Neptunium (Np)
actinideSolid
Standard Atomic Weight
[237]Electron configuration
[Rn] 7s2 5f4 6d1Melting point
643.85 °C (917 K)Boiling point
3901.85 °C (4175 K)Density
2.025000e+4 kg/m³Oxidation states
+2, +3, +4, +5, +6, +7Electronegativity (Pauling)
1.36Ionization energy (1st)
Discovery year
1940Atomic radius
175 pmDetails
Neptunium is a radioactive actinide and the first transuranium element. It is produced mainly by neutron reactions involving uranium in nuclear reactors and occurs naturally only in minute amounts from neutron capture and decay processes in uranium ores. Its chemistry bridges uranium and plutonium, with several accessible oxidation states in solution. The isotope ²³⁷Np is the most important for handling and study because of its long half-life and availability as a reactor by-product.
Neptunium metal buttons (photo courtesy Lawrence Berkeley National Laboratory)
Neptunium was first produced by Edwin M. McMillian and Philip H. Abelson, working at the University of California, Berkeley, in 1940. They produced neptunium-239, an isotope of neptunium with a half-life of about 2.4 days, by bombarding uranium with slow moving neutrons.
Named for the planet Neptune (named after the Roman god of the sea), the next planet out from the Sun after Uranus. There were many early false reports of the discovery of neptunium. The most significant was by Enrico Fermi who believed that bombarding uranium with neutrons followed by beta decay would lead to the formation of element 93. In 1934, he bombarded uranium atoms with neutrons and reported that he had produced elements 93 and 94. As it turned out, Fermi had actually fissioned or split uranium atoms into many fragment radioisotopes. The explanation and announcement of the discovery of fission was later published by Hahn and Strassman, although it was their co-worker Lisa Meitner who had correctly interpreted the results of the experiments. In 1940, with excitement about fission reaching the University of California at Berkeley, Professor Edwin McMillan and graduate student Philip Abelson bombarded uranium with cyclotron-produced moderated (slow) neutrons, resulting not in “fission” but "fusion" of the reactants forming the new element 93, which they named "neptunium":
23892U + 10n → 23992U → 23993Np + β-
Neptunium-239 was the first transuranium element produced synthetically and the first actinide series transuranium element discovered. This isotope has a beta-decay half-life of 2.3565 days, which forms daughter product plutonium-239 with a half-life of 24,000 years.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
N/A
Reactivity
N/A
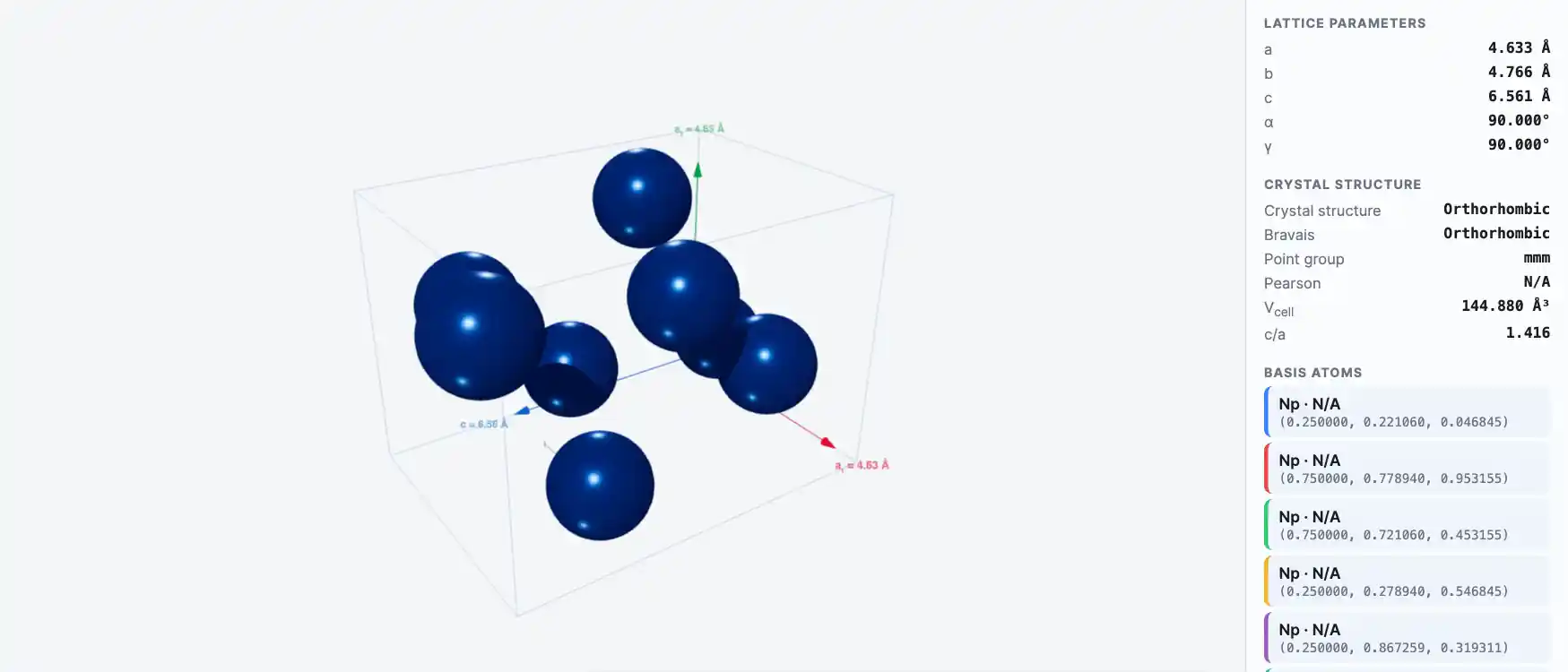
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Np: 5f⁴ 6d¹ 7s²[Rn] 5f⁴ 6d¹ 7s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p⁶ 5f⁴ 6d¹ 7s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 219 Radioactive | 219.03143 ± 0.00021 | N/A | 570 us |
| 227 Radioactive | 227.034957 ± 0.000078 | N/A | 510 ms |
| 222 Radioactive | 222.0333 ± 0.00021 | N/A | 480 ns |
| 235 Radioactive | 235.0440635 ± 0.0000021 | N/A | 396.1 days |
| 236 Radioactive | 236.04657 ± 0.000054 | N/A | 153 ky |
Phase / State
Reason: 618.9 °C below melting point (643.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 93 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Np I | 0 | 96 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Np I | 0 | 2 |
| Np II | +1 | 2 |
| Np III | +2 | 2 |
| Np IV | +3 | 2 |
| Np V | +4 | 2 |
| Np VI | +5 | 2 |
| Np VII | +6 | 2 |
| Np VIII | +7 | 2 |
| Np IX | +8 | 2 |
| Np X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 110.00000000000001 pm |
| +3 | 6 | N/A | 101 pm |
| +3 | 9 | N/A | 117.8 pm |
| +4 | 6 | N/A | 87 pm |
| +4 | 8 | N/A | 98 pm |
| +5 | 6 | N/A | 75 pm |
| +6 | 6 | N/A | 72 pm |
| +7 | 6 | N/A | 71 pm |
Compounds
Isotopes (5)
There are 25 known radioactive isotopes of neptunium ranging in atomic weights from 225 to 244 with 5 of those as metastable isotopes. The most stable are Np-237 with a half-life of 2.14 million years; Np-236 with a half-life of 154,000 years; and Np-235 with a half-life of 396 days. All of the remaining isotopes have half-lives less than 4.5 days, with most less than 50 minutes. The primary decay mode for isotopes lighter than 237Np is by electron capture with a great deal of alpha emission. The products are mostly isotopes of uranium. The primary decay mode for Np-237 is by alpha-decay forming protactinium. The primary decay mode for the isotopes heavier than Np-237 is by beta-decay, forming plutonium. Neptunium-237, after decaying to protactinium then to uranium, eventually decays to form bismuth-209 and thallium-205. Unlike most other common heavy nuclei which decay to make isotopes of lead this decay chain is known as the neptunium series.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 219 Radioactive | 219.03143 ± 0.00021 | N/A | 570 us | α =100% | |
| 227 Radioactive | 227.034957 ± 0.000078 | N/A | 510 ms | α ≈100%β+ ? | |
| 222 Radioactive | 222.0333 ± 0.00021 | N/A | 480 ns | α =100% | |
| 235 Radioactive | 235.0440635 ± 0.0000021 | N/A | 396.1 days | ε =99.99740±1.3%α =0.00260±1.3% | |
| 236 Radioactive | 236.04657 ± 0.000054 | N/A | 153 ky | ε =86.3±0.8%β- =13.5±0.8%α =0.16±0.4% |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Phase Transitions & Allotropes
| Melting point | 917.15 K |
Oxidation State Categories
Advanced Reference Data
Crystal Radii Detail (8)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 124 | ||
| 3 | VI | 115 | from r^3 vs V plots, | |
| 4 | VI | 101 | from r^3 vs V plots, | |
| 4 | VIII | 112 | from r^3 vs V plots, | |
| 5 | VI | 89 | ||
| 6 | VI | 86 | from r^3 vs V plots, | |
| 7 | VI | 85 | Ahrens (1952) ionic radius, | |
| 3 | IX | — | 131.8 |
Isotope Decay Modes (44)
| Isotope | Mode | Intensity |
|---|---|---|
| 219 | A | 100% |
| 220 | A | 100% |
| 221 | A | — |
| 222 | A | 100% |
| 223 | A | 100% |
| 224 | A | 100% |
| 225 | A | 100% |
| 225 | B+ | — |
| 226 | A | 100% |
| 226 | B+ | — |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
Not Applicable
References (1)
- [5] Neptunium https://education.jlab.org/itselemental/ele093.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Neptunium https://education.jlab.org/itselemental/ele093.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Neptunium.
The element property data was retrieved from publications.