Lutetium (Lu)
lanthanideSolid
Standard Atomic Weight
174.9668 uElectron configuration
[Xe] 6s2 4f14 5d1Melting point
1662.85 °C (1936 K)Boiling point
3401.85 °C (3675 K)Density
9840 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.27Ionization energy (1st)
Discovery year
1907Atomic radius
175 pmDetails
Lutetium is a dense, silvery lanthanide and the last element of the 4f series. In chemistry it is almost exclusively trivalent, with a filled 4f shell in Lu³⁺ and a relatively small ionic radius compared with other lanthanides. It occurs with the rare-earth elements in minerals such as monazite and xenotime, but is one of the least abundant lanthanides. Its main technological value lies in specialized scintillators, catalysts, and medical radioisotopes rather than in bulk structural use.
Lutetium occurs in very small amounts in nearly all minerals containing yttrium, and is present in monazite to the extent of about 0.003%, which is a commercial source. The pure metal has been isolated only in recent years and is one of the most difficult to prepare. It can be prepared by the reduction of anhydrous LuCl3 or LuF3 by an alkali or alkaline earth metal. The metal is silvery white and relatively stable in air. 176Lu occurs naturally (2.6%) with 175Lu (97.4%). It is radioactive with a half-life of about 3 x 1010 years.
The name derives from Lutetia, the ancient name for the city of Paris. The discovery of lutetium is credited to the French chemist Georges Urbain in 1907 although it had been separated earlier and independently by the Austrian chemist Carl Auer (Baron von Welsbach) from an ytterbium sample.
Von Welsbach had named the element cassiopeium after the constellation Cassiopeia. However, because Urbain published his results before Auer, his name for the element was adopted by IUPAC in 1949.
The mineral gadolinite ((Ce, La, Nd, Y)2FeBe2Si2O10), discovered in a quarry near the town of Ytterby, Sweden, has been the source of a great number of rare earth elements. In 1843, Carl Gustaf Mosander, a Swedish chemist, was able to separate gadolinite into three materials, which he named yttria, erbia and terbia. As might be expected considering the similarities between their names and properties, scientists soon confused erbia and terbia and, by 1877, had reversed their names. What Mosander called erbia is now called terbia and visa versa. In 1878 Jean Charles Galissard de Marignac, a Swiss chemist, discovered that erbia was itself composed of two components. One component was named ytterbia by Marignac while the other component retained the name erbia. Marignac believed that ytterbia was a compound of a new element, which he named ytterbium. Other chemists produced and experimented with ytterbium in an attempt to determine some of it's properties. Unfortunately, different scientists obtained different results from the same experiments. While some scientists believed that these inconsistent results were caused by poor procedures or faulty equipment, Georges Urbain, a French chemist, believed that ytterbium wasn't an element at all, but a mixture of two elements. In 1907, Urbain was able to separate ytterbium into two elements. Urbain named one of the elements neoytterbium (new ytterbium) and the other element lutecium. Carl Auer von Welsbach, an Austrian chemist working independently of Urbain, reached the same conclusions at nearly the same time. Welsbach chose the names albebaranium and cassiopium for these elements. Urbain was eventually credited with the discovery of the elements and won the right to name them, although chemists later changed the name neoytterbium back to ytterbium and changed the spelling of lutecium to lutetium. Today, lutetium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements.
Lutetia is the ancient name for Paris. In 1907, Urbain described a process by which Marignac's ytterbium (1879) could be separated into the two elements, ytterbium (neoytterbium) and lutetium. These elements were identical with "aldebaranium" and "cassiopeium," independently discovered at this time. The spelling of the element was changed from lutecium to lutetium in 1949.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
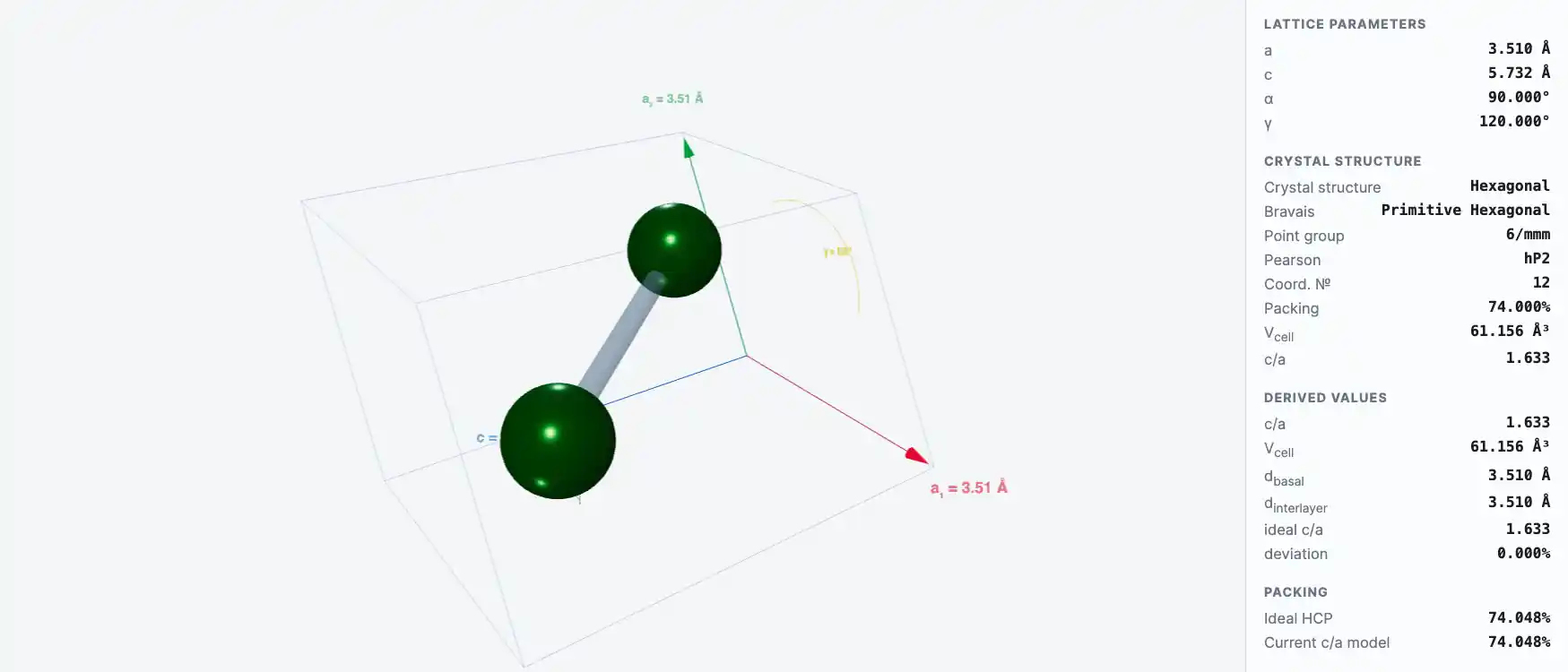
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Lu: 4f¹⁴ 5d¹ 6s²[Xe] 4f¹⁴ 5d¹ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 175 Stable | 174.9407752 ± 0.000002 | 97.4010% | Stable |
Phase / State
Reason: 1637.8 °C below melting point (1662.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 71 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Lu I | 0 | 133 | 44 | 108 |
| Lu II | +1 | 79 | 9 | 17 |
| Lu III | +2 | 64 | 0 | 0 |
| Lu IV | +3 | 100 | 0 | 0 |
| Lu V | +4 | 64 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Lu I | 0 | 234 |
| Lu II | +1 | 40 |
| Lu III | +2 | 29 |
| Lu IV | +3 | 62 |
| Lu V | +4 | 40 |
| Lu VI | +5 | 2 |
| Lu VII | +6 | 2 |
| Lu VIII | +7 | 2 |
| Lu IX | +8 | 2 |
| Lu X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 86.1 pm |
| +3 | 8 | N/A | 97.7 pm |
| +3 | 9 | N/A | 103.2 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 175 Stable | 174.9407752 ± 0.000002 | 97.4010% ± 0.0130% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1936.15 K |
| Boiling point | 3675.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.3805 |
| 2 | p | 4.389 |
| 2 | s | 18.5502 |
| 3 | d | 13.5812 |
| 3 | p | 20.8337 |
| 3 | s | 21.4655 |
| 4 | d | 35.7108 |
| 4 | f | 40.0688 |
| 4 | p | 33.8096 |
| 4 | s | 32.7308 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 100.1 | from r^3 vs V plots, | |
| 3 | VIII | 111.7 | from r^3 vs V plots, | |
| 3 | IX | 117.2 | from r^3 vs V plots, |
Isotope Decay Modes (53)
| Isotope | Mode | Intensity |
|---|---|---|
| 150 | p | 100% |
| 150 | B+ | — |
| 151 | p | — |
| 151 | B+ | — |
| 152 | B+ | 100% |
| 152 | B+p | 15% |
| 153 | A | — |
| 153 | B+ | — |
| 153 | p | 0% |
| 154 | B+ | — |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.67493 |
| 10.1617 | — | 1.63824 |
| 10.3261 | — | 1.60236 |
| 10.4931 | — | 1.56726 |
| 10.6628 | — | 1.53293 |
| 10.8353 | — | 1.49935 |
| 11.0106 | — | 1.46651 |
| 11.1886 | — | 1.43538 |
| 11.3696 | — | 1.42424 |
| 11.5535 | — | 1.41319 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8×10-1 milligrams per kilogram
References (1)
- [5] Lutetium https://education.jlab.org/itselemental/ele071.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.5×10-7 milligrams per liter
References (1)
- [5] Lutetium https://education.jlab.org/itselemental/ele071.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Lutetium.
The element property data was retrieved from publications.