadmium. Almost all cadmium is obtained as a by-product in the treatment of zinc, copper, and lead ores. It is a soft, bluish-white metal which is easily cut with a knife. It is similar in many respects to zinc. In 1927 the International Conference on Weights and Measures redefined the meter in terms of the wavelength of the red cadmium spectral line (i.e. 1m = 1.553,164.13 wavelengths). This definition has been changed (see Krypton).
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
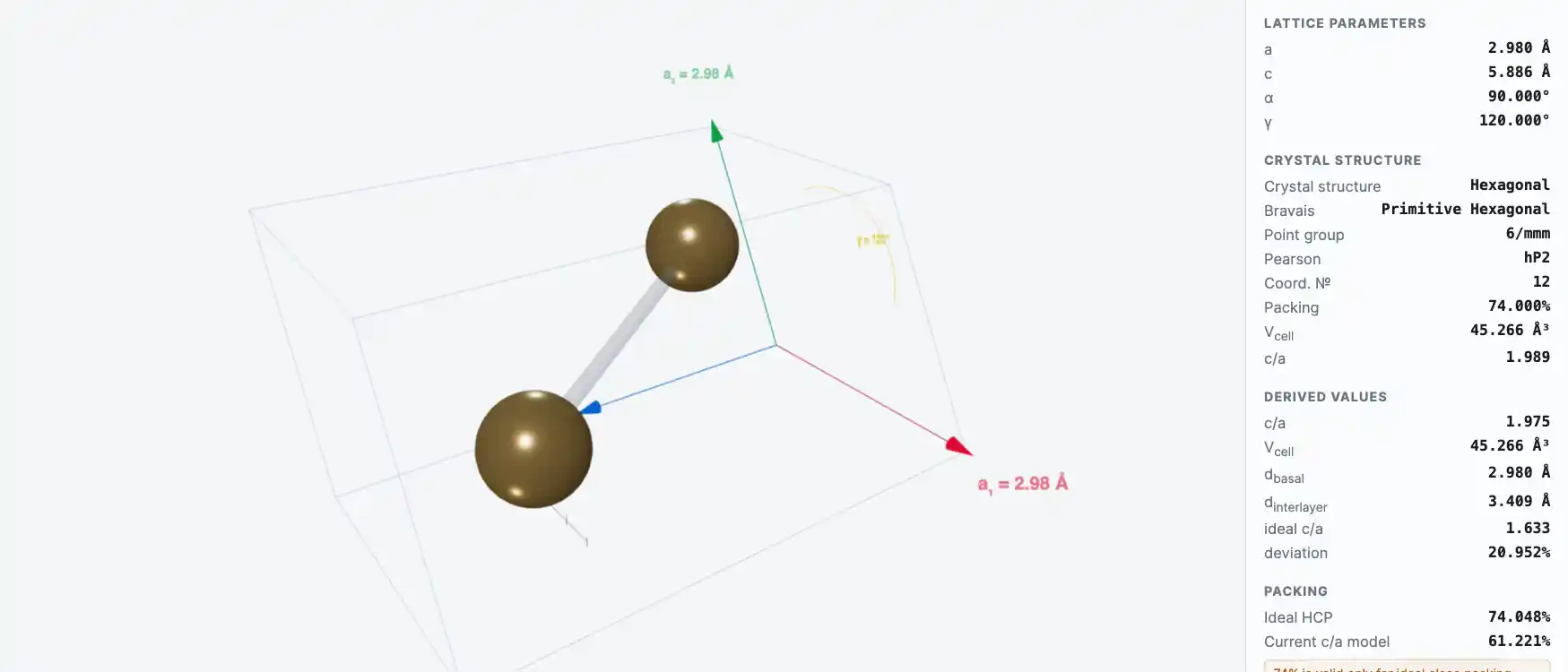
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Cd: 4d¹⁰ 5s²[Kr] 4d¹⁰ 5s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 110 Stable | 109.90300661 ± 0.00000061 | 12.4900% | Stable |
| 111 Stable | 110.90418287 ± 0.00000061 | 12.8000% | Stable |
| 112 Stable | 111.90276287 ± 0.0000006 | 24.1300% | Stable |
Phase / State
Reason: 296.1 °C below melting point (321.07 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 48 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Cd I | 0 | 69 | 18 | 69 |
| Cd II | +1 | 173 | 87 | 173 |
| Cd III | +2 | 95 | 0 | 0 |
| Cd IV | +3 | 102 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Cd I | 0 | 128 |
| Cd II | +1 | 101 |
| Cd III | +2 | 48 |
| Cd IV | +3 | 52 |
| Cd V | +4 | 2 |
| Cd VI | +5 | 2 |
| Cd VII | +6 | 2 |
| Cd VIII | +7 | 2 |
| Cd IX | +8 | 2 |
| Cd X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 78 pm |
| +2 | 5 | N/A | 87 pm |
| +2 | 6 | N/A | 95 pm |
| +2 | 7 | N/A | 103 pm |
| +2 | 8 | N/A | 110.00000000000001 pm |
| +2 | 12 | N/A | 131 pm |
Compounds
Isotopes (3)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 110 Stable | 109.90300661 ± 0.00000061 | 12.4900% ± 0.1800% | Stable | stable | |
| 111 Stable | 110.90418287 ± 0.00000061 | 12.8000% ± 0.1200% | Stable | stable | |
| 112 Stable | 111.90276287 ± 0.0000006 | 24.1300% ± 0.2100% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 398.19257 nm | 10 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.9s 1S | Measured | NIST | |
| 414.03021 nm | N/A | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.7d 1D | Measured | NIST | |
| 430.66718 nm | 8 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.8s 1S | Measured | NIST | |
| 441.29894 nm | 3 | Cd I | emission | 4d10.5s.5p 3P* → 4d10.5s.6s 1S | Measured | NIST | |
| 466.2352 nm | 8 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.6d 1D | Measured | NIST | |
| 467.815 nm | 200 | Cd I | emission | 4d10.5s.5p 3P* → 4d10.5s.6s 3S | Measured | NIST | |
| 479.99121 nm | 300 | Cd I | emission | 4d10.5s.5p 3P* → 4d10.5s.6s 3S | Measured | NIST | |
| 508.58214 nm | N/A | Cd I | emission | 4d10.5s.5p 3P* → 4d10.5s.6s 3S | Measured | NIST | |
| 515.46618 nm | 6 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.7s 1S | Measured | NIST | |
| 609.9142 nm | 300 | Cd I | emission | 4d10.5s.6s 3S → 4d10.5s.8p 3P* | Measured | NIST | |
| 611.1495 nm | 100 | Cd I | emission | 4d10.5s.6s 3S → 4d10.5s.8p 3P* | Measured | NIST | |
| 632.51689 nm | 100 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.5d 3D | Measured | NIST | |
| 633.00149 nm | 30 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.5d 3D | Measured | NIST | |
| 643.84695 nm | 2000 | Cd I | emission | 4d10.5s.5p 1P* → 4d10.5s.5d 1D | Measured | NIST | |
| 677.8116 nm | 30 | Cd I | emission | 4d10.5s.6s 1S → 4d10.5s.8p 1P* | Measured | NIST | |
| 734.5665 nm | 1000 | Cd I | emission | 4d10.5s.6s 3S → 4d10.5s.7p 3P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 594.22 K |
| Boiling point | 1040.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.9744 |
| 2 | p | 4.091 |
| 2 | s | 12.6142 |
| 3 | d | 14.3931 |
| 3 | p | 17.3085 |
| 3 | s | 17.1588 |
| 4 | d | 32.1232 |
| 4 | p | 28.5888 |
| 4 | s | 27.1308 |
| 5 | s | 39.808 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | 92 | ||
| 2 | V | 101 | ||
| 2 | VI | 109 | ||
| 2 | VII | 117 | calculated, | |
| 2 | VIII | 124 | calculated, | |
| 2 | XII | 145 |
Isotope Decay Modes (59)
| Isotope | Mode | Intensity |
|---|---|---|
| 94 | B+ | — |
| 94 | B+p | — |
| 95 | B+ | 100% |
| 95 | B+p | 4.6% |
| 96 | B+ | 100% |
| 96 | B+p | 1.6% |
| 97 | B+ | 100% |
| 97 | B+p | 7.4% |
| 98 | B+ | 100% |
| 98 | B+p | 0% |
X‑ray Scattering Factors (510)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.40004 |
| 10.1617 | — | 0.41842 |
| 10.3261 | — | 0.43764 |
| 10.4931 | — | 0.45775 |
| 10.6628 | — | 0.49362 |
| 10.8353 | — | 0.53685 |
| 11.0106 | — | 0.59258 |
| 11.1886 | — | 0.66328 |
| 11.3696 | — | 0.74242 |
| 11.5535 | — | 0.86125 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.5×10-1 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.1×10-4 milligrams per liter
References (1)
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Cadmium.
The element property data was retrieved from publications.