Hafnium (Hf)
transition-metalSolid
Standard Atomic Weight
178.49 uElectron configuration
[Xe] 6s2 4f14 5d2Melting point
2232.85 °C (2506 K)Boiling point
4602.85 °C (4876 K)Density
1.330000e+4 kg/m³Oxidation states
−2, 0, +1, +2, +3, +4Electronegativity (Pauling)
1.3Ionization energy (1st)
Discovery year
1911Atomic radius
155 pmDetails
Hafnium is a dense, corrosion-resistant transition metal in group 4, chemically close to zirconium. It occurs almost entirely with zirconium minerals and is difficult to separate because the two elements have similar ionic sizes and chemistry. A defining technological feature is its very large thermal-neutron absorption cross section, which contrasts with zirconium's low absorption and makes high-purity separation important for nuclear applications.
Hafnium is a ductile metal with a brilliant silver luster. Its properties are considerably influenced by presence of zirconium impurities. Of all the elements, zirconium and hafnium are two of the most difficult to separate. Although their chemistry is almost identical, the density of zirconium is about half of hafnium. Very pure hafnium has been produced, with zirconium being the major impurity.
Hafnium has been successfully alloyed with iron, titanium, niobium, tantalum, and other metals. Hafnium carbide is the most refractory binary composition known, and the nitride is the most refractory of all known metal nitrides (m.p. 3310C). At 700 degrees C hafnium rapidly absorbs hydrogen to form the composition HfH1.86.
Hafnium is resistant to concentrated alkalis, but at elevated temperatures reacts with oxygen, nitrogen, carbon, boron, sulfur, and silicon. Halogens react directly to form tetrahalides.
The name derives from the Latin hafnia for Copenhagen. An element named celtium was erroneously claimed to have been discovered in 1911 by the French chemist Georges Urbain in rare earth samples, until the Danish physicist Niels Bohr, predicted hafnium's properties using his theory of electronic configuration of the elements. Bohr argued that hafnium would not be a rare earth element, but would be found in zirconium ore. Hafnium was discovered by the Dutch physicist Dirk Coster and the Hungarian physicist George von Hevesy in 1923, while working at Bohr's Institute in Copenhagen.
Hafnium was discovered by Dirk Coster, a Danish chemist, and George Charles de Hevesy, a Hungarian chemist, in 1923. They used a method known as X-ray spectroscopy to study the arrangement of the outer electrons of atoms in samples of zirconium ore. The electron structure of hafnium had been predicted by Niels Bohr and Coster and Hevesy found a pattern that matched. Hafnium is difficult to separate from zirconium and is present in all of its ores. It is obtained with the same methods used to extract zirconium.
From Hafinia, the Latin name for Copenhagen. Many years before its discovery in 1923 (credited to D. Coster and G. von Hevesey), Hafnium was thought to be present in various minerals and concentrations. On the basis of the Bohr theory, the new element was expected to be associated with zirconium.
It was finally identified in zircon from Norway, by means of X-ray spectroscope analysis. It was named in honor of the city in which the discovery was made. Most zirconium minerals contain 1 to 5 percent hafnium.
It was originally separated from zirconium by repeated recrystallization of the double ammonium or potassium fluorides by von Hevesey and Jantzen. Metallic hafnium was first prepared by van Arkel and deBoer by passing the vapor of the tetraiodide over a heated tungsten filament. Almost all hafnium metal now produced is made by reducing the tetrachloride with magnesium or with sodium (Kroll Process).
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
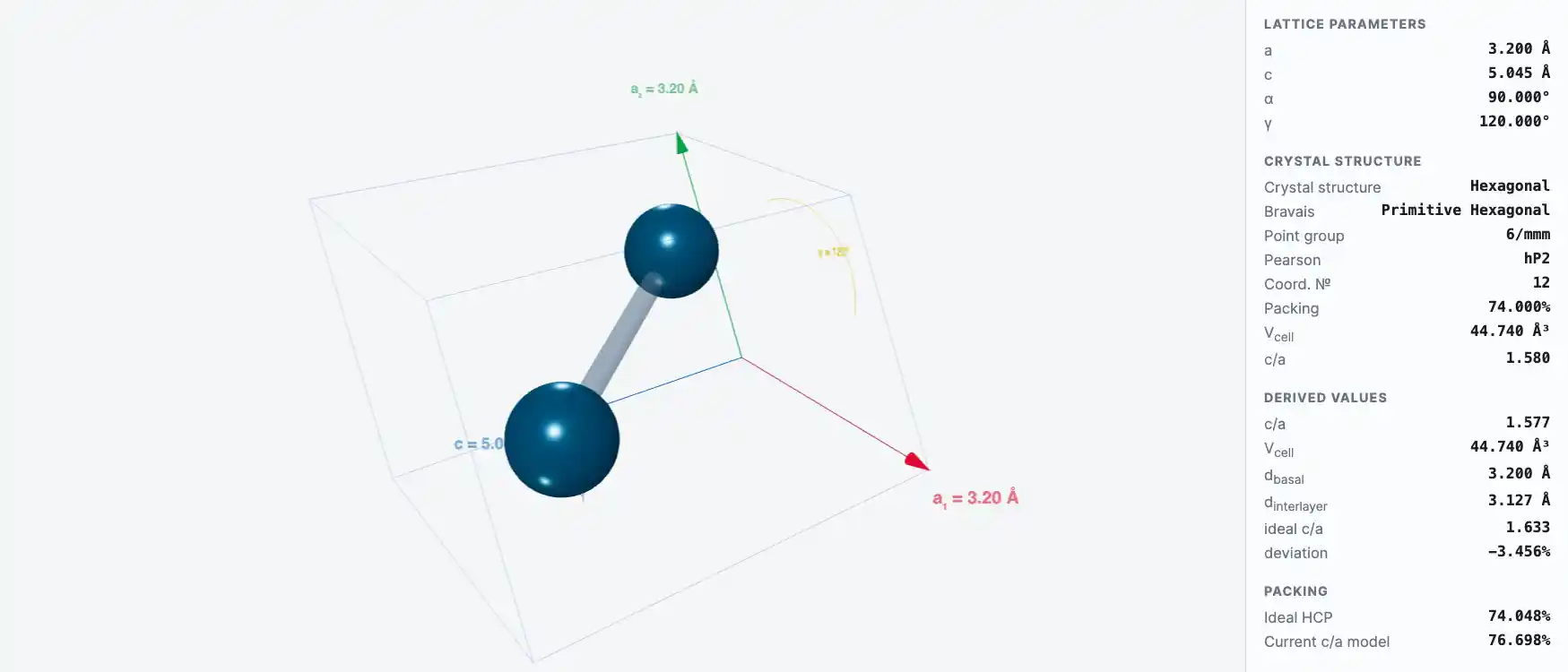
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Hf: 4f¹⁴ 5d² 6s²[Xe] 4f¹⁴ 5d² 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d² 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 176 Stable | 175.9414076 ± 0.0000022 | 5.2600% | Stable |
| 178 Stable | 177.9437058 ± 0.000002 | 27.2800% | Stable |
| 179 Stable | 178.9458232 ± 0.000002 | 13.6200% | Stable |
| 180 Stable | 179.946557 ± 0.000002 | 35.0800% | Stable |
Phase / State
Reason: 2207.8 °C below melting point (2232.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 72 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Hf I | 0 | 5341 | 187 | 3821 |
| Hf II | +1 | 218 | 2 | 10 |
| Hf III | +2 | 37 | 0 | 0 |
| Hf IV | +3 | 27 | 0 | 0 |
| Hf V | +4 | 82 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Hf I | 0 | 333 |
| Hf II | +1 | 125 |
| Hf III | +2 | 2 |
| Hf IV | +3 | 2 |
| Hf V | +4 | 2 |
| Hf VI | +5 | 2 |
| Hf VII | +6 | 2 |
| Hf VIII | +7 | 2 |
| Hf IX | +8 | 2 |
| Hf X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 4 | N/A | 57.99999999999999 pm |
| +4 | 6 | N/A | 71 pm |
| +4 | 7 | N/A | 76 pm |
| +4 | 8 | N/A | 83 pm |
Compounds
Isotopes (4)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 176 Stable | 175.9414076 ± 0.0000022 | 5.2600% ± 0.0700% | Stable | stable | |
| 178 Stable | 177.9437058 ± 0.000002 | 27.2800% ± 0.0700% | Stable | stable | |
| 179 Stable | 178.9458232 ± 0.000002 | 13.6200% ± 0.0200% | Stable | stable | |
| 180 Stable | 179.946557 ± 0.000002 | 35.0800% ± 0.1600% | Stable | stable |
Spectral Lines
Showing 50 of 1890 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 417.433998 nm | 48000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 380.03629 nm | 36000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 382.072307 nm | 34000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3F* | Measured | NIST | |
| 723.71003 nm | 34000 | Hf I | emission | 5d2.6s2 a 3F → 5d.6s2.(a 2D).6p z 3D* | Measured | NIST | |
| 384.917811 nm | 32000 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3F).6s.(a 2F).6p y 1D* | Measured | NIST | |
| 713.1807 nm | 32000 | Hf I | emission | 5d2.6s2 a 3F → 5d.6s2.(a 2D).6p z 3D* | Measured | NIST | |
| 389.993003 nm | 29000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 395.181289 nm | 26000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3F* | Measured | NIST | |
| 385.830632 nm | 25000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3D* | Measured | NIST | |
| 724.0873 nm | 21000 | Hf I | emission | 5d2.6s2 a 3F → 5d.6s2.(a 2D).6p z 3D* | Measured | NIST | |
| 393.137246 nm | 19000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3D* | Measured | NIST | |
| 480.049829 nm | 17000 | Hf I | emission | 5d2.6s2 a 1D → 5d.6s2.(a 2D).6p z 1P* | Measured | NIST | |
| 397.347912 nm | 15000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 706.38474 nm | 15000 | Hf I | emission | 5d2.6s2 a 1D → 5d.6s2.(a 2D).6p z 3P* | Measured | NIST | |
| 381.177553 nm | 14000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 3G* | Measured | NIST | |
| 555.06011 nm | 14000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5G* | Measured | NIST | |
| 555.211884 nm | 14000 | Hf I | emission | 5d2.6s2 a 1D → 5d.6s2.(a 2D).6p z 1F* | Measured | NIST | |
| 456.593715 nm | 13000 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3P).6s.(a 4P).6p y 5D* | Measured | NIST | |
| 435.630591 nm | 12000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 445.734411 nm | 12000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5F* | Measured | NIST | |
| 459.87979 nm | 12000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5F* | Measured | NIST | |
| 462.086529 nm | 12000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5F* | Measured | NIST | |
| 465.518924 nm | 12000 | Hf I | emission | 5d2.6s2 a 3P → 5d2.(a 3P).6s.(a 4P).6p z 3S* | Measured | NIST | |
| 380.044548 nm | 11000 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3P).6s.(a 4P).6p z 5S* | Measured | NIST | |
| 429.477692 nm | 11000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 681.89395 nm | 11000 | Hf I | emission | 5d2.6s2 a 3P → 5d.6s2.(a 2D).6p z 1F* | Measured | NIST | |
| 396.799621 nm | 10000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 406.28356 nm | 10000 | Hf I | emission | 5d.6s2.(a 2D).6p z 1D* → 3512 | Measured | NIST | |
| 497.525232 nm | 10000 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5F* | Measured | NIST | |
| 454.093108 nm | 8400 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3F).6s.(a 4F).6p y 3F* | Measured | NIST | |
| 443.80364 nm | 8300 | Hf I | emission | 5d2.6s2 a 3P → 5d2.(a 3P).6s.(a 4P).6p z 3S* | Measured | NIST | |
| 446.117576 nm | 8300 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3P).6s.(a 4P).6p z 3S* | Measured | NIST | |
| 459.891547 nm | 8300 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5D* | Measured | NIST | |
| 408.33549 nm | 8000 | Hf I | emission | 5d.6s2.(a 2D).6p z 1D* → 3499 | Measured | NIST | |
| 571.91718 nm | 7300 | Hf I | emission | 5d2.6s2 a 3P → 5d.6s2.(a 2D).6p z 1P* | Measured | NIST | |
| 403.225898 nm | 7200 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3F* | Measured | NIST | |
| 478.27405 nm | 7100 | Hf I | emission | 5d.6s2.(a 2D).6p z 3F* → 6p2.(3P).5d.(2D).6s c 3D | Measured | NIST | |
| 383.001314 nm | 6700 | Hf I | emission | 5d3.(b 4F).6s a 5F → 5d2.(b 1D).6s.(b 2D).6p v 3F* | Measured | NIST | |
| 504.743848 nm | 6500 | Hf I | emission | 5d2.6s2 a 3P → 5d2.(a 3F).6s.(a 4F).6p y 3D* | Measured | NIST | |
| 485.92338 nm | 6400 | Hf I | emission | 5d.6s2.(a 2D).6p z 3F* → 3512 | Measured | NIST | |
| 386.09058 nm | 6300 | Hf I | emission | 5d2.6s2 a 3P → 5d2.(a 3F).6s.(a 2F).6p y 1F* | Measured | NIST | |
| 441.790242 nm | 6200 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3F).6s.(a 4F).6p y 3D* | Measured | NIST | |
| 388.935622 nm | 5900 | Hf I | emission | 5d2.6s2 a 1D → 5d2.(a 3P).6s.(a 4P).6p y 5D* | Measured | NIST | |
| 469.90048 nm | 5900 | Hf I | emission | 5d2.(a 3F).6s.(a 4F).6p z 5G* → 5d2.(3F).6s.(a 4F).7s b 5F | Measured | NIST | |
| 678.92714 nm | 5900 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5G* | Measured | NIST | |
| 433.027751 nm | 5800 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p y 3F* | Measured | NIST | |
| 410.65431 nm | 5600 | Hf I | emission | 5d2.6s2 a 1G → 5d2.(a 3F).6s.(a 2F).6p y 1F* | Measured | NIST | |
| 426.34428 nm | 5400 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5F* | Measured | NIST | |
| 477.37157 nm | 5400 | Hf I | emission | 5d2.6s2 a 3P → 5d.6s2.(a 2D).6p z 1P* | Measured | NIST | |
| 590.29382 nm | 5400 | Hf I | emission | 5d2.6s2 a 3F → 5d2.(a 3F).6s.(a 4F).6p z 5G* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 2506.15 K |
| Boiling point | 4873.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.3984 |
| 2 | p | 4.4012 |
| 2 | s | 18.8102 |
| 3 | d | 13.5702 |
| 3 | p | 21.0168 |
| 3 | s | 21.6885 |
| 4 | d | 36.476 |
| 4 | f | 39.7904 |
| 4 | p | 34.0704 |
| 4 | s | 33.0228 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | IV | 72 | from r^3 vs V plots, | |
| 4 | VI | 85 | from r^3 vs V plots, | |
| 4 | VII | 90 | ||
| 4 | VIII | 97 |
Isotope Decay Modes (46)
| Isotope | Mode | Intensity |
|---|---|---|
| 153 | B+ | — |
| 154 | B+ | 100% |
| 154 | A | 0% |
| 155 | B+ | 100% |
| 155 | A | — |
| 156 | A | 100% |
| 156 | B+ | — |
| 157 | A | 94% |
| 157 | B+ | 14% |
| 158 | B+ | 55.7% |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.62338 |
| 10.1617 | — | 2.71485 |
| 10.3261 | — | 2.80951 |
| 10.4931 | — | 2.90326 |
| 10.6628 | — | 2.98247 |
| 10.8353 | — | 3.06384 |
| 11.0106 | — | 3.14744 |
| 11.1886 | — | 3.21346 |
| 11.3696 | — | 3.27509 |
| 11.5535 | — | 3.33789 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.0 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
7×10-6 milligrams per liter
References (1)
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Hafnium.
The element property data was retrieved from publications.