Uranium (U)
actinideSolid
Standard Atomic Weight
238.02891 uElectron configuration
[Rn] 7s2 5f3 6d1Melting point
1134.85 °C (1408 K)Boiling point
4130.85 °C (4404 K)Density
1.895000e+4 kg/m³Oxidation states
−1, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
1.38Ionization energy (1st)
Discovery year
1789Atomic radius
175 pmDetails
Uranium is a dense actinide metal and the heaviest element found in substantial natural abundance on Earth. Natural uranium is dominated by ²³⁸U, with smaller amounts of fissile ²³⁵U and trace ²³⁴U. Its chemistry is strongly oxophilic and commonly involves the uranyl ion, UO₂²⁺. Uranium is technologically important as nuclear fuel and historically important in the discovery and development of radioactivity and nuclear fission.
Pure uranium is a silvery white, weakly radioactive metal, which is harder than most elements. It is malleable, ductile, slightly paramagnetic, strongly electropositive and is a poor electrical conductor. Uranium metal has very high density, being approximately 70% denser than lead, but slightly less dense than gold. Uranium metal exhibits in three crystallographic modifications: alpha > (688°C) > beta > (776°C) > gamma. Uranium is pyrophoric when finely divided. It is a little softer than steel and is attacked by cold water in a finely divided state.In air, uranium metal becomes coated with a layer of oxide. Acids dissolve the metal, forming the +3 oxidation state which oxidizes rapidly by water and air to form higher oxidation states. Uranium metal is unaffected by alkalis. Uranium metal can be prepared by reducing uranium halides with alkali or alkaline earth metals or by reducing uranium oxides by calcium, aluminum, or carbon at high temperatures. The metal can also be produced by electrolysis of KUF5 or UF4, dissolved in a molten salt mixture of CaCl2 and NaCl. High-purity uranium can be prepared by the thermal decomposition of uranium halides on a hot filament.
Uranium metal reacts with almost all nonmetallic elements and their compounds, with reactivity increasing with temperature. Hydrochloric and nitric acids dissolve uranium, but non-oxidizing acids other than hydrochloric acid attack the element very slowly. When finely divided, it can react with cold water. In air, uranium metal oxidizes and becomes coated with a dark layer of uranium oxide. Uranium forms a variety of alloys and compounds with the most important oxidation states being uranium(IV) and uranium(VI), and their two corresponding oxides are, respectively, uranium dioxide, UO2 and uranium trioxide, UO3. Besides the oxides, other Important uranium compounds include fluorides, chlorides, bromides, iodides, carbonates, hydrides, carbides, nitrides, phosphates, etc. At room temperatures, uranium hexafluoride, UF6, has a high vapor pressure, making it useful in the gaseous diffusion process used to separate the rare U-235 from the common U-238 isotope. Uranium hydrides, nitrides and carbides are relatively inertsemimetallic compounds that are minimally soluble in acids and have been used as stable fuel pellets in nuclear power reactor technology.
Uranium exists in aqueous solutions in the +3, +4, +5, and +6 oxidation states. Oxidation state +6 as the UO22+ ion (yellow in color) is the most stable state in solution. Uranium in the +5 state as the UO2+ ion is colorless, quite unstable and disproportionates (reacts with itself) to form the +6 and +4 states. The +4 state (green) is reasonably stable in solution, but the +3 state (dark green or dark red depending on the illumination source - daylight vs fluorescent light) is unstable and easily oxidizes to +4. The +4 state in near-neutral pH solutions readily hydrolyzes to form black oxy-hydroxide precipitates.
The name derives from the planet Uranus, which in Roman mythology was "Father Heaven". The German chemist Martin-Heinrich Klaproth discovered the element in 1789, following William Hershel's discovery of the planet in 1781. The metallic uranium was first isolated by the French chemist Eugène-Melchior Peligot in 1841.
Uranium was discovered by Martin Heinrich Klaproth, a German chemist, in the mineral pitchblende (primarily a mix of uranium oxides) in 1789. Although Klaproth, as well as the rest of the scientific community, believed that the substance he extracted from pitchblende was pure uranium, it was actually uranium dioxide (UO2). After noticing that 'pure' uranium reacted oddly with uranium tetrachloride (UCl4), Eugène-Melchoir Péligot, a French chemist isolated pure uranium by heating uranium dioxide with potassium in a platinum crucible. Radioactivity was first discovered in 1896 when Antoine Henri Becquerel, a French physicist, detected it from a sample of uranium. Today, uranium is obtained from uranium ores such as pitchblende, uraninite (UO2), carnotite (K2(UO2)2VO4·1-3H2O) and autunite (Ca(UO2)2(PO4)2·10H2O) as well as from phosphate rock (Ca3(PO4)2), lignite (brown coal) and monazite sand ((Ce, La, Th, Nd, Y)PO4). Since there is little demand for uranium metal, uranium is usually sold in the form of sodium diuranate (Na2U2O7·6H2O), also known as yellow cake, or triuranium octoxide (U3O8).
The use of uranium in its natural oxide form dates back to 79 A.D. when it was used as a yellow coloring agent in ceramic glazes. Yellow glass with 1% uranium oxide was found in an ancient Roman villa near Naples, Italy. In the late Middle Ages, pitchblende was extracted from the silver mines and was used as a coloring agent in the glassmaking industry. The identification of uranium as an element is generally credited to Martin H. Klaproth. While experimenting with pitchblende in 1789, he concluded that it contained a new element, which he named after the newly discovered planet Uranus (named after the Greek god of the sky or heaven). What Klaproth actually identified was not the pure element but uranium oxide. The pure metal was first isolated in 1841 by Eugène-Melchior Péligot, who reduced anhydrous uranium tetrachloride with potassium metal.
In 1896 Antoine H. Becquerel discovered that uranium exhibited invisible light or rays; it was radioactivity. In 1934 research by Enrico Fermi and others eventually led to the use of uranium fission in the first nuclear weapon used in war and later in the peaceful use of uranium as fuel in nuclear power production. An ensuing arms race during the Cold War between the United States and the Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239. The security of those weapons and their fissile material following the breakup of the Soviet Union in 1991 is an ongoing concern.
In 1972 French physicist Francis Perrin discovered ancient and no longer active prehistoric natural nuclear fission reactors in uranium ore deposits at the Oklo mine in Gabon, West Africa, collectively known as the Oklo Fossil Reactors. The ore deposit is 1.7 billion years old; at that time, uranium-235 constituted about 3% of the total uranium on Earth (0.72% today). This is high enough to permit a sustained nuclear fission chain reaction to occur, provided other supporting geologic conditions exist.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
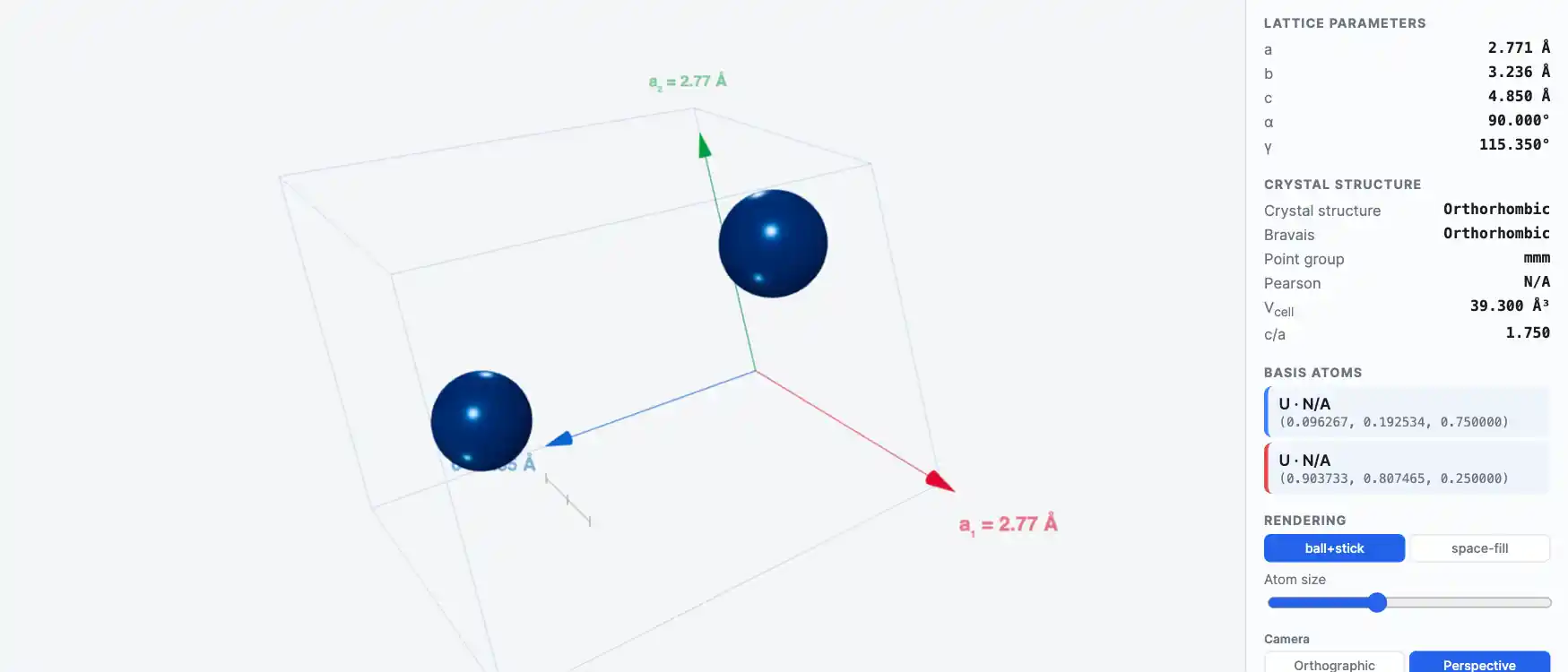
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
U: 5f³ 6d¹ 7s²[Rn] 5f³ 6d¹ 7s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p⁶ 5f³ 6d¹ 7s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 217 Radioactive | 217.02466 ± 0.00011 | N/A | 850 us |
| 235 Radioactive | 235.0439301 ± 0.0000019 | 0.7204% | 704 My |
| 221 Radioactive | 221.02628 ± 0.00011 | N/A | 660 ns |
| 224 Radioactive | 224.027605 ± 0.000027 | N/A | 396 us |
| 218 Radioactive | 218.023523 ± 0.00002 | N/A | 354 us |
Phase / State
Reason: 1109.8 °C below melting point (1134.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 92 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| U I | 0 | 216 | 57 | 0 |
| U II | +1 | 258 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| U I | 0 | 2 |
| U II | +1 | 2 |
| U III | +2 | 2 |
| U IV | +3 | 2 |
| U V | +4 | 2 |
| U VI | +5 | 2 |
| U VII | +6 | 2 |
| U VIII | +7 | 2 |
| U IX | +8 | 2 |
| U X | +9 | 2 |
Ionic Radii
Showing 10 of 14 Ionic Radii.
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 102.49999999999999 pm |
| +3 | 9 | N/A | 118.9 pm |
| +4 | 6 | N/A | 89 pm |
| +4 | 7 | N/A | 95 pm |
| +4 | 8 | N/A | 100 pm |
| +4 | 9 | N/A | 105 pm |
| +4 | 12 | N/A | 117 pm |
| +5 | 6 | N/A | 76 pm |
| +5 | 7 | N/A | 84 pm |
| +6 | 2 | N/A | 45 pm |
Compounds
Isotopes (5)
Uranium is weakly radioactive because all naturally occurring (or primordial) isotopes of uranium (238U, 235U and 234U) are unstable, with half-lives varying between 159,200 years and 4.5 billion years. There are 27 known isotopes of uranium ranging in atomic weights 217–219, 222–240 and 242, with half-lives of from billions of years to a few nanoseconds. Naturally occurring uranium consists of three major isotopes: 238U (99.28% abundance), 235U (0.71%), and 234U (0.0054%). (The US DOE has adopted the value of 0.711 as being their official percentage of 235U in natural uranium.) All three isotopes are radioactive, with small probabilities of undergoing spontaneous fission but preferentially decaying by alpha emission. The half-life of uranium-238 is about 4.47 billion years and that of uranium-235 is 704 million years, making them useful in dating the age of the Earth. It also suggests that half of the uranium that existed from the formation of the Earth has decayed to other radioactive elements and eventually to stable elements. Much of the internal heat of the earth is thought to be attributable to the decay of uranium and thorium radio-isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 217 Radioactive | 217.02466 ± 0.00011 | N/A | 850 us | α ≈100%β- ? | |
| 235 Radioactive | 235.0439301 ± 0.0000019 | 0.7204% ± 0.0006% | 704 My | IS =0.7204±0.6%α =100%SF =7e-9±0.2% | |
| 221 Radioactive | 221.02628 ± 0.00011 | N/A | 660 ns | α ≈100%β+ ? | |
| 224 Radioactive | 224.027605 ± 0.000027 | N/A | 396 us | α =100%β+ ? | |
| 218 Radioactive | 218.023523 ± 0.00002 | N/A | 354 us | α =100% |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1408.15 K |
| Boiling point | 4404.15 K |
Oxidation State Categories
Advanced Reference Data
Crystal Radii Detail (14)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 116.5 | from r^3 vs V plots, | |
| 4 | VI | 103 | ||
| 4 | VII | 109 | estimated, | |
| 4 | VIII | 114 | from r^3 vs V plots, | |
| 4 | IX | 119 | ||
| 4 | XII | 131 | estimated, | |
| 5 | VI | 90 | ||
| 5 | VII | 98 | estimated, | |
| 6 | II | 59 | ||
| 6 | IV | 66 |
Isotope Decay Modes (60)
| Isotope | Mode | Intensity |
|---|---|---|
| 215 | A | — |
| 215 | B+ | — |
| 216 | A | 100% |
| 217 | A | 100% |
| 217 | B- | — |
| 218 | A | 100% |
| 219 | A | 100% |
| 219 | B+ | — |
| 220 | A | — |
| 220 | B+ | — |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.627 |
| 10.1617 | — | 1.61282 |
| 10.3261 | — | 1.59877 |
| 10.4931 | — | 1.58416 |
| 10.6628 | — | 1.56547 |
| 10.8353 | — | 1.547 |
| 11.0106 | — | 1.52874 |
| 11.1886 | — | 1.5107 |
| 11.3696 | — | 1.49287 |
| 11.5535 | — | 1.47526 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.7 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3.2×10-3 milligrams per liter
References (1)
Sources
Sources of this element.
Uranium is the heaviest naturally-occurring element available in large quantities. The heavier “transuranic” elements are either man-made or they exist only as trace quantities in uranium ore deposits as activation products. Uranium occurs naturally in low concentrations of a few parts per million in soil, rock and water, and is commercially extracted from uranium-bearing minerals. Uranium, not as rare as once thought, is now considered to be more plentiful than mercury, antimony, silver, or cadmium, and is about as abundant as molybdenum or arsenic. It occurs in numerous natural minerals such as pitchblende, uraninite, carnotite, autunite, uranophane, and tobernite. It is also found in phosphate rocks, lignite, monazite sands, and is recovered commercially from these sources. The United States Department of Energy purchases uranium in the form of acceptable U3O8 concentrates. This incentive program has greatly increased the known uranium reserves.
References (1)
- [6] Uranium https://periodic.lanl.gov/92.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Uranium.
The element property data was retrieved from publications.