Nitrogen (N)
nonmetalGas
Standard Atomic Weight
14.007 u [14.00643, 14.00728]Electron configuration
2s2.2p3Melting point
-210 °C (63.15 K)Boiling point
-195.79 °C (77.36 K)Density
1.2506 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
3.04Ionization energy (1st)
Discovery year
1772Atomic radius
65 pmDetails
Nitrogen is a light nonmetal and the main constituent of Earth’s atmosphere as N₂. Its strong N≡N triple bond makes the element relatively inert at ordinary conditions, yet nitrogen chemistry is exceptionally rich once that bond is broken. It is essential in amino acids, nucleic acids, and many cofactors, and industrial fixation of N₂ underpins modern fertilizer production. Nitrogen commonly forms covalent compounds and spans oxidation states from −3 to +5.
Colourless, gaseous element which belongs to group 15 of the periodic table. Constitutes ~78% of the atmosphere and is an essential part of the ecosystem. Nitrogen for industrial purposes is acquired by the fractional distillation of liquid air. Chemically inactive, reactive generally only at high temperatures or in electrical discharges. It was discovered in 1772 by D. Rutherford.
The name derives from the Latin nitrum and Greek nitron for "native soda" and genes for "forming". Nitrogen was discovered by the Scottish physician and chemist Daniel Rutherford in 1772.
Nitrogen was discovered by the Scottish physician Daniel Rutherford in 1772. It is the fifth most abundant element in the universe and makes up about 78% of the earth's atmosphere, which contains an estimated 4,000 trillion tons of the gas. Nitrogen is obtained from liquefied air through a process known as fractional distillation.
From the Latin word nitrum, Greek Nitron, native soda; and genes, forming. Nitrogen was discovered by chemist and physician Daniel Rutherford in 1772. He removed oxygen and carbon dioxide from air and showed that the residual gas would not support combustion or living organisms. At the same time there were other noted scientists working on the problem of nitrogen. These included Scheele, Cavendish, Priestley, and others. They called it "burnt" or" dephlogisticated air," which meant air without oxygen.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
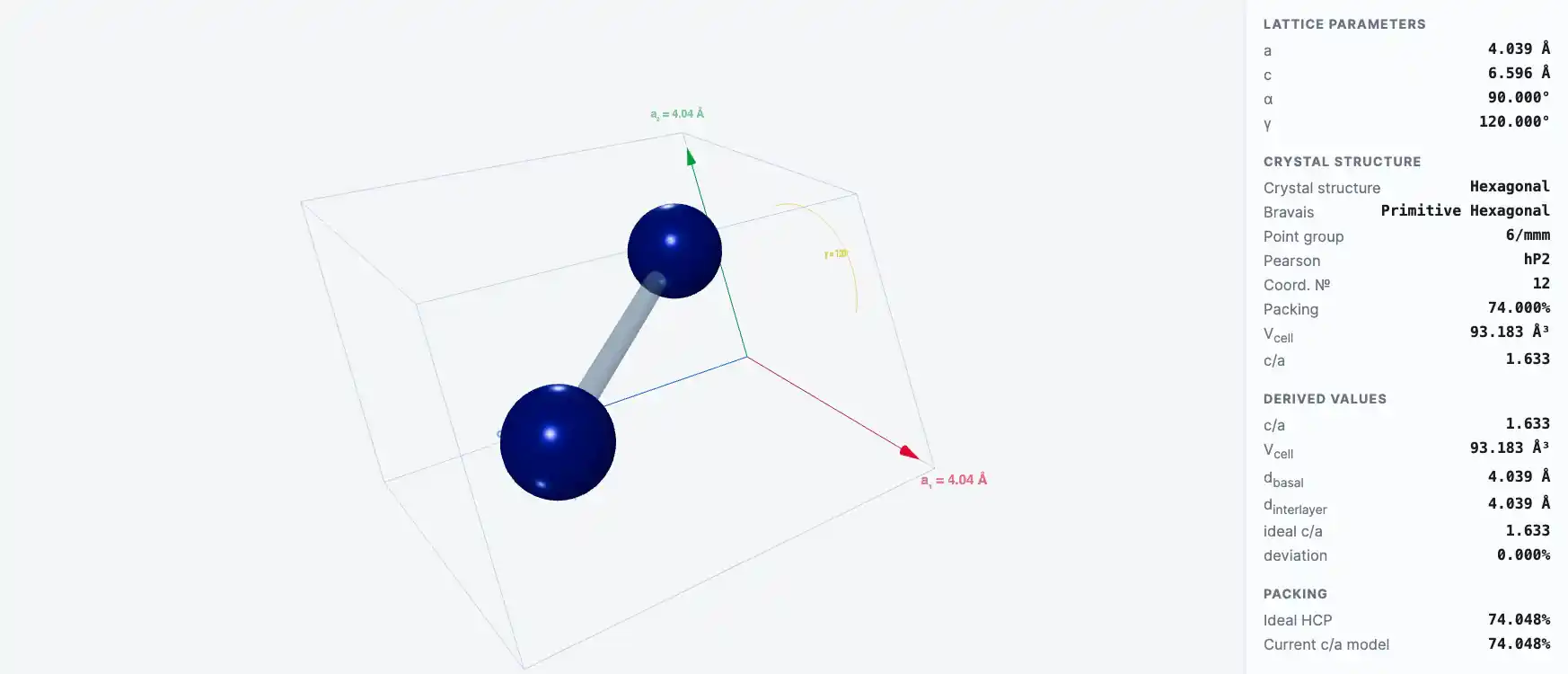
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
N: 2s² 2p³[He] 2s² 2p³1s² 2s² 2p³Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 14 Stable | 14.00307400443 ± 0.0000000002 | 99.6360% | Stable |
| 15 Stable | 15.00010889888 ± 0.00000000064 | 0.3640% | Stable |
Phase / State
Reason: 220.8 °C above boiling point (-195.79 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| N I | 0 | 1309 | 1287 | 1287 |
| N II | +1 | 809 | 786 | 786 |
| N III | +2 | 1378 | 1359 | 1359 |
| N IV | +3 | 536 | 531 | 531 |
| N V | +4 | 442 | 402 | 433 |
| N VI | +5 | 91 | 90 | 91 |
| N VII | +6 | 137 | 137 | 137 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| N I | 0 | 381 |
| N II | +1 | 197 |
| N III | +2 | 347 |
| N IV | +3 | 292 |
| N V | +4 | 151 |
| N VI | +5 | 148 |
| N VII | +6 | 149 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -3 | 4 | N/A | 146 pm |
| +3 | 6 | N/A | 16 pm |
| +5 | 6 | N/A | 13 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 14 Stable | 14.00307400443 ± 0.0000000002 | 99.6360% ± 0.0200% | Stable | stable | |
| 15 Stable | 15.00010889888 ± 0.00000000064 | 0.3640% ± 0.0200% | Stable | stable |
Spectral Lines
Showing 50 of 731 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 399.4997 nm | 1000 | N II | emission | 2s2.2p.3s 1P* → 2s2.2p.3p 1D | Measured | NIST | |
| 746.8312 nm | 900 | N I | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4S* | Measured | NIST | |
| 463.0539 nm | 870 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 500.515 nm | 870 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3F* | Measured | NIST | |
| 567.956 nm | 870 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3D | Measured | NIST | |
| 744.2298 nm | 785 | N I | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4S* | Measured | NIST | |
| 648.205 nm | 750 | N II | emission | 2s2.2p.3s 1P* → 2s2.2p.3p 1P | Measured | NIST | |
| 661.056 nm | 750 | N II | emission | 2s2.2p.3p 1D → 2s2.2p.3d 1F* | Measured | NIST | |
| 575.2499 nm | 700 | N I | emission | 2s2.2p2.(3P).3p 4P* → 2s2.2p2.(3P).5d 4D | Measured | NIST | |
| 742.3641 nm | 685 | N I | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4S* | Measured | NIST | |
| 444.703 nm | 650 | N II | emission | 2s2.2p.3p 1P → 2s2.2p.3d 1D* | Measured | NIST | |
| 500.1474 nm | 650 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3F* | Measured | NIST | |
| 566.663 nm | 650 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3D | Measured | NIST | |
| 594.165 nm | 650 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3D* | Measured | NIST | |
| 460.1478 nm | 550 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 464.3086 nm | 550 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 500.7328 nm | 550 | N II | emission | 2s2.2p.3p 3S → 2s2.2p.3d 3P* | Measured | NIST | |
| 504.5099 nm | 550 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3S | Measured | NIST | |
| 567.602 nm | 550 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3D | Measured | NIST | |
| 593.178 nm | 550 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3D* | Measured | NIST | |
| 395.5851 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 1D | Measured | NIST | |
| 460.7153 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 462.1393 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 480.3287 nm | 450 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3D* | Measured | NIST | |
| 499.436 nm | 450 | N II | emission | 2s.2p2.(4P).3s 5P → 2s.2p2.(4P).3p 5P* | Measured | NIST | |
| 501.0621 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3S | Measured | NIST | |
| 549.5655 nm | 450 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3P* | Measured | NIST | |
| 568.621 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3D | Measured | NIST | |
| 571.077 nm | 450 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3D | Measured | NIST | |
| 391.9001 nm | 360 | N II | emission | 2s2.2p.3p 1P → 2s2.2p.3d 1P* | Measured | NIST | |
| 461.3868 nm | 360 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3P | Measured | NIST | |
| 500.2703 nm | 360 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 3S | Measured | NIST | |
| 501.6381 nm | 360 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3F* | Measured | NIST | |
| 502.5659 nm | 360 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3F* | Measured | NIST | |
| 592.781 nm | 360 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3D* | Measured | NIST | |
| 637.962 nm | 360 | N II | emission | 2s2.2p.3s 3P* → 2s2.2p.3p 1P | Measured | NIST | |
| 648.2699 nm | 360 | N I | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).4d 4F | Measured | NIST | |
| 460.374 nm | 350 | N V | emission | 1s2.3s 2S → 1s2.3p 2P* | Measured | NIST | |
| 648.4808 nm | 325 | N I | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).4d 4F | Measured | NIST | |
| 648.3753 nm | 300 | N I | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).4d 4F | Measured | NIST | |
| 383.8374 nm | 285 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.4s 3P* | Measured | NIST | |
| 422.7736 nm | 285 | N II | emission | 2s2.2p.3p 1D → 2s2.2p.4s 1P* | Measured | NIST | |
| 478.8138 nm | 285 | N II | emission | 2s2.2p.3p 3D → 2s2.2p.3d 3D* | Measured | NIST | |
| 489.5117 nm | 285 | N II | emission | 2s.2p3 1D* → 2s2.2p.3p 1P | Measured | NIST | |
| 498.7376 nm | 285 | N II | emission | 2s2.2p.3p 3S → 2s2.2p.3d 3P* | Measured | NIST | |
| 553.5347 nm | 285 | N II | emission | 2s.2p2.(4P).3s 5P → 2s.2p2.(4P).3p 5D* | Measured | NIST | |
| 574.73 nm | 285 | N II | emission | 2s2.2p.3s 1P* → 2s2.2p.3p 3D | Measured | NIST | |
| 594.024 nm | 285 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3D* | Measured | NIST | |
| 595.239 nm | 285 | N II | emission | 2s2.2p.3p 3P → 2s2.2p.3d 3D* | Measured | NIST | |
| 616.775 nm | 285 | N II | emission | 2s2.2p.3d 3F* → 2s2.2p.4p 3D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 63.15 K |
| Boiling point | 77.35 K |
| Critical point (temperature) | 126.19 K |
| Critical point (pressure) | 3.4 MPa |
| Triple point (temperature) | 63.15 K |
| Triple point (pressure) | 12.52 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3349 |
| 2 | p | 3.166 |
| 2 | s | 3.1526 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -3 | IV | 132 | ||
| 3 | VI | 30 | Ahrens (1952) ionic radius, | |
| 5 | III | 4.4 | ||
| 5 | VI | 27 | Ahrens (1952) ionic radius, |
Isotope Decay Modes (33)
| Isotope | Mode | Intensity |
|---|---|---|
| 10 | p | — |
| 11 | p | 100% |
| 12 | B+ | 100% |
| 12 | B+A | 1.9% |
| 13 | B+ | 100% |
| 16 | B- | 100% |
| 16 | B-A | 0% |
| 17 | B- | 100% |
| 17 | B-n | 95.1% |
| 17 | B-A | 0% |
X‑ray Scattering Factors (503)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.67785 |
| 10.1617 | — | 0.68963 |
| 10.3261 | — | 0.70162 |
| 10.4931 | — | 0.71382 |
| 10.6628 | — | 0.72623 |
| 10.8353 | — | 0.73885 |
| 11.0106 | — | 0.7517 |
| 11.1886 | — | 0.76584 |
| 11.3696 | — | 0.78603 |
| 11.5535 | — | 0.80674 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.9×101 milligrams per kilogram
References (1)
- [5] Nitrogen https://education.jlab.org/itselemental/ele007.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
5×10-1 milligrams per liter
References (1)
- [5] Nitrogen https://education.jlab.org/itselemental/ele007.html
Sources
Sources of this element.
Nitrogen gas (N2) makes up 78.1% of the Earth’s air, by volume. The atmosphere of Mars, by comparison, is only 2.6% nitrogen. From an exhaustible source in our atmosphere, nitrogen gas can be obtained by liquefaction and fractional distillation. Nitrogen is found in all living systems as part of the makeup of biological compounds.
References (1)
- [6] Nitrogen https://periodic.lanl.gov/7.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Stable hydrogen, carbon, and nitrogen isotopic compositions are used to determine the origin of pseudoephedrine from seized methyl-amphetamine made from the pseudoephedrine (drug used as a nasal decongestant or as a stimulant) [91] [91] H. Salouros, G. J. Sutton, J. Howes, D. B. Hibbert, M. Collins. Anal. Chem.85, 9400 (2013).[91] H. Salouros, G. J. Sutton, J. Howes, D. B. Hibbert, M. Collins. Anal. Chem.85, 9400 (2013)..
References (2)
- [91] H. Salouros, G. J. Sutton, J. Howes, D. B. Hibbert, M. Collins. Anal. Chem.85, 9400 (2013).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Nitrogen.
The element property data was retrieved from publications.