Carbon (C)
nonmetalSolid
Standard Atomic Weight
12.011 u [12.0096, 12.0116]Electron configuration
[He] 2s2 2p2Melting point
3549.85 °C (3823 K)Boiling point
3824.85 °C (4098 K)Density
2267 kg/m³Oxidation states
−4, −3, −2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
2.55Ionization energy (1st)
Discovery year
1797Atomic radius
70 pmDetails
Carbon is a nonmetal in group 14 and the defining element of organic chemistry. Its small atoms form strong covalent bonds with carbon and many other elements, allowing chains, rings, networks, and multiple bonds of great diversity. It occurs naturally as graphite, diamond, amorphous carbon-rich materials, carbonate minerals, fossil carbon, dissolved carbon species, and as a central element in living matter.
Carbon is a member of group 14 of the periodic table. It has three allotropic forms of it, diamonds, graphite and fullerite. Carbon-14 is commonly used in radioactive dating. Carbon occurs in all organic life and is the basis of organic chemistry. Carbon has the interesting chemical property of being able to bond with itself, and a wide variety of other elements.
The name derives from the Latin carbo for "charcoal". It was known in prehistoric times in the form of charcoal and soot. In 1797, the English chemist Smithson Tennant proved that diamond is pure carbon.
Carbon, the sixth most abundant element in the universe, has been known since ancient times. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Three naturally occurring allotropes of carbon are known to exist: amorphous, graphite and diamond.
From the Latin word carbo: charcoal. Carbon, an element of prehistoric discovery, is very widely distributed in nature. It is found in abundance in the sun, stars, comets, and atmospheres of most planets. Carbon in the form of microscopic diamonds is found in some meteorites.
Natural diamonds are found in kimberlite of ancient volcanic "pipes," found in South Africa, Arkansas, and elsewhere. Diamonds are now also being recovered from the ocean floor off the Cape of Good Hope. About 30% of all industrial diamonds used in the U.S. are now made synthetically.
The energy of the sun and stars can be attributed at least in part to the well-known carbon-nitrogen cycle.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
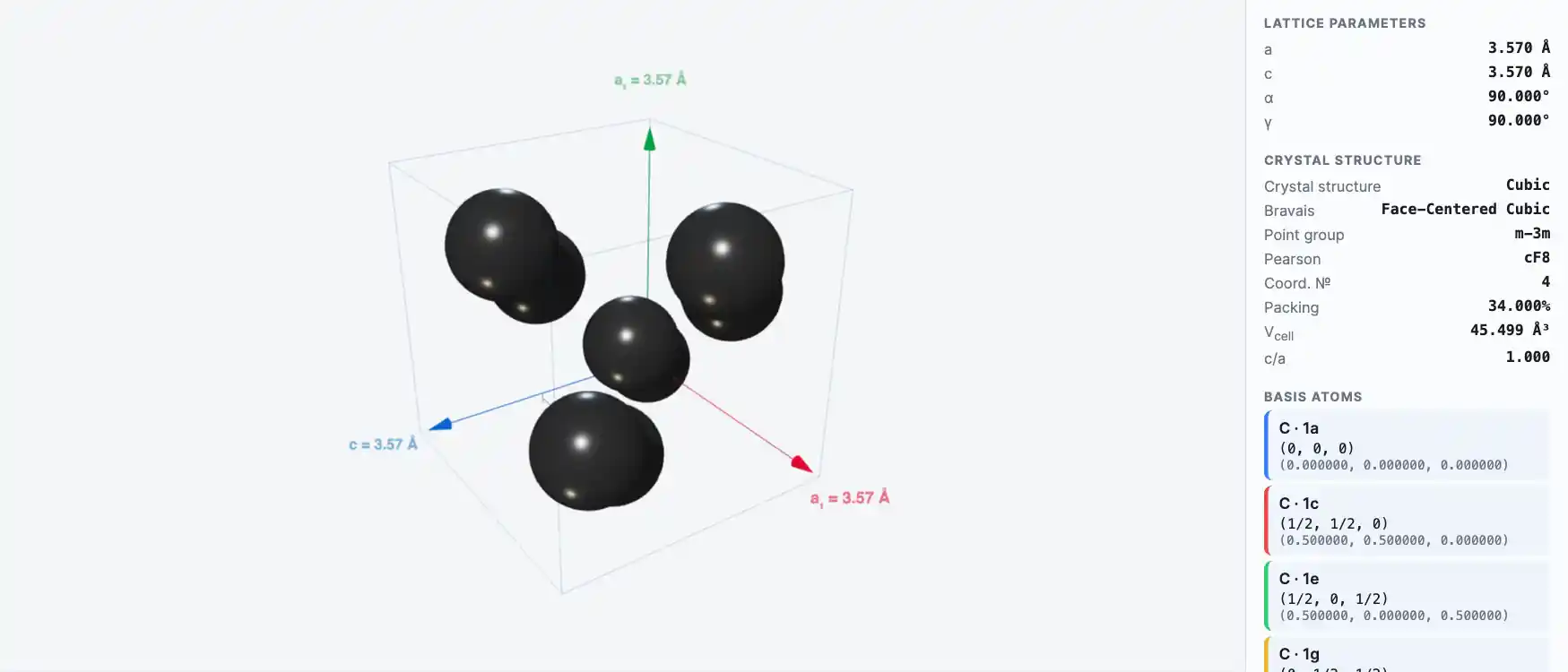
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
C: 2s² 2p²[He] 2s² 2p²1s² 2s² 2p²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 12 Stable | 12 | 98.9300% | Stable |
| 13 Stable | 13.00335483507 ± 0.00000000023 | 1.0700% | Stable |
Phase / State
Reason: 3799.8 °C below sublimation point (3824.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 11 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| C I | 0 | 2102 | 1616 | 2102 |
| 12C I Isotope | 0 | 89 | 0 | 89 |
| 13C I Isotope | 0 | 89 | 0 | 89 |
| 12C II Isotope | +1 | 187 | 0 | 187 |
| 14C II Isotope | +1 | 187 | 0 | 187 |
| C II | +1 | 1605 | 1433 | 1605 |
| 13C II Isotope | +1 | 187 | 0 | 187 |
| C III | +2 | 882 | 878 | 878 |
| C IV | +3 | 259 | 224 | 255 |
| C V | +4 | 149 | 146 | 147 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| C I | 0 | 435 |
| 12C I Isotope | 0 | 33 |
| 13C I Isotope | 0 | 33 |
| 12C II Isotope | +1 | 36 |
| 14C II Isotope | +1 | 36 |
| C II | +1 | 415 |
| 13C II Isotope | +1 | 36 |
| C III | +2 | 201 |
| C IV | +3 | 107 |
| C V | +4 | 156 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 4 | N/A | 15 pm |
| +4 | 6 | N/A | 16 pm |
Compounds
Isotopes (2)
Carbon has seven isotopes. In 1961 the International Union of Pure and Applied Chemistry adopted the isotope carbon-12 as the basis for atomic weights. Carbon-14, an isotope with a half-life of 5715 years, has been widely used to date such materials as wood, archaeological specimens, etc.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 12 Stable | 12 | 98.9300% ± 0.0800% | Stable | stable | |
| 13 Stable | 13.00335483507 ± 0.00000000023 | 1.0700% ± 0.0800% | Stable | stable |
Spectral Lines
Showing 50 of 993 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 505.214927 nm | 160000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.4p 1D | Measured | NIST | |
| 538.033014 nm | 120000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.4p 1P | Measured | NIST | |
| 711.31656 nm | 110000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.4d 3F* | Measured | NIST | |
| 493.202524 nm | 73000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.4p 1S | Measured | NIST | |
| 477.173374 nm | 69000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.4p 3P | Measured | NIST | |
| 711.697758 nm | 45000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5s 3P* | Measured | NIST | |
| 658.76211 nm | 40000 | C I | emission | 2s2.2p.3p 1P → 2s2.2p.4d 1P* | Measured | NIST | |
| 579.311495 nm | 38000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3P | Measured | NIST | |
| 711.96559 nm | 37000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5s 3P* | Measured | NIST | |
| 580.059993 nm | 35000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3P | Measured | NIST | |
| 600.1123 nm | 35000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.6s 3P* | Measured | NIST | |
| 477.589266 nm | 34000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.4p 3P | Measured | NIST | |
| 437.13814 nm | 33000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.5p 1P | Measured | NIST | |
| 711.145795 nm | 32000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.4d 3F* | Measured | NIST | |
| 682.814076 nm | 27000 | C I | emission | 2s2.2p.3p 1P → 2s2.2p.4d 1D* | Measured | NIST | |
| 504.149039 nm | 25000 | C I | emission | 2s.2p3 3D* → 2s2.2p.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 477.002376 nm | 24000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.4p 3P | Measured | NIST | |
| 600.6012 nm | 23000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5d 3D* | Measured | NIST | |
| 665.55294 nm | 20000 | C I | emission | 2s2.2p.3p 1P → 2s2.2p.5s 1P* | Measured | NIST | |
| 710.011312 nm | 19000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5s 3P* | Measured | NIST | |
| 566.894 nm | 18000 | C I | emission | 2s2.2p.3p 1P → 2s2.2p.5d 1P* | Measured | NIST | |
| 596.933151 nm | 18000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3D | Measured | NIST | |
| 708.782188 nm | 18000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.4d 3D* | Measured | NIST | |
| 402.94119 nm | 16000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.5p 3P | Measured | NIST | |
| 601.64487 nm | 16000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5d 3F* | Measured | NIST | |
| 473.426281 nm | 15000 | C I | emission | 2s.2p3 3D* → 2s2.2p.5p 3P | Measured | NIST | |
| 481.737213 nm | 15000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.4p 3S | Measured | NIST | |
| 579.446608 nm | 15000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3P | Measured | NIST | |
| 748.344451 nm | 15000 | C I | emission | 2s2.2p.3p 3S → 2s2.2p.4d 3P* | Measured | NIST | |
| 406.52425 nm | 14000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.5p 3D | Measured | NIST | |
| 580.52017 nm | 14000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3P | Measured | NIST | |
| 601.4833 nm | 14000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.6s 3P* | Measured | NIST | |
| 710.89263 nm | 14000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.5s 3P* | Measured | NIST | |
| 400.9928 nm | 13000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.6p 1P | Measured | NIST | |
| 422.83269 nm | 13000 | C I | emission | 2s2.2p.3s 1P* → 2s2.2p.5p 1S | Measured | NIST | |
| 447.85821 nm | 13000 | C I | emission | 2s.2p3 3D* → 2s2.2p.(2P*<1/2>).5f 2[5/2] | Measured | NIST | |
| 504.012903 nm | 12000 | C I | emission | 2s.2p3 3D* → 2s2.2p.(2P*<1/2>).4f 2[7/2] | Measured | NIST | |
| 601.0669 nm | 12000 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.6s 3P* | Measured | NIST | |
| 406.4264 nm | 11000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.5p 3D | Measured | NIST | |
| 639.7965 nm | 11000 | C I | emission | 2s2.2p.3p 3S → 2s2.2p.6s 3P* | Measured | NIST | |
| 666.3043 nm | 11000 | C I | emission | 2s2.2p.3p 3P → 2s2.2p.5d 3D* | Measured | NIST | |
| 667.1849 nm | 11000 | C I | emission | 2s2.2p.3p 3P → 2s2.2p.6s 3P* | Measured | NIST | |
| 403.180216 nm | 10000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.5p 3P | Measured | NIST | |
| 482.679468 nm | 10000 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.4p 3S | Measured | NIST | |
| 598.903753 nm | 10000 | C I | emission | 2s.2p3 3D* → 2s2.2p.4p 3D | Measured | NIST | |
| 707.649944 nm | 9900 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.4d 3D* | Measured | NIST | |
| 402.284321 nm | 9700 | C I | emission | 2s2.2p.3s 3P* → 2s2.2p.5p 3P | Measured | NIST | |
| 555.1578 nm | 9600 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.7s 3P* | Measured | NIST | |
| 600.7173 nm | 9600 | C I | emission | 2s2.2p.3p 3D → 2s2.2p.6s 3P* | Measured | NIST | |
| 502.492938 nm | 9400 | C I | emission | 2s.2p3 3D* → 2s2.2p.(2P*<3/2>).4f 2[7/2] | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Boiling point | 4098.15 K |
| Triple point (temperature) | 4762.15 K |
| Triple point (pressure) | 10300 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3273 |
| 2 | p | 2.8642 |
| 2 | s | 2.7834 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | III | 6 | ||
| 4 | IV | 29 | Pauling's (1960) crystal radius, | |
| 4 | VI | 30 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (27)
| Isotope | Mode | Intensity |
|---|---|---|
| 8 | 2p | 100% |
| 9 | B+ | 100% |
| 9 | B+p | 7.5% |
| 9 | B+A | 38.4% |
| 10 | B+ | 100% |
| 11 | B+ | 100% |
| 14 | B- | 100% |
| 15 | B- | 100% |
| 16 | B- | 100% |
| 16 | B-n | 99% |
X‑ray Scattering Factors (502)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.80688 |
| 10.1617 | — | 0.85152 |
| 10.3261 | — | 0.89863 |
| 10.4931 | — | 0.94834 |
| 10.6628 | — | 1.0008 |
| 10.8353 | — | 1.05755 |
| 11.0106 | — | 1.12167 |
| 11.1886 | — | 1.18968 |
| 11.3696 | — | 1.26181 |
| 11.5535 | — | 1.33832 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.00×102 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2.8×101 milligrams per liter
References (1)
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Variations in the isotope-amount ratio n(13C)/n(12C) of biological products can be observed using isotope-ratio mass spectrometry (IRMS) to detect adulteration (the addition of inferior ingredients) in honey and other food products.
The isotope-amount ratio n(13C)/n(12C) can fluctuate between carbon sources, for example C3 plants (found in temperate climates and which use atmospheric carbon dioxide to make a 3-carbon molecule during photosynthesis — examples include rice, potatoes, tomatoes, and sugar beets), C4 plants (found in hot climates and which use atmospheric carbon dioxide to make a 4-carbon molecule during photosynthesis — examples include corn and sugar cane), animal carbon, atmospheric CO2, etc. This commonly makes it possible to detect whether these different carbon sources have been mixed by using isotope or mass balance to distinguish, for example, between beet sugar and cane sugar. Complications in source identification can arise with plants that open stomata at night to collect carbon dioxide to use a third mechanism to fix atmospheric carbon dioxide (CAM or crassulacean acid metabolism). The isotope-amount ratio n(13C)/n(12C) of CAM plants overlaps that of C3 or C4 plants — examples include pineapples and jade plants. The following adulterations are commonly detected using stable carbon isotope IRMS:
–Variations in the isotope-amount ratio n(13C)/n(12C) of honey are used to detect the addition (and potential adulteration) of high fructose corn syrup, corn, or sugar cane [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)..
–Variations in the isotope-amount ratio n(13C)/n(12C) of fruit juice have been used to detect the addition of a sugar [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)..
–Variations in the isotope-amount ratio n(13C)/n(12C) of natural vanilla extract have been used to detect the addition of artificial vanillin or p-hydroxybenzaldehyde [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)..
–Variations in the isotope-amount ratio n(13C)/n(12C) of beer are used to detect C4 carbon, which would indicate that a beer company may have added ingredients that are not traditionally used in brewing beer. Therefore, this ratio is used to detect the misrepresentation of a product as being pure [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)., [68] [68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).[68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002)..
Stable carbon IRMS has been used to determine if the botanical origin of an alcoholic spirit has been mislabeled and if chaptalization (the process of adding sugar to increase the alcoholic content) of wine has occurred [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)., [68] [68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).[68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).. 14C scintillation counting has been used to determine the age of wine and alcoholic spirits [67] [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).[67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002)., [68] [68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).[68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).. Variations in the isotope-amount ratio n(13C)/n(12C) of urine has been used to determine if steroids in urine are natural or of synthetic origin. These measurements enable anti-doping laboratories to perfect their methods for detecting steroid doping in athletes [69] [69] B. D. Ahrens, A. W. Butch. Drug Test Anal.5, 534 (2013).[69] B. D. Ahrens, A. W. Butch. Drug Test Anal.5, 534 (2013)., [70] [70] E. Bulska, D. Gorczyca, I. Zalewska, A. Pokrywka, D. Kwiatkowska. J. Pharm. Biomed. Anal.106, 159 (2015).[70] E. Bulska, D. Gorczyca, I. Zalewska, A. Pokrywka, D. Kwiatkowska. J. Pharm. Biomed. Anal.106, 159 (2015)., [71] [71] A. Casilli, T. Piper, F. A. de Oliveira, M. Costa Padilha, H. Marcelo Pereira, M. Thevis, F. R. de Aquino Neto. Drug Test Anal.8, 1204 (2016).[71] A. Casilli, T. Piper, F. A. de Oliveira, M. Costa Padilha, H. Marcelo Pereira, M. Thevis, F. R. de Aquino Neto. Drug Test Anal.8, 1204 (2016).. Variations in the isotope-amount ratio n(13C)/n(12C) of marijuana can provide information to determine if the plants were grown “inside” a building or greenhouse or were “open grown” (Fig. IUPAC.6.4). Plant carbon isotopic compositions are controlled by atmospheric CO2 and the supply and demand of CO2 in photosynthesis (the process used by plants to convert light energy from the sun into chemical energy). “Open grown” plants are grown in an area that is well ventilated and receives natural CO2. In contrast, plants grown “inside” receive supplemented CO2 and the photosynthesis process is more confined. Additionally, CO2 from a tank of compressed gas used to augment atmospheric CO2 to increase the growth of marijuana plants is commonly highly depleted in 13C as a refinery by-product. These differences change the carbon isotope ratios of the plants and the ratios vary enough to enable the determination of the growing and cultivation process of marijuana [72] [72] E. K. Shibuya, J. E. Souza Sarkis, O. N. Neto, M. Z. Moreira, R. L. Victoria. Forensic Sci. Int.160, 35 (2006).[72] E. K. Shibuya, J. E. Souza Sarkis, O. N. Neto, M. Z. Moreira, R. L. Victoria. Forensic Sci. Int.160, 35 (2006)., [73] [73] J. B. West, J. M. Hurley, J. R. Ehleringer. J Forensic Sci.54, 84 (2009).[73] J. B. West, J. M. Hurley, J. R. Ehleringer. J Forensic Sci.54, 84 (2009)..
References (9)
- [67] C. Cordella, I. Moussa, A. C. Martel, N. Sbirrazzuoli, L. Lizzani-Cuvelier. J. Agric. Food. Chem.50, 1751 (2002).
- [68] J. R. Brooks, N. Buchmann, S. Phillips, B. Ehleringer, R. D. Evans, M. Lott, L. A. Martinelli, W. T. Pockman, D. Sandquist, J. P. Sparks, L. Sperry, D. Williams, J. R. Ehleringer. J. Agric. Food. Chem.50, 6413 (2002).
- [69] B. D. Ahrens, A. W. Butch. Drug Test Anal.5, 534 (2013).
- [70] E. Bulska, D. Gorczyca, I. Zalewska, A. Pokrywka, D. Kwiatkowska. J. Pharm. Biomed. Anal.106, 159 (2015).
- [71] A. Casilli, T. Piper, F. A. de Oliveira, M. Costa Padilha, H. Marcelo Pereira, M. Thevis, F. R. de Aquino Neto. Drug Test Anal.8, 1204 (2016).
- [72] E. K. Shibuya, J. E. Souza Sarkis, O. N. Neto, M. Z. Moreira, R. L. Victoria. Forensic Sci. Int.160, 35 (2006).
- [73] J. B. West, J. M. Hurley, J. R. Ehleringer. J Forensic Sci.54, 84 (2009).
- [74] United States Drug Enforcement Administration. Marijuana-Indoor Marijuana Grow, United States Department of Justice (2014), Feb. 22; http://www.justice.gov/dea/pr/multimedia-library/image-gallery/images_marijuana.shtml.
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Carbon.
The element property data was retrieved from publications.