Europium (Eu)
lanthanideSolid
Standard Atomic Weight
151.964 uElectron configuration
[Xe] 6s2 4f7Melting point
821.85 °C (1095 K)Boiling point
1528.85 °C (1802 K)Density
5240 kg/m³Oxidation states
0, +2, +3Electronegativity (Pauling)
N/AIonization energy (1st)
Discovery year
1896Atomic radius
185 pmDetails
Europium is a lanthanide rare-earth metal with atomic number 63. It is chemically notable for the relative stability of both Eu³⁺ and Eu²⁺, a contrast to most lanthanides, which are dominated by the +3 state. This redox flexibility controls much of its mineral behavior and its optical technology. Europium is best known for intense, narrow luminescence from Eu³⁺ and Eu²⁺ ions in solid hosts, especially in phosphors and security materials.
As with other rare-earth metals, except for lanthanum, europium ignites in air at about 150 to 180°C. Europium is about as hard as lead and is quite ductile. It is the most reactive of the rare-earth metals, quickly oxidizing in air. It resembles calcium in its reaction with water. Bastnasite and monazite are the principal ores containing europium.
The name derives from the continent of Europe. It was separated from the mineral samaria in magnesium- samarium nitrate by the French chemist Eugène-Anatole Demarçay in 1896. It was also first isolated by Demarçay in 1901.
Europium was discovered by Eugène-Antole Demarçay, a French chemist, in 1896. Demarçay suspected that samples of a recently discovered element, samarium, were contaminated with an unknown element. He was able to produce reasonably pure europium in 1901. Today, europium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements.
Named after Europe. In 1890 Boisbaudran obtained basic fractions from samarium-gadolinium concentrates which had spark spectral lines not accounted for by samarium or gadolinium. These lines subsequently have been shown to belong to europium. The discovery of europium is generally credited to Demarcay, who separated the rare earth in reasonably pure form in 1901. The pure metal was not isolated until recent years.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
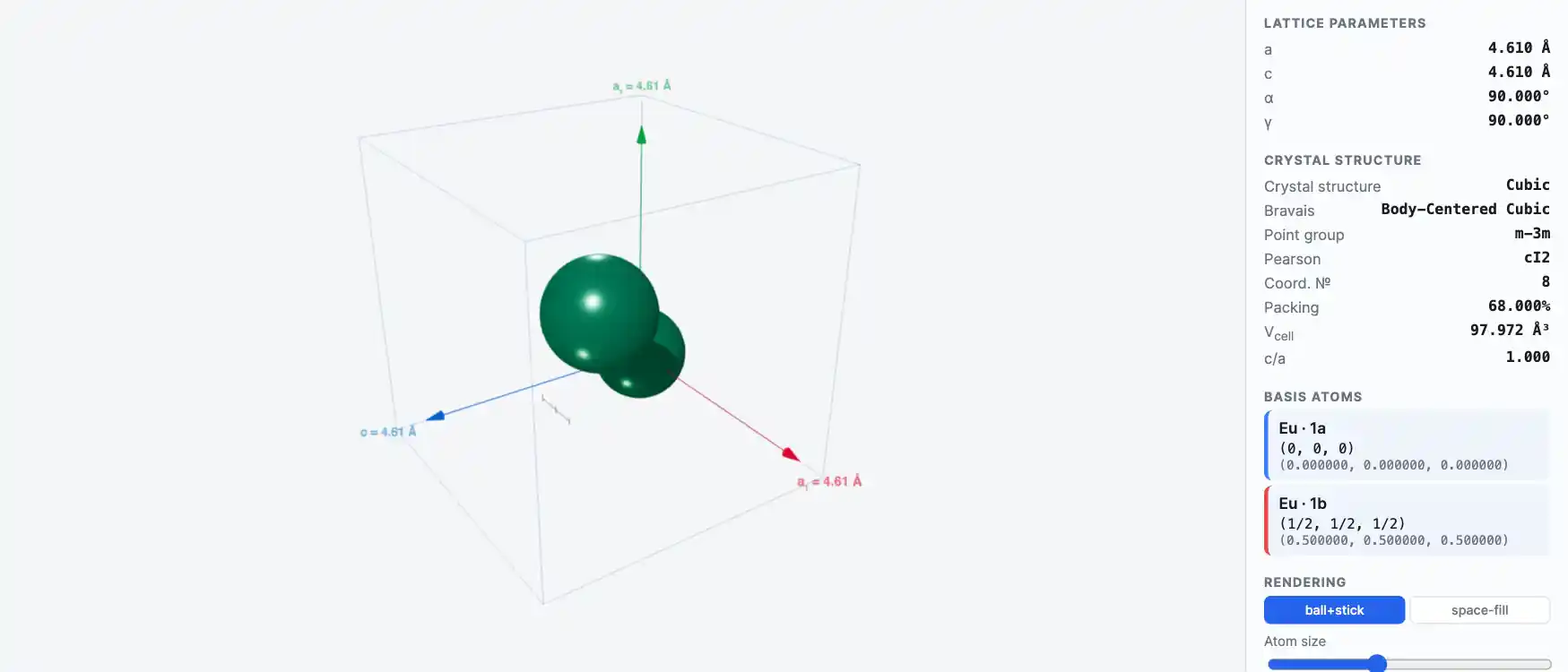
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Eu: 4f⁷ 6s²[Xe] 4f⁷ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f⁷ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 153 Radioactive | 152.921238 ± 0.0000018 | 52.1900% | 550 Py |
| 134 Radioactive | 133.9464 ± 0.00032 | N/A | 500 ms |
| 169 Radioactive | 168.961717 ± 0.000537 | N/A | 420 ms |
| 133 Radioactive | 132.94929 ± 0.00032 | N/A | 200 ms |
| 168 Radioactive | 167.957863 ± 0.000429 | N/A | 200 ms |
Phase / State
Reason: 796.9 °C below melting point (821.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 63 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Eu I | 0 | 350 | 152 | 343 |
| Eu II | +1 | 218 | 13 | 13 |
| Eu III | +2 | 229 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Eu I | 0 | 592 |
| Eu II | +1 | 163 |
| Eu III | +2 | 118 |
| Eu IV | +3 | 13 |
| Eu V | +4 | 2 |
| Eu VI | +5 | 2 |
| Eu VII | +6 | 2 |
| Eu VIII | +7 | 2 |
| Eu IX | +8 | 2 |
| Eu X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 117 pm |
| +2 | 7 | N/A | 120 pm |
| +2 | 8 | N/A | 125 pm |
| +2 | 9 | N/A | 130 pm |
| +2 | 10 | N/A | 135 pm |
| +3 | 6 | N/A | 94.69999999999999 pm |
| +3 | 7 | N/A | 101 pm |
| +3 | 8 | N/A | 106.60000000000001 pm |
| +3 | 9 | N/A | 112.00000000000001 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 153 Radioactive | 152.921238 ± 0.0000018 | 52.1900% ± 0.0600% | 550 Py | IS =52.19±0.6% | |
| 134 Radioactive | 133.9464 ± 0.00032 | N/A | 500 ms | β+ =100%β+p =? | |
| 169 Radioactive | 168.961717 ± 0.000537 | N/A | 420 ms | β- ? | |
| 133 Radioactive | 132.94929 ± 0.00032 | N/A | 200 ms | β+ ?β+p ? | |
| 168 Radioactive | 167.957863 ± 0.000429 | N/A | 200 ms | β- =100%β-n ? |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1095.15 K |
| Boiling point | 1802.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.2391 |
| 2 | p | 4.282 |
| 2 | s | 16.5292 |
| 3 | d | 13.7472 |
| 3 | p | 19.716 |
| 3 | s | 20.1318 |
| 4 | d | 34.0592 |
| 4 | f | 38.68 |
| 4 | p | 31.1252 |
| 4 | s | 30.132 |
Crystal Radii Detail (9)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 131 | ||
| 2 | VII | 134 | ||
| 2 | VIII | 139 | ||
| 2 | IX | 144 | ||
| 2 | X | 149 | ||
| 3 | VI | 108.7 | from r^3 vs V plots, | |
| 3 | VII | 115 | ||
| 3 | VIII | 120.6 | from r^3 vs V plots, | |
| 3 | IX | 126 | from r^3 vs V plots, |
Isotope Decay Modes (63)
| Isotope | Mode | Intensity |
|---|---|---|
| 130 | p | 100% |
| 130 | B+ | — |
| 130 | B+p | — |
| 131 | p | 89% |
| 131 | B+ | — |
| 131 | B+p | — |
| 132 | B+ | — |
| 132 | B+p | — |
| 132 | p | 0% |
| 133 | B+ | — |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.18583 |
| 10.1617 | — | 0.19489 |
| 10.3261 | — | 0.20439 |
| 10.4931 | — | 0.21435 |
| 10.6628 | — | 0.22479 |
| 10.8353 | — | 0.23529 |
| 11.0106 | — | 0.24598 |
| 11.1886 | — | 0.25716 |
| 11.3696 | — | 0.26817 |
| 11.5535 | — | 0.27854 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.0 milligrams per kilogram
References (1)
- [5] Europium https://education.jlab.org/itselemental/ele063.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.3×10-7 milligrams per liter
References (1)
- [5] Europium https://education.jlab.org/itselemental/ele063.html
Sources
Sources of this element.
Europium has been identified spectroscopically in the sun and certain stars. Seventeen isotopes are now recognized. Europium isotopes are good neutron absorbers and are being studied for use in nuclear control applications.
References (1)
- [6] Europium https://periodic.lanl.gov/63.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Europium is now prepared by mixing Eu2O3 with a 10%-excess of lanthanum metal and heating the mixture in a tantalum crucible under high vacuum. The element is collected as a silvery-white metallic deposit on the walls of the crucible.
References (1)
- [6] Europium https://periodic.lanl.gov/63.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Europium.
The element property data was retrieved from publications.