Gadolinium (Gd)
lanthanideSolid
Standard Atomic Weight
157.25 uElectron configuration
[Xe] 6s2 4f7 5d1Melting point
1312.85 °C (1586 K)Boiling point
3272.85 °C (3546 K)Density
7900 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.2Ionization energy (1st)
Discovery year
1886Atomic radius
180 pmDetails
Gadolinium is a silvery lanthanide metal and one of the middle rare-earth elements. Its chemistry is dominated by the +3 oxidation state, but its seven unpaired 4f electrons give it unusually strong magnetic behavior for a rare-earth element. Natural gadolinium is a mixture of stable isotopes, with ¹⁵²Gd very long-lived and weakly radioactive. The element is technologically important in magnetic materials, neutron absorption, phosphors, and medical contrast agents.
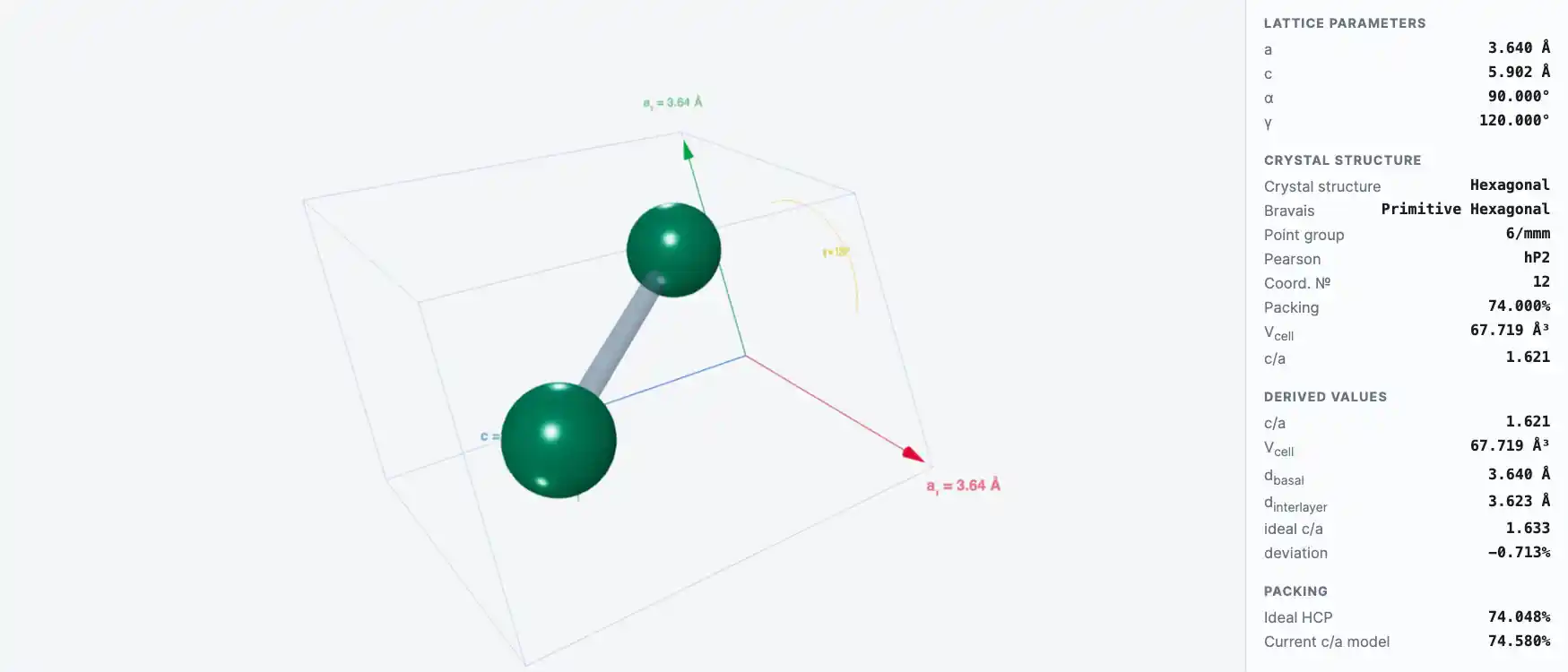
As with other related rare-earth metals, gadolinium is silvery white, has a metallic luster, and is malleable and ductile. At room temperature, gadolinium crystallizes in the hexagonal, close-packed alpha form. Upon heating to 1235°C, alpha gadolinium transforms into the beta form, which has a body-centered cubic structure.
The metal is relatively stable in dry air, but tarnishes in moist air and forms a loosely adhering oxide film which falls off and exposes more surface to oxidation. The metal reacts slowly with water and is soluble in dilute acid.
Gadolinium has the highest thermal neutron capture cross-section of any known element (49,000 barns).
The name derives from the mineral gadolinite, in which it was found, and that had been named for the Finnish rare earth chemist Johan Gadolin. Gadolinium was discovered by the Swiss chemist Jean-Charles Galissard de Marignac in 1886, who produced a white oxide in a samarskite mineral. In 1886, the French chemist Paul-Emile Lecoq de Boisbaudran gave the name gadolinium.
Spectroscopic evidence for the existence of gadolinium was first observed by the Swiss chemist Jean Charles Galissard de Marignac in the minerals didymia and gadolinite ((Ce, La, Nd, Y)2FeBe2Si2O10) in 1880. Today, gadolinium is primarily obtained from the minerals monazite ((Ce, La, Th, Nd, Y)PO4) and bastnasite ((Ce,La,Y)CO3F).
From gadolinite, a mineral named for Gadolin, a Finnish chemist. The rare earth metal is obtained from the mineral gadolinite. Gadolinia, the oxide of gadolinium, was separated by Marignac in 1880 and Lecoq de Boisbaudran independently isolated it from Mosander's yttria in 1886.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Gd: 4f⁷ 5d¹ 6s²[Xe] 4f⁷ 5d¹ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f⁷ 5d¹ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 154 Stable | 153.9208741 ± 0.0000017 | 2.1800% | Stable |
| 155 Stable | 154.9226305 ± 0.0000017 | 14.8000% | Stable |
| 156 Stable | 155.9221312 ± 0.0000017 | 20.4700% | Stable |
| 157 Stable | 156.9239686 ± 0.0000017 | 15.6500% | Stable |
| 158 Stable | 157.9241123 ± 0.0000017 | 24.8400% | Stable |
Phase / State
Reason: 1287.8 °C below melting point (1312.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 64 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Gd I | 0 | 371 | 16 | 19 |
| Gd II | +1 | 465 | 0 | 17 |
| Gd III | +2 | 158 | 0 | 0 |
| Gd IV | +3 | 50 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Gd I | 0 | 634 |
| Gd II | +1 | 321 |
| Gd III | +2 | 28 |
| Gd IV | +3 | 5 |
| Gd V | +4 | 2 |
| Gd VI | +5 | 2 |
| Gd VII | +6 | 2 |
| Gd VIII | +7 | 2 |
| Gd IX | +8 | 2 |
| Gd X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 93.8 pm |
| +3 | 7 | N/A | 100 pm |
| +3 | 8 | N/A | 105.3 pm |
| +3 | 9 | N/A | 110.7 pm |
Compounds
Isotopes (5)
Natural gadolinium is a mixture of seven isotopes, but 17 isotopes of gadolinium are now recognized. Although two of these, 155Gd and 157Gd, have excellent capture characteristics, they are only present naturally in low concentrations. As a result, gadolinium has a very fast burnout rate and has limited use as a nuclear control rod material.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 154 Stable | 153.9208741 ± 0.0000017 | 2.1800% ± 0.0300% | Stable | stable | |
| 155 Stable | 154.9226305 ± 0.0000017 | 14.8000% ± 0.1200% | Stable | stable | |
| 156 Stable | 155.9221312 ± 0.0000017 | 20.4700% ± 0.0900% | Stable | stable | |
| 157 Stable | 156.9239686 ± 0.0000017 | 15.6500% ± 0.0200% | Stable | stable | |
| 158 Stable | 157.9241123 ± 0.0000017 | 24.8400% ± 0.0700% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1586.15 K |
| Boiling point | 3546.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.2565 |
| 2 | p | 4.2946 |
| 2 | s | 16.783 |
| 3 | d | 13.723 |
| 3 | p | 19.8508 |
| 3 | s | 20.2903 |
| 4 | d | 34.3664 |
| 4 | f | 38.9864 |
| 4 | p | 31.3532 |
| 4 | s | 30.556 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 107.8 | from r^3 vs V plots, | |
| 3 | VII | 114 | ||
| 3 | VIII | 119.3 | from r^3 vs V plots, | |
| 3 | IX | 124.7 | from r^3 vs V plots, calculated, |
Isotope Decay Modes (57)
| Isotope | Mode | Intensity |
|---|---|---|
| 133 | B+ | — |
| 133 | B+p | — |
| 134 | B+ | — |
| 134 | B+p | — |
| 135 | B+ | 100% |
| 135 | B+p | 2% |
| 136 | B+ | — |
| 136 | B+p | — |
| 137 | B+ | 100% |
| 137 | B+p | — |
X‑ray Scattering Factors (719)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.59886 |
| 10.1152 | — | 2.63957 |
| 10.2317 | — | 2.68119 |
| 10.3496 | — | 2.72415 |
| 10.4688 | — | 2.7678 |
| 10.5894 | — | 2.81214 |
| 10.7114 | — | 2.8572 |
| 10.8348 | — | 2.90298 |
| 10.9596 | — | 2.94949 |
| 11.0859 | — | 2.99675 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
6.2 milligrams per kilogram
References (1)
- [5] Gadolinium https://education.jlab.org/itselemental/ele064.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
7×10-7 milligrams per liter
References (1)
- [5] Gadolinium https://education.jlab.org/itselemental/ele064.html
Sources
Sources of this element.
Gadolinium is found in several other minerals, including monazite and bastnasite, both of which are commercially important. With the development of ion-exchange and solvent extraction techniques, the availability and prices of gadolinium and the other rare-earth metals have greatly improved. The metal can be prepared by the reduction of the anhydrous fluoride with metallic calcium.
References (1)
- [6] Gadolinium https://periodic.lanl.gov/64.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Gadolinium.
The element property data was retrieved from publications.