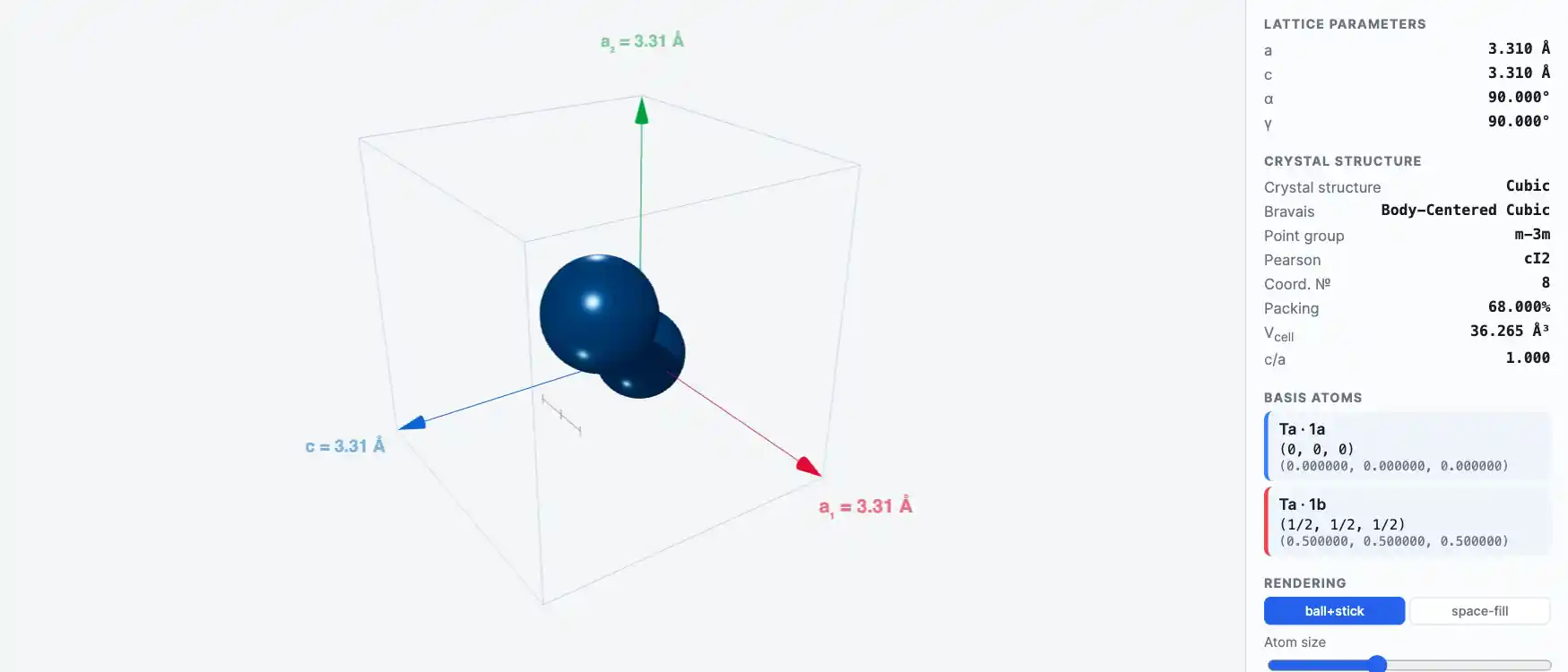
Tantalum (Ta)
transition-metalSolid
Standard Atomic Weight
180.94788 uElectron configuration
[Xe] 6s2 4f14 5d3Melting point
3016.85 °C (3290 K)Boiling point
5457.85 °C (5731 K)Density
1.640000e+4 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
1.5Ionization energy (1st)
Discovery year
1802Atomic radius
145 pmDetails
Tantalum is a dense, refractory transition metal in group 5, closely associated geologically and chemically with niobium. It is noted for exceptional resistance to corrosion, a very high melting point, and the stable, high-permittivity oxide film that forms on its surface. Most natural tantalum is ¹⁸¹Ta, with a small contribution from the long-lived nuclear isomer ¹⁸⁰ᵐTa. Its chemistry is dominated by the +5 oxidation state.
Tantalum is a gray, heavy, and very hard metal. When pure, it is ductile and can be drawn into fine wire, which is used as a filament for evaporating metals such as aluminum. Tantalum is almost completely immune to chemical attack at temperatures below 150°C, and is attacked only by hydrofluoric acid, acidic solutions containing the fluoride ion, and free sulfur trioxide. Alkalis attack it only slowly. At high temperatures, tantalum becomes much more reactive. The element has a melting point exceeded only by tungsten and rhenium. Tantalum is used to make a variety of alloys with desirable properties such as high melting point, high strength, good ductility, etc. Tantalum has a good "gettering" ability at high temperatures, and tantalum oxide films are stable and have good rectifying and dielectric properties.
The name derives from the Greek mythological character Tantalus who was banished to Hades, the region of lost souls where he was placed up to his chin in water, which receded whenever he tried to drink it, and under branches of fruit, which drew back whenever he tried to pick their fruit. This name was selected because of the insolubility of tantalum in acids; thus, when placed in the midst of acids, it is incapable of taking any of them up. Tantalum was discovered by the Swedish chemist and mineralogist Anders- Gustav Ekeberg in 1802.
Tantalum was discovered by Anders Gustaf Ekenberg, a Swedish chemist, in 1802 in minerals obtained from Ytterby, Sweden. Many scientists believed that he had only discovered an allotrope of niobium, an element that is chemically similar to tantalum. The issue was finally settled in 1866 when, Jean Charles Galissard de Marignac, a Swiss chemist, proved that tantalum and niobium were two distinct elements. The first relatively pure samples of tantalum were first produced in 1907. Today, tantalum is primarily obtained from the minerals columbite ((Fe, Mn, Mg)(Nb, Ta)2O6), tantalite ((Fe, Mn)(Ta, Nb)2O6) and euxenite ((Y, Ca, Er, La, Ce, U, Th)(Nb, Ta, Ti)2O6).
Named after Tantalos, a Greek a mythological character, father of Niobe. Discovered in 1802 by Ekeberg, but many chemists thought niobium and tantalum were identical elements until Rowe in 1844, and Marignac, in 1866, showed that niobic and tantalic acids were two different acids. The early investigators only isolated the impure metal. The first relatively pure ductile tantalum was produced by von Bolton in 1903. Tantalum occurs principally in the mineral columbite-tantalite.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ta: 4f¹⁴ 5d³ 6s²[Xe] 4f¹⁴ 5d³ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d³ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 181 Stable | 180.9479958 ± 0.000002 | 99.9880% | Stable |
Phase / State
Reason: 2991.8 °C below melting point (3016.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 73 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ta I | 0 | 526 | 200 | 510 |
| Ta II | +1 | 141 | 0 | 13 |
| Ta IV | +3 | 83 | 0 | 0 |
| Ta V | +4 | 12 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ta I | 0 | 301 |
| Ta II | +1 | 134 |
| Ta III | +2 | 2 |
| Ta IV | +3 | 2 |
| Ta V | +4 | 2 |
| Ta VI | +5 | 2 |
| Ta VII | +6 | 2 |
| Ta VIII | +7 | 2 |
| Ta IX | +8 | 2 |
| Ta X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 72 pm |
| +4 | 6 | N/A | 68 pm |
| +5 | 6 | N/A | 64 pm |
| +5 | 7 | N/A | 69 pm |
| +5 | 8 | N/A | 74 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 181 Stable | 180.9479958 ± 0.000002 | 99.9880% ± 0.0003% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 3290.15 K |
| Boiling point | 5728.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.4163 |
| 2 | p | 4.4136 |
| 2 | s | 19.0702 |
| 3 | d | 13.5589 |
| 3 | p | 21.1996 |
| 3 | s | 21.9085 |
| 4 | d | 36.676 |
| 4 | f | 39.5296 |
| 4 | p | 34.2652 |
| 4 | s | 33.2412 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 86 | estimated, | |
| 4 | VI | 82 | estimated, | |
| 5 | VI | 78 | ||
| 5 | VII | 83 | ||
| 5 | VIII | 88 |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 155 | p | 100% |
| 156 | p | 71% |
| 156 | B+ | 29% |
| 157 | A | 96.6% |
| 157 | p | 3.4% |
| 157 | B+ | — |
| 158 | A | 100% |
| 158 | B+ | — |
| 159 | B+ | 66% |
| 159 | A | 34% |
X‑ray Scattering Factors (716)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.16064 |
| 10.1152 | — | 3.23709 |
| 10.2317 | — | 3.31539 |
| 10.3496 | — | 3.39558 |
| 10.4688 | — | 3.47772 |
| 10.5894 | — | 3.56683 |
| 10.7114 | — | 3.65875 |
| 10.8348 | — | 3.75304 |
| 10.9596 | — | 3.84976 |
| 11.0859 | — | 3.94897 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.0 milligrams per kilogram
References (1)
- [5] Tantalum https://education.jlab.org/itselemental/ele073.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-6 milligrams per liter
References (1)
- [5] Tantalum https://education.jlab.org/itselemental/ele073.html
Sources
Sources of this element.
Tantalum ores are found in Australia, Brazil, Mozambique, Thailand, Portugal, Nigeria, Zaire, and Canada.
References (1)
- [6] Tantalum https://periodic.lanl.gov/73.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Separation of tantalum from niobium requires several complicated steps. Several methods are used to commercially produce the element, including electrolysis of molten potassium fluorotantalate, reduction of potassium fluorotantalate with sodium, or reacting tantalum carbide with tantalum oxide. Twenty five isotopes of tantalum are known to exist. Natural tantalum contains two isotopes.
References (1)
- [6] Tantalum https://periodic.lanl.gov/73.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Tantalum.
The element property data was retrieved from publications.